Introduction
Subependymal giant cell astrocytoma (SEGA) is a benign, slow-growing, circumscribed tumor classified as astrocytic glioma, grade 1 by the World Health Organization (WHO), and is typically associated with the tuberous sclerosis complex (TSC), an autosomal dominant hereditary disorder caused by a mutation in one of the TSC genes1. TSC1 (9q34) codes for hamartin, and TSC2 (16p13.3) codes for tuberin2. TSC incidence is one in 5000-10,000 live births. 5-20% of patients with TSC develop SEGAs3. SEGA can be single or multiple with a diameter of 1-10 cm, typically located adjacent to the lateral wall of the ventricles or at the foramen of Monro4. Histologically, with large polygonal and elongated cells, SEGA shows co-expression of glial and neuronal markers5. By identifying the mTOR pathway as the protagonist of SEGA pathogenesis, inhibitors of this pathway have been developed6. However, in some cases, medical treatment with mTOR inhibitors is not totally effective to control tumor growth7. We investigated the participation of the Hippo pathway in tumor growth, and we applied an immunohistochemistry (IHC) panel to reveal whether the tumor expresses stem cell markers that could suggest its origin in these cells.
The objectives of this study were to describe the clinicopathological features of patients with SEGA, distinguishing, if possible, between TSC-associated and solitary cases. In addition, immunohistochemical studies aim to provide novel insights into the cell of origin and other involved metabolic pathways, potentially offering therapeutic applications.
Materials and methods
It was a retrospective and descriptive study with a convenience sample from January 2013 to December 2022. The protocol was reviewed and approved by the Research and Ethics Committees, and the registration number 36/23 was assigned. Since it was a retrospective study where personal data were not exposed, the Research and Ethics Committees did not consider it necessary to obtain a letter of informed consent. The study was conducted according to the principles established in the Declaration of Helsinki.
All histologically corroborated SEGA cases with clinical and radiological records, as well as paraffin blocks for the IHC study, were included. Patients with incomplete clinical data, neuroimaging, or lacking tumor tissue were excluded. Information about sex, age at diagnosis, association with TSC, initial symptom or sign, neurological data, clinical evolution, treatment, post-surgical complications, recurrences, and magnetic resonance imaging findings was obtained.
The surgical specimens were fixed in 4% buffered formalin and embedded in paraffin. 5 μm sections were stained with hematoxylin and eosin for evaluation. The standard streptavidin-biotin technique was performed using the following primary antibodies: Glial fibrillary acidic protein (GFAP; catalog MU020-UC, prediluted, Biogenex, Fremont, CA, USA); Neurofilaments (NF; catalog M-0762, prediluted, Dako, Carpinteria, CA, USA); Cytokeratins AE1/AE3 (CK-AE1/AE3; catalog PDM072, prediluted, diagnostic biosystems, Pleasanton, CA, USA); Ki67 (catalog MU410- UC, prediluted, Biogenex, Fremont, CA, USA); Signal transducer and activator of transcription 6 (STAT-6; catalog Sc-1689, prediluted, Santa Cruz Biotechnology, Dallas, TX, USA); Octamer-binding transcription factor 4 (OCT-4; catalog MC0598, prediluted, Medayasis, Livermore, CA, USA); Nestin (catalog Sc-23927, prediluted, Santa Cruz Biotechnology, Dallas, TX, USA); Hamartin (catalog MC0598, prediluted, Medayasis, Livermore, CA, USA); Tuberin (catalog RC0317, prediluted, Medayasis, Livermore, CA, USA); Integrase interactor 1 (INI-1, catalog LS-B6039, prediluted, Life-Span BioSciences, Seattle, WA, USA); Thyroid transcription factor 1 (TTF-1, catalog 343-M-96, prediluted, Cell Marque, Rocklin, CA, USA); Vimentin (catalog MU074-UC, prediluted, Biogenex, Fremont, CA, USA); Marker of T helper cells (CD4, catalog MU421-UC, prediluted, Biogenex, Fremont, CA, USA); Cytotoxic T cell marker (CD8, catalog MU261-UC, prediluted, Biogenex, Fremont, CA, USA); Yes associated protein (YAP65, catalog #14074, prediluted, cell signaling technology, Danvers, MA, USA). Immunohistochemical staining was performed manually, according to the suppliers instructions. The immunohistochemical score was determined whether the positive staining was in the membrane, cytoplasm, or nuclear. Percentage: negative stain (0-4%); (+) 5-49% of cells; (++) 50-79%; (+++) 80-100% of cells. To evaluate this percentage, the number of positive cells in 10 high-power fields was quantified.
