Introduction
Amphibian diversity in Mexico is one of the highest worldwide (Ramírez-Bautista et al., 2023); 42 species of amphibians have been reported in the State of Colima alone, representing 11% of the total species reported in Mexico (García et al., 2022), although Colima only represents 0.3% of the total country area (INEGI, 2015). Knowledge on the helminth fauna of amphibians and its biology provides valuable information about the conservation state of the ecosystems and can be used as indicators of the health of host populations (Brooks et al., 2014; Marcogliese et al., 2003). In spite of their potential, studies on amphibian helminths in Colima are scarce, and most of them are isolated records. To date, 17 species of helminths (8 digeneans, 1 cestode and 8 nematodes) have been recorded, out of 6 species of anurans (Cabrera-Guzmán et al., 2022). Two of the most common anuran species in Colima are Leptodactylus melanonotus (Hallowell, 1861) and Incilius marmoreus (Weigman, 1833) (García & Ceballos, 1994), nevertheless, there is only 1 study on the helminth fauna of L. melanonotus (Mata-López et al., 2013) and there is nothing for I. marmoreus (Table 1).
Table 1 Helminth records of Leptodactylus melanonotus and Incilius marmoreus in Mexico. A = Adult, L = larvae, Ch = Chiapas, C = Colima, G = Guerrero, M = Michoacán, N = Nayarit, O = Oaxaca, QR = Quintana Roo, S = Sonora, T = Tabasco, Ta = Tamaulipas.
| Helminth species | Host species | Site of infection | Reference, locality |
|---|---|---|---|
| Acanthocephala | |||
| Acanthocephala gen. sp. L | I. marmoreus | Mesentery | Trejo-Meléndez et al. (2019), O |
| Centrorhynchus sp.L | I. marmoreus | Body cavity | Galicia-Guerrero et al. (2000), J |
| L. melanonotus | Intestine | Terán-Juárez (2011), QR | |
| Mesentery | Goldberg et al. (2002), V | ||
| Cestoda | |||
| Cyclophyllidae gen. sp. L | L. melanonotus | Body cavity | Terán-Juárez (2011), QR |
| Cylindrotaenia americanaA | L. melanonotus | Intestine | Goldberg and Bursey (2002), S |
| Nematotaeniidae gen. sp. A | I. marmoreus | Intestine | Trejo-Meléndez et al. (2019), O |
| Digenea | |||
| Catadiscus propinquusA | L. melanonotus | Intestine | Brooks et al. (2006), V; Mata-López et al. (2013), G, J, N, O, T |
| Choledocystus hepaticumA | I. marmoreus | Liver | Trejo-Meléndez et al. (2019), O |
| Clinostomum tataxumuiA | I. marmoreus | Liver | Trejo-Meléndez et al. (2019), O |
| Clinostomum sp. L | L. melanonotus | Mesentery | Mata-López et al. (2013), C |
| Glypthelmins facioiA | L. melanonotus | Intestine | Goldberg et al. (2002), V |
| Gorgoderina attenuataA | I. marmoreus | Urinary bladder | Trejo-Meléndez et al. (2019), O |
| L. melanonotus | Urinary bladder | Mata-López et al. (2013), Ch, C; Goldberg and Bursey (2002), S; Goldberg et al. (2002), V | |
| Gorgoderina festoniA | L. melanonotus | Urinary bladder | Mata-López and León-Règagnon (2005), C |
| Mata-López and León-Règagnon (2005); Mata-López et al. (2013), G | |||
| Haematoloechus longiplexusA | L. melanonotus | Lungs | Goldberg and Bursey (2002), S |
| Megalodiscus temperatusA | L. melanonotus | Intestine | Goldberg and Bursey (2002), S |
| Megalodiscus sp. A | L. melanonotus | Intestine | Goldberg et al. (2002), V |
| Mesocoelium americanumA | I. marmoreus | Intestine | Trejo-Meléndez et al. (2019), O |
| Rauschiella poncedeleoniA | L. melanonotus | Intestine | Razo-Mendívil and León-Règagnon (2001), T, V; Razo-Mendívil et al. (2006), G; Mata-López et al. (2013), C, G, J, O; Terán-Juárez (2011), QR |
| R. tineriA | L. melanonotus | Intestine | Mata-López et al. (2013), G, T |
| Strigeidae gen. sp.L | L. melanonotus | Intestine | Mata-López et al. (2013), C |
| Mesentery | Mata-López et al. (2013), O | ||
| Nematoda | |||
| Aplectana incertaA | I. marmoreus | Intestine | This study, C |
