Introduction
The constant need for high-quality foods and related raw materials has put substantial pressure on agriculture to increase the yield of essential crops like soybean, rice, cotton, wheat, and corn. It is expected that by 2050 the need for food will double, an unreachable goal under the current climate change scenario [1]. One of the major strategies to improve food production is pest, phytopathogen, and weed control, which modern agriculture has addressed using a variety of agrochemicals [2,3]. Nevertheless, herbicides can be traced from crop fields through the food distribution and consumption chain, and the indiscriminate use of pesticides due to the resistance of weeds and insects endangers the health of future generations of wildlife and humans [4-7].
Glyphosate (n-phosphonomethyl-glycine), registered as a pesticide by Monsanto Company in 1974, is the most successful and widely used herbicide worldwide, and it has been detected in fresh and processed food and human fluids[3]. The golden age of glyphosate in weed management came with the introduction of glyphosate-resistant crops such as soybean, maize, and cotton [8]. Currently, several glyphosate-based herbicides are available on the market and sold under several brand names in various countries around the world [9]. Glyphosate is a non-selective, broad-spectrum herbicide that enters the plant through the leaf system and disperses to the rest of the plant’s tissues, killing all meristems [10]. The mechanism of action of this compound involves the arresting of the biosynthesis of aromatic amino acids by inhibiting the enzyme 5-enolpyruvylshikimate-3-phosphate-synthetase (EPSPS) [11]. However, several weed species have now evolved resistance to glyphosate worldwide, which points out the implementation of new application strategies for this herbicide. It is therefore necessary to generate new technologies for weed control in agriculture [12]. In addition, the use of glyphosate has generated controversy due to its association with severe health conditions such as the increase in the probability of developing cancer, endocrine alterations, neurological disorders, and autism [13-16]. Consequently, glyphosate was reclassified in 2015 as "probably carcinogenic" in Group 2A by the International Agency for Research on Cancer [17].
The main concern about the massive use of glyphosate is its potentially detrimental effect on human health. This raises the need to establish reliable and robust analytical approaches to survey glyphosate, which can persist for several months in soil, water, and other substrates [18]. Due to glyphosate’s chemical features (amphoterism, high polarity, non-volatility, low molecular weight, and hydrophilicity), mass spectrometry has become the keystone for detection and quantification [19,20]. Both gas and liquid chromatography have been coupled to mass spectrometry to detect glyphosate successfully [18-20]. For example, glyphosate content has been determined by capillary gas chromatography with mass-selective detection, however, this technique has become tedious due to the previous column conditioning (clean up) and the need for a derivatization step, [21], which can lead to the loss of analytes of interest, content underestimation when the derivatization reaction yield is poor, low reproducibility, and variation in the detection and quantification limits [22-24]. In addition, there are reports about glyphosate determination by liquid chromatography coupled to mass [25,26], fluorescence [27] and ultraviolet detectors [28,29], where most of these, also it is required a derivatization step to facilitate the detection, which is caused by the high water solubility, low volatility, and the lack of chromophore groups resulting in time-consuming and more complex methods [30,31]. Glyphosate derivatization step can be made before or after column separation to form fluorescent-adducts to allow its detection and/or to reduce its polar characteristics and thus facilitate its chromatographic retention [32,33]. The extraction procedures used for the determination of glyphosate depend mainly on the type of matrix to be analyzed, and the most frequent approaches are solid-phase extraction, ultrasonication [20,27,34,35], agitation [27], and accelerated solvent extraction (ASE) [27,36]. However, there have been few efforts to systematically determine the best and most efficient extraction approach by comparing different protocols applied to different matrices. Most of the latest published methods related to analytical methods for the quantification of glyphosate and related compounds report the modification of protocols previously described where it is evaluated changes in pH, solvents, concentrations, derivatization steps, etc. [37-41].