Results
In total, four cases were studied, with a median age at diagnosis of 23.5 years (range 19-46). The main features are described in Table 1.
Table 1 Clinical features of patients with SEGA
| Patient No | Age (years)/sex | TSC | Age at TSC diagnosis | TSC features | Initial symptoms | Hydrocephalus | SEGA location (size/mm) | Surgical treatment/post-surgical complications | Mortality |
|---|---|---|---|---|---|---|---|---|---|
| Patient 1 | 22/F | + | 10 years | Hypochromic spots and periungual fibroma | Headache and nausea | + | RLV (18×22×30) | Endoport, VPS/Epidural hematoma | Alive |
| Patient 2 | 25/F | + | 12 years | Facial angiofibromas, gingival fibromas, pits in teeth, hypochromic spots | Headache and general weakness | + | RLV (31.7×22.7×26) | Endoport, VPS, Ventriculostomy/- | Alive, recurrence after 2 years |
| Patient 3 | 46/M | - | - | - | General weakness and somnolence | - | RLV (Not available) | Endoport, ventriculostomy/- | Alive |
| Patient 4 | 19/M | - | - | - | Headache | - | LLV (31×17×20) | Endoport/- | Alive |
SEGA: subependymal giant cell astrocytoma; M: male; F: female; TSC: tuberous sclerosis complex; VPSS: ventriculoperitoneal shunt; RLV: right lateral ventricle; LLV: left lateral ventricle.
Morphology
In all cases, tumors were moderately cellular, with a fibrillar background. TSC patients showed compact nest-forming giant cells (80-200 μm) circumscribed by elongated cells (30-120 μm) in fascicles. In solitary SEGA, the giant cells were smaller (40-120 μm) and the nodules were not well defined, or the giant cells were scattered throughout the tumor tissue, interspersed with elongated cells (20-100 μm). Mitoses were not observed. The vessels were wide-lumen capillaries and arterioles, and in one case of each group, sclerotic walls were observed. Inflammatory cells had a perivascular pattern in all cases (Fig. 1).
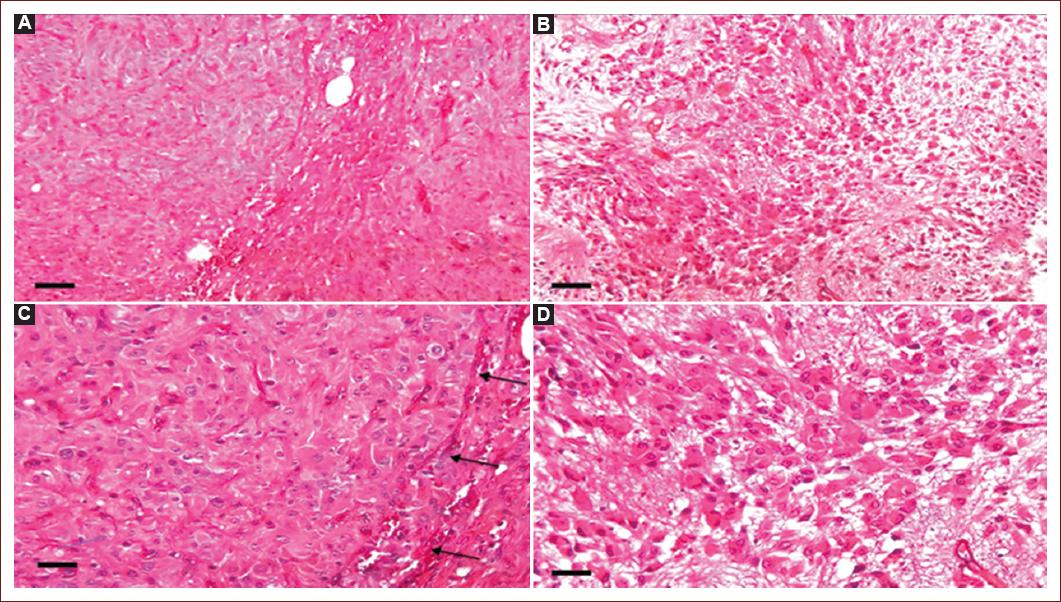
Figure 1 Histological comparison between tuberous sclerosis complex-associated and solitary subependymal giant cell astrocytoma. A and C: the giant cells show nodules with a compact, and well-defined borders (C, arrows), delimited by elongated cells in patients with tuberous sclerosis complex. B and D: in the solitary subependymal giant cell astrocytoma, the giant cells form less well-defined nests or are intermingled with the elongated cells that form fascicles. 50× (A, B); 100× (C, D). Hematoxylin and eosin. Bar: A, B = 500 μm; C, D = 200 μm.