| Galicia-Guerrero et al. (2000), J | |||
| Mata-López et al. (2013), T | |||
| A. itzocanensisA | L. melanonotus | Intestine | Goldberg and Bursey (2002), S |
| Mata-López et al. (2013), G, N, O, V | |||
| Aplectana sp.A | I. marmoreus | Intestine | Trejo-Meléndez et al. (2019), O |
| L. melanonotus | Intestine | Mata-López et al. (2013), Ch, G, O, T | |
| Ascarididae gen. sp.L | L. melanonotus | Mesentery | Mata-López et al. (2013), C, G, O, T |
| Ascarops sp. L | L. melanonotus | Stomach | Goldberg et al. (2002), V |
| Contracaecum sp. L | L. melanonotus | Stomach | Terán-Juárez (2011), QR |
| Cosmocerca colimense n. sp. A | L. melanonotus | Intestine | This study, C |
| C. parvaA | L. melanonotus | Intestine | Mata-López et al. (2013), O |
| C. podicipinusA | L. melanonotus | Intestine | Mata-López et al. (2013); Ch, C, G, J, M, O, T, Ta; Goldberg et al. (2002), V, Terán-Juárez (2011), QR; Goldberg and Bursey (2002), S |
| Cosmocerca sp. A | L. melanonotus | Stomach | Terán-Juárez (2011), QR |
| Eustrongylides sp. L | L. melanonotus | Mesentery | Mata-López et al. (2013), Ch |
| Kalicephalus sp. A | L. melanonotus | Intestine | Mata-López et al. (2013), G |
| Oswaldocruzia lescureiA | I. marmoreus | Intestine | Trejo-Meléndez et al. (2019), O |
| O. pipiensA | L. melanonotus | Intestine | Goldberg and Bursey (2002), S |
| O. cf pipiensA | I. marmoreus | Intestine | This study, C |
| O. subauricularisA | I. marmoreus | Intestine | Trejo-Meléndez et al. (2019), O |
| L. melanonotus | Intestine | This study, C; Mata-López et al. (2013), C, G, J, O, T, Ta | |
| Oswaldocruzia sp. A | I. marmoreus | Intestine | Trejo-Meléndez et al. (2019), O |
| L. melanonotus | Intestine | Mata-López et al. (2013), Ch, G; Terán-Juárez (2011), QR; Goldberg et al. (2002), V | |
| Physocephalus sp. | I. marmoreus | Body cavity | Galicia-Guerrero et al. (2000), J |
| Stomach | Trejo-Meléndez et al. (2019), O | ||
| Porrocaecum sp.L | L. melanonotus | Mesentery | Goldberg et al. (2002), V |
| Rhabdias americanusA | L. melanonotus | Lungs | Goldberg et al. (2002), V |
| R. elegansA | L. melanonotus | Lungs | Goldberg et al. (2002), V; Terán-Juárez (2011), QR |
| R. fuellernorniA | I. marmoreus | Lungs | Galicia-Guerrero et al. (2000), J |
| R. pseudosphaerocephalaA | I. marmoreus | Lungs | Trejo-Meléndez et al. (2019), O |
| Rhabdias ranaeA | L. melanonotus | Lungs | Goldberg and Bursey (2002), S |
| Rhabdias sp. A | L. melanonotus | Lungs | This study, C; Mata-López et al. (2013), G, O, T, Ta, V |
| Schrankiana sp. A | L. melanonotus | Intestine | Terán-Juárez (2011), QR, |
| Spiroxys sp. L | L. melanonotus | Intestine | Terán-Juárez (2011), QR |
| Mesentery | Goldberg and Bursey (2002), S; Mata-López et al. (2013), T, Ta | ||
| Stomach | Goldberg et al. (2002), V | ||
| Subulascaris falcaustriformisA | L. melanonotus | Intestine | Goldberg et al. (2002), V |
Leptodactylus Fitzinger, 1826 includes around 84 species, being Leptodactylus fragilis (Brocchi, 1877) and L. melanonotus the only 2 species distributed in Mexico (Flores-Villela, 1993; Flores-Villela & Canseco-Márquez, 2004). Leptodactylus melanonotus (Sabinal frog) is a small frog (svl = 50mm) with indirect development that occurs from Sonora and Tamaulipas in Mexico, throughout Central America, and into South America west of the Andes to Ecuador; inhabit the edges of ponds or flooded pastures, at the base of tufts of grass, or within burrows in the mud through dry and humid premontane habitats (Lee, 1996). To date, 36 helminth taxa had been recorded in L. melanonotus from a wide geographical range in México (Table 1), being Guerrero, Oaxaca, Quintana Roo, and Veracruz the states with a larger number of recorded helminth taxa (Goldberg & Bursey, 2002; Mata-López et al., 2013; Terán-Juárez, 2011).