The coupling of a mass spectrometry detector to a chromatographic separation system allows solving efficiently the problems of identification and quantification of metabolites. Mass spectrometry is used to identify unknown compounds and quantify known compounds and elucidate the structure and chemical properties of molecules. In principle, the mass spectrum of each compound is unique and can be used as its "chemical fingerprint". The mass spectrometry foundation is the formation of ions from organic molecules in a gas phase, and the separation according to their mass/charge ratio [42-44]. The dynamic multiple reaction monitoring (dMRM) acquisition methods in a triple quadrupole mass analyzer consist of selecting the m/z value from the molecular ion (parent ion) in the first quadrupole, breaking the molecule in the second quadrupole (collision cell-kind), and selecting an m/z from a diagnostic fragment (daughter ion) in the third quadrupole. So, this approach is highly selective and sensitive.
Given the aforementioned gaps, we evaluated different extraction protocols for glyphosate recovery, including ultrasonication, ASE, and the energized dispersive guided extraction (EDGE) approaches. Our extracts from corn flour were analyzed with a dMRM method in ultra-performance liquid chromatography coupled to a triple quadrupole mass spectrometer (UPLC-MS-QqQ). Our comparative scrutiny showed that the extraction methods tested allowed optimal recovery percentages of glyphosate-2-13C, leading to the development of more alternative analytical approaches for regulatory purposes in the agricultural and health sectors.
Experimental
Material and methods
Chemicals and standards
Glyphosate-2-13C (PubChem ID 329759134, molecular weight 170.07) and aminomethylphosphonic acid (AMPA, PubChem ID 24859469, molecular weight 111.04), PESTANAL analytical standards (purity > 98.0 %) were purchased from Sigma-Aldrich (St. Louise, MO, USA). AMPA, which is a glyphosate-related compound found in treated matrices with the herbicide, was used to validate the specificity of the method. Formic acid (99 %), water Optima LC/MS grade, and methanol CHROMASOLV LC/MS were purchased from Thermo Scientific (Fisher chemical, Waltham, Massachusetts, USA) and Honeywell Riedel-de Haën (Wabash, Indiana, Estados Unidos), respectively.
Preparation of standards
Glyphosate-2-13C and AMPA standard stock solutions were prepared at a concentration of 1 mg/mL in water LC/MS grade and stored at 4 °C in dark conditions until their use. The standard stock solutions for the calibration curve for glyphosate-2-13C and AMPA were 0.1, 0.2, 0.4, 1, 2, 4, 6, 8, 10, 12, 14, 16, 18, and 20 µM (1 mL each) for chromatographic analysis.
Glyphosate extraction systems
Three different extraction protocols were evaluated using in two cases completely automated instruments (for details see Fig.1), an Accelerated Solvent System (ASE 350, Dionex Corporation, Thermo-Scientific, Sunnyvale, California, USA), an automated Energized Dispersive Guided Extraction system (EDGE™, CEM Corporation, Charlotte, North Caroline, USA), and an ultrasonic bath (Quick Start CPXH Series, Cole-Parmer, Vernon Hills, Illinois, USA). Condition parameters were established and modified for each system according to the manual and previously reported extraction efficiencies [45-47]. Extraction methods using ASE and ultrasonication have been previously reported and were used and adapted in this work [27,48]. For ASE, samples were dispersed with 0.1 g diatomaceous earth and then placed separately in each 5 mL extraction cell. We set a single static cycle for 5 minutes at 40 °C. Methanol/water (20:80 and 50:50) with 1 % (v/v) formic acid (FA) was used as the extraction and rinse solvent. The obtained extracts were filtered and stored for further analysis. For the ultrasonic extraction, samples were transferred to a 50 mL polypropylene centrifuge tube, and methanol/water (20:80 and 50:50) with 1 % (v/v) formic acid was used as the extraction solvent. The tube was capped, shaken, and vortexed for 1 minute, and stored at -20 °C for 1 hour. Then, the sample was defrosted to continue to be sonicated in a cold bath with four 5-minutes pulses. Finally, the tube was centrifuged for 20 minutes at 4 °C and the supernatant was recovered and filtered for further analysis.