IHC
It showed positive staining in neoplastic cells for GFAP, NF, vimentin, and nestin, with negative Ki67 staining. The main results with the different antibodies are shown in Table 2 and Fig. 2.
Table 2 Histological findings in immunohistochemistry
| Antibody | TSC | Solitary | ||
|---|---|---|---|---|
| Patient 1 | Patient 2 | Patient 3 | Patient 4 | |
| GFAP* | GC and EC and fibrillar process | Diffuse cytoplasmic stain in EC and GC | ||
| +++ | +++ | +++ | +++ | |
| NF* | Diffuse pattern in GC and EC | Strong staining in EC and fibrillar processes, slight staining in EC | ||
| ++ | ++ | ++ | ++ | |
| CKAE1/AE3* | Diffuse staining in cytoplasm and processes of GC and in EC | Diffuse, intense staining in fibrillar processes of EC | ||
| +++ | +++ | +++ | +++ | |
| Ki67n | Negative | Negative | ||
| STAT6n | Cytoplasmic staining in GC and EC | Diffuse homogeneous cytoplasm stain | ||
| ++ | ++ | + | + | |
| OCT4n | Cytoplasm staining in EC and GC | Negative | Cytoplasmic and fibrillar processes in EC | |
| + | + | | ++ | |
| Nestin* | Staining in GC and their extensions, and in ES | Diffuse pattern in cytoplasm and fibrillar processes (GC and EC) | ||
| +++ | +++ | +++ | +++ | |
| Hamartin* | Staining in GC and EC | Negative | Negative | Staining in GC, EC |
| ++ | | | +++ | |
| Tuberin* | Negative | Negative | Slight checkerboard staining in both GC and EC | |
| | | | + | |
| INI1n | Diffuse pattern with nuclear staining and slight cytoplasmic staining | Negative | Diffuse pattern with cytoplasm staining in GC and EC | |
| +++ | +++ | | ++ | |
| TTF1n | Strong nuclear staining and slight cytoplasmic staining in GC, and SE | Strong nuclear staining in GC, and EC and slight cytoplasmic staining | ||
| +++ | +++ | ++ | ++ | |
| Vimentin* | Strong, diffuse, staining in cytoplasm in all cells | Strong, diffuse, staining in cytoplasm in all cells | ||
| +++ | +++ | +++ | +++ | |
| CD4* | Lymphoid cells in perivascular distribution and surrounding GC | Scarce lymphoid cells in perivascular distribution | ||
| +++ | +++ | + | + | |
| CD8* | Perivascular lymphocytes and among tumor cells. 1015 cells cells per HPF | Perivascular lymphocytes and scattered among tumor cells. 2 cells per HPF | ||
| +++ | +++ | ++ | ++ | |
| YAPn | Diffuse pattern of cytoplasm staining in GC and EC | Positive nuclear staining in 5% of the cells. Diffuse pattern of cytoplasm staining in GC and EC | Negative | Strong nuclear staining in GC and EC |
| + | + | | +++ | |
Negative stain.
+ 5-49%.
++ 50-79%.
+++ 80-100%.
*Cytoplasmic/ membrane stain
nNuclear stain.
TSC: tuberous sclerosis complex; GFAP: glial fibrillary acidic protein; NF: neurofilaments; CK-AE1/AE3: cytokeratins AE1/AE3; STAT-6: signal transducer and activator of transcription 6; OCT-4: octamer-binding transcription factor 4; INI-1: integrase interactor 1; TTF-1: thyroid transcription factor 1; YAP: yes-associated protein, GC: giant cells; EC: elongated cells; HPF: high-power field (400×).