Incilius Cope, 1863 comprehends 39 species (Mendelson III et al., 2011), 23 of which are native to Mexico and 10 (or 43% of Mexican species) are endemic (Ramírez-Bautista et al., 2023). Incilius marmoreus (Marbled toad) is a small toad (svl = 76 mm) with indirect development, and an endemic species inhabiting tropical areas along the Mexican Pacific coast from northern Sinaloa throughout Chiapas, where it is found in the litter of forest floor (García & Ceballos, 1994; Rueda-Hernández, 2023); some populations also occur in the Balsas basin and coast of the state of Veracruz. The only previous helminth records for I. marmoreus are from the states of Jalisco and Oaxaca (Galicia-Guerrero et al., 2000; Trejo-Meléndez et al., 2019), both in the Pacific coast of Mexico, including 15 taxa (Table 1).
The aim of this study was to describe a new species of nematode parasite in the intestine of L. melanonotus and contribute to the knowledge of the helminth fauna of L. melanonotus and I. marmoreus in Colima.
Materials and methods
Specimens of L. melanonotus (n = 137) and I. marmoreus (n = 31) were collected from July to October 2021, in several sites in the state of Colima, Mexico, distributed along the riparian zones of 3 rivers (Comala, Colima, and Manríquez) and in flooded crop fields and suburban areas in Coquimatlán and Colima municipalities. Specimens were collected under the scientific collection permit FAUT0056 issued to VLR by Secretaría del Medio Ambiente y Recursos Naturales (Semarnat). Amphibians were captured by hand and euthanized by an overdose of sodium pentobarbital, frozen and preserved in 96% ethanol until helminthological examination. Amphibians were dissected and examined under stereomicroscope; worms were recovered and preserved in 70% ethanol for morphological study.
Platyhelminths were stained with Mayer´s paracarmine and Delafield´s hematoxylin, and permanently mounted in Canada balsam. Nematodes were cleared with a mixture of glycerine and ethanol solution 1:1, for 24 h and mounted between coverslips. In the case of the new species description, measurements are given in millimetres (unless otherwise indicated), with minimum and maximum, mean and standard deviation in parentheses. Morphometric data were obtained with an Olympus CX40 optical microscope; micrographs were taken with a Leica DM750 optical microscope equipped with a digital camera Leica ICC50 E. Line drawings were made on the basis of digital micrographs using a Wacom One drawing tablet with Adobe Illustrator CS6 and Adobe Photoshop CS6. Scale bars of all figures are given in micrometers. Specimens were deposited in the Colección Nacional de Helmintos, Instituto de Biología, Universidad Nacional Autónoma de México, Mexico City (CNHE).
Results
Six helminth species were collected during this study, 1 digenean, Rauschiella poncedeleoniRazo-Mendívil & León-Règagnon, 2001 and 5 nematodes, Aplectana incerta Bravo-Hollis, 1943, Cosmocerca colimense n. sp., Oswaldocruzia subauricularis (Rudolphi, 1819), Oswaldocruzia pipiens Walton, 1929, and Rhabdias sp. Rauschiella poncedeleoni (prevalence 5.8%), C. colimense n. sp. (31.4%), O. subauricularis (2.2%), and Rhabdias sp. (1.45%) were collected from L. melanonotus, while A. incerta (38.7%) and O. pipiens (6.4%) were found in I. marmoreus.