For EDGE, samples were placed in the extraction cell. G1 (glass fibre, 0.3 µm) and C9 (cellulose, 40 µm) filters were used in a stacked C9+G1+C9 configuration for extraction. Methanol/water (20:80 and 50:50) with 1 % (v/v) formic acid was used as the extraction and rinse solvent. The cycle consisted of adding 5 mL to the top and rinsing with 15 mL. The temperature was set at 40 °C with a hold time of two minutes, and the rinse step was set at 15 seconds.
Fortified sample preparation
The glyphosate-2-13C internal standard was added to corn flour (Maseca™, purchased from the local market) samples (300 mg) and homogenized to obtain fortified samples. The 20:80 methanol/water + 1 % FA samples were fortified with 34 µg of glyphosate-2-13C to obtain a final concentration of 113.33 ppm (w/w). Additionally, 50:50 methanol/water + 1 % FA samples were fortified with 68 µg of glyphosate-2-13C to obtain a final concentration of 226.66 ppm (w/w). Only for the 50:50 methanol/water + 1 % FA in the ultrasonication system, samples were fortified with 13.62 µg of glyphosate-2-13C equivalent to 45.40 ppm (w/w). To evaluate the specificity of the method, 20 µM of the standard stock solution of AMPA was prepared and added to the corn samples.
Analysis by UPLC-MS-QqQ
An Ultra-High Performance Liquid Chromatography Agilent 1290 (Agilent Technologies Santa Clara, California, USA) coupled with a triple quadrupole mass spectrometer (Agilent 6460; Serial# SG13369040-UPLC-MS-QqQ) with a dynamic multiple reaction monitoring (dMRM) method was employed. Chromatographic separation was performed in a ZORBAX Eclipse Plus C18 column (2.1 x 50 mm, 1.8 Micron, 600 Bar) with a flow rate of 0.1 mL/min. The spectrometric analyses were conducted using an electrospray ionization source in negative mode. The injected volume was 5 µL. The binary mobile phase consisted of 1 % FA in water (phase A) and 1 % FA in methanol (phase B). The initial composition consisted of 95 % A and 5 % B (v/v). This condition was held for 3 minutes, followed by linear ramping to 95 % B over 1 minute, maintained for 5 minutes. Then, the phases were returned to the initial composition over 1 minute (5 % B) and held for 5 minutes. The total chromatographic run time was 15 minutes. The dMRM method was optimized by evaluating four different transitions and three collision energies. For quantification, the glyphosate transition of m/z 169.0>151.1 was used. The chromatographic conditions used in this work were selected by reviewing the literature, considering instrumental similarities, derivatization steps, reagents, and the objective of the study [19,20,25,27,48,49]. The data were acquired using the Agilent Mass Hunter Workstation software, processed with the Agilent Mass Hunter Quantitative module to establish the calibration curves and with the Agilent Qualitative software to determine the glyphosate-2-13C and AMPA peaks (Agilent, Santa Clara, CA, USA).
Validation method of glyphosate-2-13C determination and quantification.
The validation of the method was carried out following the method validation guide for the analysis of pesticides issued by the Center of the National Reference for Pesticides and Contaminants issued by the National Service of Health and Food Quality (SENASICA) from Mexico [50]. The identity confirmation of glyphosate-2-13C was carried out with the combination of two validation parameters: selectivity and specificity. Selectivity is the aptitude of a method to qualify the compound of interest in the presence of other components of similar behaviour under the established conditions of the test and was validated by determining the presence of glyphosate-2-13C in fortified and non-fortified flour samples. For specificity, which is the aptitude of a method to respond exclusively to the analyte in the presence of similar compounds or the ability to differentiate isomers of a compound under the established conditions of the test, the presence of glyphosate-2-13C was determined in a sample fortified with AMPA (at 20 µM), which is a product of microbial degradation of glyphosate and a structurally related compound [30,51].