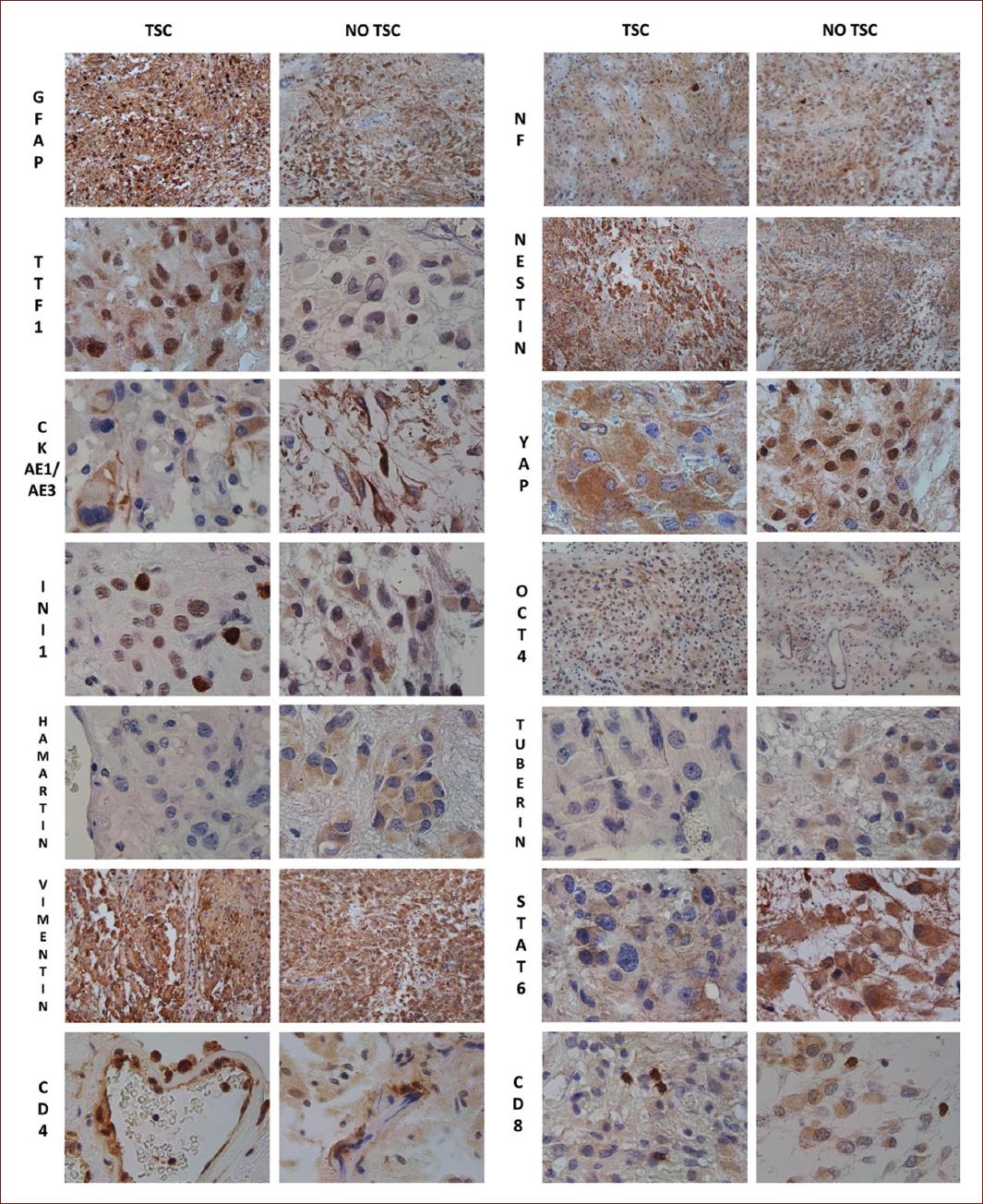
Figure 2 Comparative immunohistochemical study in tuberous sclerosis complex -associated and solitary subependymal giant cell astrocytomas. GFAP: glial fibrillary acidic protein; NF: neurofilaments; CK: cytokeratins AE1/AE3; STAT-6: signal transducer and activator of transcription 6; OCT-4: octamer-binding transcription factor 4; INI-1: integrase interactor 1; TTF-1: thyroid transcription factor 1; YAP: yes-associated protein; YAP65. 100×: GFAP, NF, Nestin, Octamer-binding transcription factor 4, Vimentin. 400×: Thyroid transcription factor 1, cytokeratins AE1/3, yes-associated protein, Integrase interactor 1, Hamartin, Tuberin, signal transducer, and activator of transcription 6, CD4, CD8.