Description
Family Cosmocercidae Railliet, 1916 (Travassos, 1925)
Genus Cosmocerca Diesing, 1861
Cosmocerca colimense n. sp. (Figs. 1, 2)

Figure 1 Drawings of Cosmocerca colimense n. sp. A, Male, entire, lateral view; B, female, entire, lateral view; C; male, detail of the cloacal region, lateral view. Scale bars: A = 100 µm, B = 150 µm, C = 25 µm.
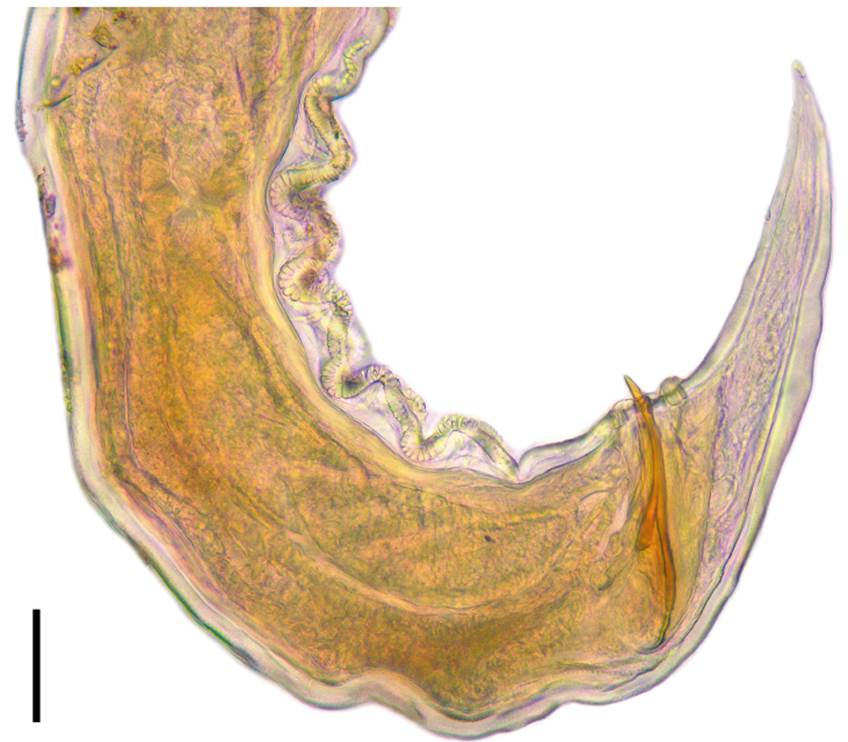
Figure 2 Digital photography of Cosmocerca colimense n. sp. Male. Detail of the cloacal region, lateral view. Scale bar = 50 µm.
http://zoobank.org/urn:lsid:zoobank.org:act:C8ADE45C-E6FF-4380-8B90-6854F9DF6EBE
Diagnosis. Robust, small and fusiform nematodes, males smaller than females (Fig. 1A, B). Cuticle with transverse striations along the entire body. Oesophageal bulb well developed, with sclerotized valves (Fig. 1A, B). Protruding excretory pore, anterior to oesophageal bulb (Fig. 1A, B). Females with lateral alae (Fig. 1B), absent in males. Caudal alae absent. Spicules equal in length, with a conical, sclerotized gubernaculum (Figs. 1C, 2). Females with a monodelphic-prodelphic reproductive system (Fig. 1B), without larvated nor undeveloped eggs, only with free larvae.