For quantification, it was made calibration curves for glyphosate-2-13C and AMPA with 14 concentration points (0.1, 0.2, 0.4, 1, 2, 4, 6, 8, 10, 12, 14, 16, 18, 20 µM). The limit of detection (LOD) was determined as the minimum concentration of the analyte in a sample which can be detected with a signal/noise ratio value >3 [50]. The limit of quantification (LOQ) was determined as the minimum concentration of the substance of interest that can be reliably quantified. The method’s accuracy was determined considering the Alimentarius Codex which refers to this parameter as the coincidence between the mean value obtained from a series of results and an accepted reference value [52]. For this work, this parameter was evaluated in two different ways, first by the quantification of glyphosate-2-13C at a known concentration in different samples analyzed on the same day. The second way was by establishing the recovery percentages obtained with different extraction protocols according to the following formula:
Where RM = Recovered mass (in µg) and IM = Initial mass (in µg).
The precision of the method was validated by considering the repeatability and reproducibility. Repeatability is the proximity of the results obtained with the analytical method under similar working conditions (e.g., same analyst, same instrument in a short time scale). To validate this parameter, serial analyses of samples fortified with glyphosate-2-13C were performed at the same concentration on the same day. The reproducibility parameter considers changes in the protocol, and we validated this parameter with glyphosate-2-13C determinations at a known concentration in similar working conditions but on three different days.
Statistics
We used a one-way analysis of variance (One Way-ANOVA) to determine whether there were statistically significant differences between the means of the recovery percentage among the extraction systems and solvents (methanol/water + 1% FA at 20:80 versus 50:50 v/v). When the ANOVA exhibited significant differences, we calculated all pairwise multiple comparisons using a Tukey Test. The statistical analyses were done in SigmaStat Software for Windows, Version 3.5.
Results
Optimal parameters for multiple reaction monitoring determinations
Four different transitions and three collision energies were evaluated for optimal glyphosate detection, and three different transitions and a single voltage were evaluated for AMPA as well. We carried out our spectrometric analyses in positive and negative ionization modes. However, a higher sensitivity was obtained in the negative mode. The selection criterion was the abundance of the product ion. Therefore, we selected for glyphosate-2-13C the precursor ion of m/z 169.0 [M−H]- and the product ion of 151.1. For AMPA, we selected the m/z 110.0 [M−H]- as the precursor ion (parent) and the m/z 79.0 as the product ion (daughter). The retention time obtained for glyphosate-2-13C was 1.41 minutes and for AMPA was 1.35 minutes (Fig. 2).
Standard calibration curves for glyphosate-2-13C.
We used 14 different concentration points to build calibration curves for glyphosate-2-13C and AMPA in a concentration range of 0.1-20 µM. The coefficients of determination for those curves were 0.99763831 and 0.99806048 for glyphosate-2-13C and AMPA, respectively (Fig. 3). The LOD for glyphosate-2-13C and AMPA were 0.1 and 0.2 µM, respectively (Table 1) while the LOQ was 0.2 µM for Glyphosate-2-13C µM and 1 µM for AMPA (Table 2).
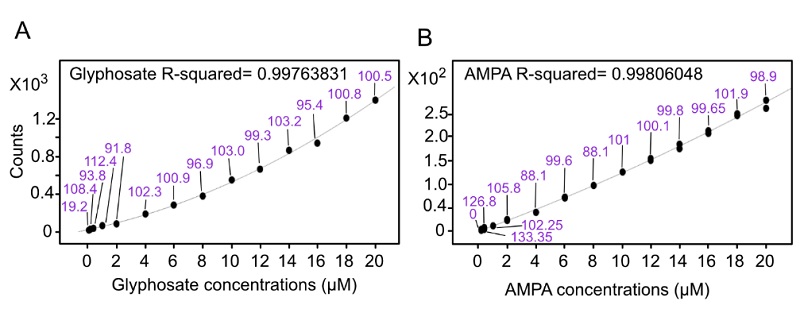
Fig. 3 Calibration curves for glyphosate (A) and AMPA (B) in a concentration range of 0.1-20 µM. Numbers in purple along the trend line specify the percent accuracy at each point.