Discussion
There was no significant difference in relation to the clinical features of the tumor between patients with and without TSC. However, a compacted nodular morphology of giant cells surrounded by fascicles was observed in patients with TSC, while in the two sporadic cases, the giant cells were smaller, did not form well-defined nodules, or were individually intermingled with the cells that formed fascicles. Although SEGA continues to be classified as an astrocytoma by the WHO, it has glioneuronal expression, which was confirmed in our study using GFAP and NF5. Glial and neuronal marker uptake in giant cells and their processes were observed in solitary forms and in those associated with TSC. In a previous study on SEGA, TTF-1 expression was observed in this tumor7. We corroborated a strong nuclear stain in TSC cases (90-100% of cells); solitary SEGA also showed nuclear staining, but with less intensity and a lower percentage of tumor cells (> 50%). The expression of TTF-1 in the fetal medial ganglionic eminence, a transient fetal structure between the caudate nucleus and thalami, suggests that SEGA may originate from progenitor cells in this region7,8.
The IHC panel that we performed provided new findings. We wanted to determine if SEGA expressed markers associated with stem cells, such as Nestin and OCT-4, or if they were capable of expressing markers from other cell lines.
Nestin and CK-AE1/AE3
Nestin is an intermediate filament of neuroepithelial stem cells. In adults, nestin-positive cells serve as a resource for cells capable of proliferating, differentiating, and migrating when required9. In certain tumors, nestin-positive cells are a marker of cancer stem cells associated with an invasive phenotype and angiogenesis10. CKs are intermediate filaments of the epithelial cytoskeleton that allow cells to cope with mechanical stress and are useful for confirming the epithelial nature of tissues11. All the tumors were positive for nestin and CK, with intense cytoplasmic staining. Previously, we reported CK expression in a case of SEGA, and we confirmed its presence in these cases12. SEGA expresses nestin and CK, which supports its origin in a cell capable of expressing markers of several cell lines in the same tumor.
INI-1 and OCT-4
INI-1 is a tumor suppressor gene (22q11.2) whose protein controls cell growth, division, and death and encodes a core subunit of the adenosine triphosphate-dependent switch/sucrose non-fermentable chromatin remodeling complex, which is involved in the regulation of gene expression13. Due to its role in the regulation of tumor growth and suppression, it was important to identify whether there was a loss of expression in SEGA. Interestingly, nuclear expression was preserved in cases of TSC but lost in solitary SEGA, and diffuse cytoplasmic expression was observed in one case in giant cells. This is a novel finding that could be helpful in differentiating between solitary SEGAs and SEGAs associated with TSC. OCT-4 (POU5F1) is a transcription factor that is involved in the control of pluripotency, self-renewal, and maintenance of stem cells. Its expression is associated with the degree of malignancy in gliomas and promotes self-renewal, chemoresistance, and tumorigenicity in neoplastic stem cells. OCT-4 generates at least three protein isoforms: OCT-4A, OCT-4B, and OCT-4B1. When OCT-4A is translocated to the nucleus, it functions as a transcription factor responsible for pluripotency and the regulation of angiogenesis14. The tumors analyzed did not show nuclear expression, but cytoplasmic staining was observed in three cases. The expression of OCT-4 in SEGA has not been previously reported and is a novel marker for understanding tumor pathogenesis.
Hamartin and tuberin
To determine the possible practical diagnostic value, we analyzed the expression of wild-type hamartin and tuberin proteins15. As can be seen in Table 2, a patient with TSC expressed hamartin but not tuberin; it is known that the TSC2 mutation is more frequent, and therefore, this would explain its absence in this tumor. In the other TSC case and in one case that was not associated with TSC, both proteins were absent. In another solitary case, hamartin expression was observed, but tubulin was stained in a mosaic checkerboard pattern. We believe that the expression of the wild-type protein is useful, especially in cases not associated with TSC, because it helps show which protein is not expressed or if there is an abnormal pattern.