Male (based on 3 mature specimens). Fusiform nematodes with robust, small and cylindrical body, ventrally curved, anteriorly truncated by the obtuse oral region and acuminated at posterior end. Finely transversely striated cuticle along the entire body, without caudal alae. Total body length 1.872-2.043 (1.944 ± 0.088, n = 3), maximum width 0.135-0.198 (0.171 ± 0.032, n = 3) at middle region. Lateral alae absent. Oesophagus divided into an anterior and comparatively short pharynx, measuring 0.022-0.028 (0.025 ± 0.003, n = 3) in length, a sub-cylindrical corpus 0.179-0.222 (0.198 ± 0.021, n = 3) long, a well-defined isthmus 0.033-0.042 (0.039 ± 0.005, n = 3) long and a sub-pyriform oesophageal bulb with sclerotized valves measuring 0.076-0.058 (0.067 ± 0.009, n = 3) wide and 0.057-0.075 (0.066 ± 0.008, n = 3) long. Nerve ring located at 0.144-0.155 (0.151 ± 0.005, n = 3) from anterior end. Evident excretory pore, slightly protruding, located near to the anterior margin of oesophageal bulb, at 0.255 (0.243 ± 0.023, n = 3) from anterior end. Cloacal opening at 0.147-0.162 (0.159 ± 0.011, n = 3) from posterior end; this region is markedly muscular and ventrally curved, with 5 pairs of pre-cloacal plectanes each one forming 2 rows of 16-18 cuticular tubercles (1 in the anterior side and 1 in the posterior side) with 1 central papilla rounded by 8-10 internal punctuations and 9-10 external punctuations. Three pairs of rosettes (1 pre-cloacal, 1 ad-cloacal and 1 post-cloacal), and several pairs of small sub ventral simple papillae at surface of cloacal region: 17-18 ventral, 4 lateral and 9 dorsal pairs. Both spicules 0.071-0.091 (0.082 ± 0.010, n = 3) in length, slightly sclerotized and tapering in point, with a well sclerotized gubernaculum, conical in shape, measuring 0.097-0.124 (0.117 ± 0.010, n = 3) in length.
Female (based on 5 gravid specimens). Fusiform nematodes with robust and cylindrical body, usually ventrally curved at middle level, anteriorly truncated by the obtuse oral region and subulated at posterior end. Total body length 2.678-3.53 (3.185 ± 0.383, n = 5), maximum width 0.277-0.495 (0.353 ±0.098, n = 5) at vulva level. Finely transversely striated cuticle along the whole body, with lateral alae beginning at isthmus level, at 0.199-0.277 (0.234 ± 0.039, n = 4) from the anterior end, and ending at the posterior region at 0.459-0.577 (0.523 ± 0.059, n = 4) from de posterior end, slightly anterior to the anus. Oesophagus is divided into an anterior and short pharynx, measuring 0.024-0.039 (0.031 ± 0.006, n = 5) in length, a sub-cylindrical corpus 0.235-0.277 (0.256 ± 0.019, n = 5) long, a well-defined isthmus 0.044-0.055 (0.047 ± 0.005, n = 5) long and a sub-pyriform oesophageal bulb with sclerotized valves, measuring 0.055-0.077 (0.064 ± 0.010, n = 5) wide, and 0.075-0.099 (0.086 ± 0.009, n = 5) long. Nerve ring located at 0.155-0.261 (0.187 ± 0.049, n = 5) from anterior end. Evident excretory pore, noticeably protruding from, located about the level of corpus basis, between the nerve ring and oesophageal bulb, at 0.207-0.360 (0.281 ± 0.062, n = 5) from anterior end. Vulva located at the middle region, at 1.008-1.395 (1.196 ± 0.169, n = 5) from anterior end, with slightly dilated lips. Short and muscular vagina, with a monodelphic-prodelphic reproductive system, without larvated nor undeveloped eggs, only with numerous free larvae.
Taxonomic summary
Type host: Leptodactylus melanonotus (Hallowell, 1861). Representative specimens deposited at Colección Nacional de Anfibios y Reptiles, Instituto de Biología, Universidad Nacional Autónoma de México (CNAR)
Site of infection: posterior intestine.
Type locality: Coquimatlán-Pueblo Juárez Road, Coquimatlán, in Colima, Mexico (19°9’39.43” N, 103°53’0.51” W).
Other localities: Río Colima, Arroyo Pereyra, Río Manríquez, Colima, Río Comala, Comala, in Colima, Mexico.
Prevalence of infection: 43 of 137 examined (31.4%).
Type specimens deposited: CNHE 12079, holotype; CNHE 12080, paratypes.
Etymology: the name of the new species refers to the Mexican state of Colima, where the specimens were collected.
Remarks
Species of Cosmocerca are common intestinal parasites associated with amphibians and reptiles worldwide. To date, 38 nominal species have been assigned to this genus (Alcántara et al., 2022); with this study, this number increases to 39 species. Of these, 35 are associated with amphibians, and 4 with reptiles (Table 2). The biogeographical realm with the highest number of described species is the Neotropical realm, with 13 species, followed by the Oriental and the Palearctic realms with 7 and 5 species, respectively (Table 2).