Table 1 Determination for Limit of detection (LOD) for glyphosate -2-13C, and AMPA.
| Compound | Concentration (µM) | Signal/Noise ratio |
| Glyphosate -2-13C | 0.1 | 3.5 |
| 0.2 | 5.4 | |
| 0.4 | 7.3 | |
| 1 | 15.8 | |
| AMPA | 0.2 | 5.2 |
| 0.4 | 9.4 | |
| 1 | 5.2 |
Table 2 Determination for the limit of quantification (LOQ) for glyphosate -2-13C, and AMPA.
| Calculated concentration (µM) | Glyphosate-2-13C | AMPA | ||
| Final concentration (µM) | Accuracy (%) | Final concentration (µM) | Accuracy (%) | |
| 0.1 | 0.0192 | 19.2 | 0 | 0 |
| 0.2 | 0.2168 | 108.4 | 0.2667 | 133.35 |
| 0.4 | 0.3750 | 93.8 | 0.50725 | 126.8 |
| 1 | 1.1242 | 112.4 | 1.02245 | 102.25 |
| 2 | 1.8353 | 91.8 | 2.1162 | 105.8 |
| 4 | 4.0913 | 102.3 | 3.5238 | 88.1 |
| 6 | 6.0560 | 100.9 | 5.97505 | 99.6 |
| 8 | 7.7545 | 96.9 | 8.05655 | 100.7 |
| 10 | 10.3036 | 103.0 | 10.10195 | 101 |
| 12 | 11.9176 | 99.3 | 12.01165 | 100.1 |
| 14 | 14.4511 | 103.2 | 13.9678 | 99.8 |
| 16 | 15.2718 | 95.4 | 15.9429 | 99.65 |
| 18 | 18.1509 | 100.8 | 18.34165 | 101.9 |
| 20 | 20.0951 | 100.5 | 19.7587 | 98.8 |
Validation and selectivity of glyphosate-2-13C and AMPA detection
Method selectivity addresses the method’s ability to quantify the substance of interest in the presence of other components with analogous behavior under specific assay conditions. This parameter, along with the specificity, allows us to establish the confirmation of a compound identity, which ensures that the signal produced in the measurement is attributed exclusively to the analyte, and not to the presence of a different (physically or chemically related) substance [50]. Fig. 4, shows representative chromatograms of the matrix fortified with glyphosate-2-13C (Fig. 4(A)), the non-fortified matrix (Fig. 4(B) ), and the matrix fortified with AMPA (Fig. 4(C), 4(D)). AMPA is a product of the microbial degradation of glyphosate, and it is therefore structurally related to glyphosate [30]. When intending to detect glyphosate-2-13C in AMPA-fortified samples, no glyphosate-2-13C was detected (Fig. 4(C)), and there was no interference in the retention time established for each compound. These two results confirm the selectivity and specificity of the glyphosate assay.
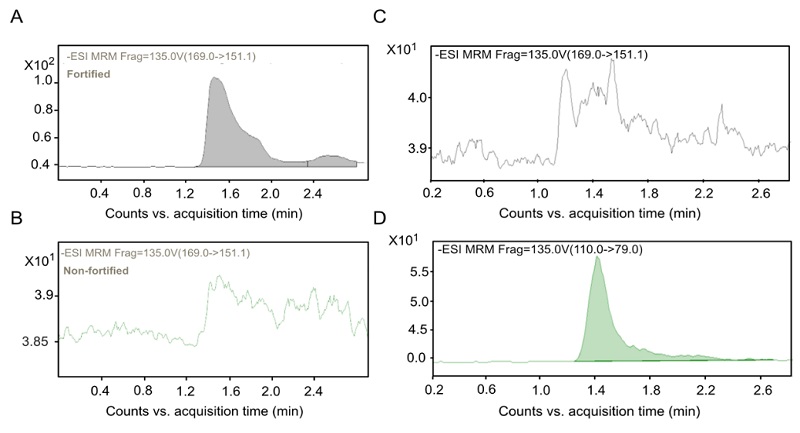
Fig. 4 Selectivity and specificity of the glyphosate quantification method in corn flour. Chromatograms of glyphosate (A, B, and C) and AMPA (D) in samples fortified with glyphosate (A) and non-fortified with glyphosate (B) and fortified with AMPA (C) and (D). The ASE extraction method was used for these tests.