The inflammatory component
STAT6 is a member of the STAT family. It mediates the biological effects of IL-4, which is necessary for type 2 differentiation of T cells, B-cell survival and proliferation, and class switching to immunoglobulin E16. The presence of lymphocytic and mast cell infiltration has been reported in SEGA; therefore, we aimed to determine the expression of STAT-6, CD4 (T helper cells), and CD8 (cytotoxic T cells) in SEGA17. The inflammatory component in our cases included an admixture of T lymphocytes (helper and cytotoxic), macrophages, and plasma cells. All cases showed tumor cells with positive cytoplasmic staining (not active) for STAT-6; CD4 lymphocytes were predominantly in the perivascular pattern. CD8 lymphocytes also showed a perivascular location in all cases; however, in the two TSC cases, there were also clusters of 10-15 cells between the giant and fascicular cells.
Possible pathways of SEGA pathogenesis
In glioblastomas, the Hippo pathway promotes tumor growth. Its downstream effectors, YAP and transcriptional activator with PDZ-binding motif (YAP/TAZ), regulate cell proliferation and differentiation and maintain crosstalk with the mTOR and WNT pathways. The Hippo pathway is a highly conserved signaling pathway involved in the regulation of cell proliferation, apoptosis, and tissue growth during embryonic development and functions as a tumor suppressor. Its dysregulation has been implicated in various diseases, including cancer, due to tissue overgrowth18. The Hippo pathway was first discovered in Drosophila melanogaster (fruit flies) and has since been found to have counterparts in mammals, including humans. In the human Hippo pathway, its key components include Hippo kinases (MST 1/2), Warts kinases (LATS 1/2), and the TEA domain family (TEAD). Under optimal conditions, Hippo pathway regulation starts with MST 1/2, which is a serine-threonine kinase that binds to the Salvador homolog 1 protein and phosphorylates Warts kinases18. Once phosphorylated, LATS 1/2 kinases phosphorylate the Hippo pathway effector YAP/TAZ, acting as inhibitors. Phosphorylated YAP and TAZ remain inactive in the cytoplasm, but when dephosphorylated or unable to be retained in the cytoplasm, YAP and TAZ translocate to the nucleus and anchor with TEAD transcription factors18. The binding between YAP/TAZ and TEAD promotes the expression of genes involved in cell proliferation and inhibition of apoptosis18. Furthermore, the mTOR prooncogenic and Hippo tumor-suppressive signaling pathways are cell-signaling cascades regulated by Merlin, a member of the ezrin, radixin, and moesin family of proteins that regulate cytoskeleton and cell signaling and are encoded by the NF2 gene (tumor suppressor gene)19. As observed in glioblastomas, multiple extracellular stimuli and pathways can trigger cell proliferation. Therefore, it is essential to consider other pathways that may regulate mTOR expression. In this study, 2/4 patients presented nuclear expression of YAP, meaning a dephosphorylated YAP, translocated to the nucleus, and capable of activating cell proliferation20. This is important due to the possibility of using a combination of drugs to regulate the growth of SEGA21. The combination of pharmacological inhibitors of mTORC1/2 and YAP is currently used in triple-negative breast cancer with efficient tumor shrinkage. Verteporfin-induced YAP inhibition leads to apoptosis, and torin1-mediated inhibition of mTORC1/2 promotes macropinocytosis. Macropinocytosis facilitates verteporfin uptake, greatly enhancing pro-apoptotic effects on cancer cells22. Furthermore, SEGA-like tumors have been reported in NF1 patients, and Merlin is known to regulate the mTOR and Hippo pathways19.
The main limitation of this study is the number of cases; however, the results are very clear and show several differences between TSC-associated and solitary SEGA, which should be confirmed by other authors in other cases.
Conclusion
The clinical manifestations of the tumor were similar in patients with sporadic SEGA and those associated with TSC. Diffuse expression of nestin and CK suggests the origin of SEGA in neuroepithelial stem cells. SEGA is a glioneuronal tumor whose diagnosis must be supported by an IHC panel that includes GFAP, NF, synaptophysin, nestin, and TTF-1. Morphological differences were observed between TSC-associated and unassociated SEGAs, specifically in the compaction of giant cell nodules in the former and in the retention of INI-1 nuclear expression in TSC patients. Nuclear expression of YAP-1 was demonstrated in two of the four tumors studied. Therefore, the possibility of the joint use of Hippo and mTOR pathway regulators should be evaluated in the management of SEGA.











 nueva página del texto (beta)
nueva página del texto (beta)