Table 2 Geographical, biological and morphometric characteristics relevant to the taxonomy of nominal species in Cosmocerca.
| Geographical realm Species | Type host group | Body length mm | Spicules µm | Gubernaculum µm | Plectane pairs | Lateral alae | Reference (description) |
|---|---|---|---|---|---|---|---|
| Afrotropical | |||||||
| C. daly | Anura | 1-2.7 | 61-102 | 60-89 | 5 | Present | Harnoster et al. (2022) |
| C. makhadooensis | Anura | 1.50-2.29 | 38-103 | 98-110 | 5 | Present | Harnoster et al. (2022) |
| C. monicae | Anura | 1.77-2.13 | 87-107 | 100-108 | 5 | Present | Harnoster et al. (2022) |
| Australian | |||||||
| C. archeyi | Anura | 1.77 | 95 | 87 | 4 | Present | Baker and Green (1988) |
| C. lymnodynastes | Anura | 1.6 | 50-75 | 110 | 5 | Absent | Johnston and Simpson (1942) |
| C. multipapillata | Anura | 3.10-3.55 | 169-219 | 125-146 | 10-12 | Present | Ni et al. (2020) |
| C. australis | Anura | 1.28-1.78 | 99-123 | 82-88 | 4 | Present | Baker and Green (1988) |
| Nearctic | |||||||
| C. acanthurum | Caudata | 2.18-2.38 | 51-66 | 75-90 | 5 | Present | Falcón-Ordaz et al. (2007) |
| Neotropical | |||||||
| C. colimense n. sp. | Anura | 1.87-2.04 | 71-91 | 97-124 | 5 | Absent | Present study |
| C. paraguayensis | Anura | 1.33-1.79 | 83-108 | 106-122 | 5 | Present | Baker and Vaucher (1984) |
| C. rara | Anura | 2.8 | 200 | 206 | 6 | Present | Baker and Vaucher (1984) |
| C. albopunctata | Anura | 6.25 | 89.6-99.8 | 119.7-157.8 | 7 | Present | Alcántara et al. (2022) |
| C. gymnophthalmicola | Squamata | 2.2 | 266 | 108 | 6 | Absent | Ávila and Da Silva (2019) |
| C. podicipinus | Anura | 2.97 | 94 | 134 | 5 | Present | Baker and Vaucher (1984) |
| C. brasiliensie | Anura | 2.24-3.99 | 158-185 | 140-156 | 8 | Absent | Dyer and Altig (1976) |
| C. cruzi | Anura | 2.99 | 74 | 85 | 5 | Absent | Rodrigues and Fabio (1970) |
| C. parva | Anura | 1.43-2.01 | 90-110 | 85-108 | 5-7 | Present | Mordeglia and Digiani (1998) |
| C. travassosi | Anura | 3.66 | 167 | 130 | 5 | Absent | Rodrigues and Fabio (1970) |
| C. vrcibradici | Squamata | 1.62-1.86 | 171-183 | 92-98 | 7 | Present | Bursey and Goldberg (2004) |
| C. chilensis | Anura | 1.4 | 80 | 84 | 6 | Absent | Lent and Freitas (1948) |
| C. uruguayensis | Anura | 2.42 | 155 | 133 | 7 | Absent | Lent and Freitas (1948) |
| Oceanian | |||||||
| C. novaeguineae | Anura | 1.90 | 69 | 111 | 5 | Present | Moravec and Sey (1990) |
| C. oroensis | Anura | 0.77-0.99 | 37-52 | 24-29 | 4 | Present | Bursey et al. (2013) |
| C. tyleri | Anura | 1.02-1.43 | 37-43 | 58-61 | 9 | Present | Bursey et al. (2006) |
| C. zugi | Squamata | 1.63-1.84 | 79-116 | 92-122 | 4 | Present | Bursey et al. (2005) |
| Oriental | |||||||
| C. asansolensis | Anura | 2.09-3.89 | 120-150 | 100-102 | 5 | Present | Banerjee and Sou (2020) |
| C. bengalensis | Anura | 1.90-2.69 | 100-130 | Absent | 5 | Present | Sou et al. (2018) |
| C. ishaqui | Anura | 2.92 | 100 | Absent | 11 | Absent | Islam et al. (1981) |
| C. simile | Anura | .93-2.71 | 79-99 | 50-69 | 5-6 | Present | Chen et al. (2020) |
| C. kalesari | Anura | 1.11-1.92 | 82-90 | 85-90 | 5 | Present | Rizvi et al. (2011) |
| C. microhylae | Anura | 0.74-0.79 | 100-112 | 65-68 | 5 | Present | Sou and Nandi (2015) |
| C. leytensis | Squamata | 1.91 | 110 | 79 | 4 | Present | Bursey et al. (2015) |
| Palearctic | |||||||