To validate the repeatability of the method, three serial glyphosate determinations were made in fortified samples on the same day (Table 3), obtaining standard deviations of 0.1961 and 0.2372 µM for glyphosate-2-13C and AMPA quantifications, respectively. The reproducibility was calculated by determining the concentration of glyphosate-2-13C on three different days; the average of the three injections was 18.5120 ± 1.0703 µM (Table 4).
Table 3 Repeatability of glyphosate -2-13C and AMPA determinations.
| Compound | Replicates (µM) | Average | Standard deviation | ||
| 1 | 2 | 3 | |||
| Glyphosate -2-13C | 18.5192 | 18.8817 | 18.5705 | 18.6571 | 0.1961 |
| AMPA | 15.9053 | 16.3779 | 16.1058 | 16.1297 | 0.2372 |
Glyphosate-2-13C extraction protocol
In this work, the recovery percentage varied from 41.65 % to 109.19 % (Table 5). In all cases, the methanol-water mixture in a 20:80 ratio, acidified with 1 % of FA exhibited the highest recovery percentages (Table 5). The extraction with ASE and ultrasonication exhibited the highest recovery values with 109.19 % and 93.12 %, respectively (Table 5). In contrast, the EDGE protocol exhibited recovery values of 41.49 - 58.49 % (Table 5) but in a shorter time (2 h, 20 min) (Table 6).
Table 5 The recovery percentage of glyphosate with three different extraction methods and different solvent proportions.
| Extraction system | MeOH -H2O* | Concentration (ppm) | Recovery (%) | |
| Expected | Observed | |||
| EDGE | 20:80 | 113.33 | 66.29 + 11.55 A | 58.49 + 10.19a |
| 50:50 | 226.67 | 94.40 + 59.12 A | 41.65 + 26.08a | |
| ASE | 20:80 | 113.33 | 123.75 + 9.36 C | 109.19 + 8.26c |
| 50:50 | 226.67 | 148.91 + 29.80 AB | 65.70 + 13.15ab | |
| Ultrasonication | 20:80 | 113.33 | 105.53 + 1.67 BC | 93.12 + 1.48bc |
| 50:50 | 45.40 A | 21.98 + 0.32 A | 48.42 + 0.71a | |
The highest recovery percentages are shown in bold (ASE n=3, EDGE n=3, and Ultrasonication n=4). *Formic acid (0.1 %) was added to the solvent. Different superscript letters among concentration values denote statistically significant differences (P < 0.05).
Table 6 Time invested in each extraction system workflow for 12 samples.
| Procedure steps | Extraction methods | ||
| ASE | EDGE | ULTRASONICATION | |
| Assembly of extractions cells* | 45 min | 10 min | NA |
| Sample weighing | 60 min | 30 min | 40 min |
| Equipment assembly | 20 min | 10 min | NA |
| Extraction time | 180 min | 80 min | 101 min |
| Dismantling of extraction cells | 45 min | 10 min | NA |
| Centrifugation | NA | NA | 20 min |
| Total working time (h:min) | 5:50 | 2: 20 | 2:41 |
*Cells are used by ASE and Q-cup for EDGE.
The highest recovery percentages are shown in bold (ASE n=3, EDGE n=3, and Ultrasonication n=4). *Formic acid (0.1 %) was added to the solvent. Different superscript letters among concentration values denote statistically significant differences (P < 0.05).
Discussion
The development of methods to detect and quantify glyphosate in different food-related matrices are necessary to safeguard human health from the harmful effects of this pesticide. The main challenge for the detection and quantification of glyphosate is that it can be masked by the chemical components in the food matrix. Due to the chemical nature of glyphosate, most of the published methods include a derivatization step to increase the possibility of detection. However, this can also introduce bias during the estimation of the total content of these pesticides during routine analysis. In our study, we tested three different alternatives for glyphosate extraction and one quantification method that avoids the derivatization step.
In studies on pesticide detection, different columns have been used (including anion exchange, HILIC, etc.) and each one works for a specific condition [19,20,49,53]. However, considering that most of the reported glyphosate and AMPA assays are performed by reverse phase HPLC, in this study a C18 column was used. In addition, C18 columns are used as a stationary phase because it offers a very simple hydrophobic interaction, allowing the retention of highly polar ionizable molecules, as is the case of glyphosate and its highly polar catabolites. The short retention time did not result in a lower sensitivity of its detection, so the direct determination of glyphosate-2-13C and AMPA was also feasible. The column remained stable throughout all the operating conditions, and no modification in retention time or peak shape degradation was observed at more than 250 injections.
The ASE and ultrasonication methods provided high percentages of glyphosate recovery. The major advantage of ASE extraction over the ultrasonication method is its reliability and process automatization, which enables several continuous extractions and does not require centrifugation. However, the ASE requires additional consumables like gaseous nitrogen. In contrast, the ultrasonication method requires conventional items like polypropylene test tubes, pipette tips, and considerably fewer solvents, decreasing the cost of the analysis. However, it is a mostly manual process which may be impractical for many samples. For comparison purposes, we summarize reported methods for glyphosate analysis in several food and soil matrixes (Table 7). One of the best protocols proposed for glyphosate and AMPA determination used water as the extraction solvent. Crude extracts of crops were homogenized with maceration at high speed (3-5 min). Then the sample preparation pipeline included cation exchange cleanup and derivatization with heptafluorobutanol and trifluoroacetic anhydride. The analysis of the sample mentioned above with capillary gas chromatography with mass-selective detection (MSD) exhibited significant performance [19,21]. The highest glyphosate recovery percentage has been reported in potatoes (106 %). This study tested the detection of glyphosate in different fruits and vegetables by using water as a solvent and avoided derivatization. The proposed protocol also included a mixed-mode hydrophilic interaction/weak anion-exchange liquid chromatography (HILIC/WAX) coupled with electrospray tandem mass spectrometry [19,21]. The protocols mentioned above provide invaluable tools for detecting and quantifying glyphosate and AMPA. However, in terms of cost, time, and sample handling steps does not provide a feasible method for heterogeneous matrices like corn flour and related products. In contrast, simple methods like agitation/ultrasonic had percentage recoveries values in maize samples ranging from 85 to 104 % [20,48,49,54] (Table 7).
Table 7 The recovery percentage of glyphosate and AMPA reported using different extraction systems, solvents, and matrices.
| Matrix | Extraction method | Solvent | Recovery % | References | |
| Glyphosate | AMPA | ||||
| Soil | ASE | H2O | 0 | 0 | [27] |
| NaOH 0.1 M | 12.4 | ND | |||
| Ultrasonic | H2O | 9.8 | 16.4 | ||
| NaOH 0.1 M | 9 | 24 | |||
| Agitation | H2O | 1.3 | 3.8 | ||
| H3PO4 | 0 | 0 | |||
| CaCl2 | 7 | 17 | |||
| Ultrasonic | KH2PO4 | 44 | ND | [34] | |
| Maize | Agitation/Centrifugation | H2O: MeOH 50:50 + 1 % formic acid | 85 | ND | [48] |
| Ultrasonic | H2O: MeOH 50:50 | 85 | ND | [20] | |
| Homogenization | H2O | 89.2 | 98.4 | [54] | |
| Agitation | Acetic acid 50 mM: Na2 EDTA 10 mM | 104 | ND | [49] | |
| Potato | AOAC Official Method 2000.05 | H2O | 106 | 80.8 | [19,24] |
| Canola | Homogenization | H2O: dichloromethane (1:1; v/v) | 58.6 | 53.7 | [25] |
| H2O: dichloromethane (2:1; v/v) | 64.8 | 59.4 | |||
| Borates Buffer (pH=9) | 30.5 | 28 | |||
| KOH solution (0.5 M) | 26.7 | 24.5 | |||
| H2O: methanol: dichloromethane (2:2:1; v/v/v) | 71.2 | 65.3 | |||
| H2O | 82 | 75.2 | |||
| MeOH | 68.2 | 62.5 | |||
| Acetonitrile | 58.4 | 48.7 | |||
| Tetrahydrofuran | 32.5 | 28.7 | |||
| H2O: MeOH (1:1; v/v) | 75.6 | 69.3 | |||
| H2O + 1 % formic acid | 88.1 | 80.7 | |||
| H2O + 1 % HCl | 83.3 | 76.3 | |||
| H2O + 0.1 % formic acid | 93.4 | 85.6 | |||
| H2O + 0.1 % HCl | 88.3 | 80.9 | |||
| Cereals | Ultrasonic | H2O | 93 | ND | [58] |
| Cornflour | ASE | H2O + MeOH (80:20) 1 % formic acid | 109.19 | ND | Méndez-Barredo et al. |
Druart and collaborators included the evaluation of three extraction systems such as ASE, ultrasonication, and agitation (magnetic stirring, using an agitation from 1 to 15 hours) for glyphosate, AMPA, and glufosinate, using a derivatization step [27]. The authors suggested that ASE is not optimal for the extraction of hydrophilic herbicides like glyphosate in soil, but it is optimal for samples soluble in organic solvents. Besides, the recovery percentage obtained with ultrasonication extraction outperformed that of ASE (18 % for glyphosate and 34 % for AMPA). We considered some points of the aforementioned studies in our proposed protocol. For example, that study suggested mixing clay-rich soils with sand to allow the solvent to pass through the cell. In addition, the authors also suggested the use of water as the sole extraction solvent and a short extraction time (10 min). In our protocols, we mixed the samples with diatomaceous earth (3:1 ratio) to avoid compaction of the sample in the cell and improve solvent contact with the entire sample surface area for efficient extraction. Furthermore, we used methanol-water with 1 % FA, obtaining recovery percentages up to 109 % for glyphosate. Another study reported the extraction of glyphosate from the soil by ultrasonication in phosphate buffer for 60 minutes and obtained a recovery efficiency of 44 % [34].
We also evaluated the glyphosate extraction efficiency with the automated EDGE system. The conditions used in the equipment were adapted according to the available literature from pesticide extraction application notes [45]. This system has exhibited high extraction efficiency for pesticides in products for human consumption, without derivatization and with automated extraction protocols, obtaining recovery percentages comparable to the QuEChERS method for pesticides [39]. Reported extraction methods involving the EDGE system exhibited recovery percentages greater than 80 %, identifying more than 140 pesticides in green tea, and validating 361 pesticides in cocoa beans [55]. To the best of our knowledge, our study is the first report on the use of EDGE to detect and quantify glyphosate in food matrices. Previous studies that included EDGE system comprised post-sowing foliar herbicides, such as 2,4-D (84 % recovery), corn growth inhibitors such as pendimethalin (120 % recovery), and herbicides from the group of photosynthesis inhibitors used for corn cultivation, such as atrazine (112 % recovery) [55-57]. We obtained glyphosate recovery percentages between 41 % and 58 % in commercial corn flour samples fortified with glyphosate (depending on the solvent, Table 5). Considering previous reports with EDGE, our protocol might be improved by using 100 % water as the main extraction solvent. Therefore, it is necessary to standardize each technique according to the food matrices as well as the availability of workload, work staff, and variety of samples to process.
Conclusions
We compared different extraction protocols and developed a dMRM method for glyphosate quantification in fortified corn flour samples without derivation steps or column regeneration. This provides a rapid method by combining the advantages of using ultra-high resolution liquid chromatography with the sensitivity and specificity provided by tandem mass spectrometry, obtaining high recovery percentages. The selective analytical method demonstrated here was validated in terms of selectivity, specificity, repeatability, and reproducibility. The established protocol is a simple and low-cost method, which can be adaptable and modifiable for use in other food matrices, which could be considered in the near future for regulatory agencies in the food industry.











 nueva página del texto (beta)
nueva página del texto (beta)