| C. banyulensis | Anura | 0.97 | 10 | 80 | 5-6 | Present | Chabaud and Campana (1955) |
| C. commutate | Anura | 4.03-4.39 | 180 | 186-213 | 7 | Present | Moravec and Vojykova (1974) |
| C. longicauda | Caudata | 2.90 | 92 | 190 | 6 | Present | Skrjabin et al. (1961) |
| C. ornata | Anura | 1.1-2.8 | Rudimentary | 100-120 | 5 | Present | Skrjabin et al. (1961) |
| C. sardiniae | Caudata | 3.41-4.18 | 375 | 260-303 | 4 | Absent | Ricci (1987) |
| Panamanian | |||||||
| C. longispicula | Anura | 1.52-2.38 | 294-300 | 138 | 7 | Present | Moravec and Kaiser (1994) |
| Sino-Japanese | |||||||
| C. japonica | Anura | 1.8 | 104 | 84 | 5 | Present | Yamaguti (1938) |
Until the present study, only 3 species of Cosmocerca had been recorded in Mexico (C. acanthurumFalcón-Ordaz, Winfield-Pérez, Mendoza-Garfias, Parra-Olea & Pérez-Ponce de León, 2007, C. parvaMordeglia & Digiani, 1998, and C. podicipinusBaker & Vaucher, 1984), being the new species the second described in the country. Cosmocerca colimense n. sp. shares the number of plectane pairs and the absence of lateral alae with C. travassosiRodrigues & Fabio, 1970 and C. cruziRodrigues & Fabio, 1970 from the Neotropical realm, and with C. lymnodynastesJohnston & Simpson, 1942, from the Australian realm (Table 2). The new species differs from C. travassosi in the body length, being smaller in the new species (1.944 vs. 3.66). Regarding the male reproductive system, the spicules and gubernaculum are larger in C. travassosi than in C. colimense n. sp. (0.167 and 0.130 vs. 0.082 and 0.117, respectively). They also differ in the geographical distribution and the host group: C. travassosi was described in Brazil (Rio de Janeiro) as a parasite of Hyla faber (Rodrigues & Fabio, 1970). On the other hand, C. colimense n. sp. can be clearly distinguished from C. cruzi by the size of the spicules, which are larger in C. colimense n. sp. (0.082 vs. 0.074). Another differential character is the gubernaculum size, which is smaller in C. cruzi (0.08 vs. 0.117). Finally, one additional character that differentiates the 2 species is the position of the excretory pore; it opens at the level of the nerve ring in C. cruzi (Rodrigues & Fabio, 1970), but it is located near to the anterior margin of the oesophageal bulb in C. colimense n. sp. Regarding the Australian species, the new species can be differentiated from C. lymnodynastes on the basis of the body length, because this species is smaller than C. colimense n. sp. (1.6 vs. 1.94). Also, the spicules are slightly smaller in the Australian species than in C. colimense n. sp. (50-75 vs. 71-91). However, it should be noted that Johnston and Simpson (1942) indicated that the length of the spicules was not determined with certainty in C. lymnodynastes. Therefore, this character should be considered with caution. More evident is the difference in the gubernaculum shape, which is spicule-shaped in C. lymnodynastes, while it shows a conical shape in C. colimense n. sp.
Mata-López et al. (2013) reported C. podicipinus in L. melanonotus of Colima, but after examining the material deposited at the CNHE (4608-10) from that study, we observed that those specimens correspond to C. colimense n. sp., because males lack the lateral alae that is characteristic in C. podicipinus, and also differ from that species and are similar to C. colimense in the body, spicules and gubernaculum length, which are shorter in the new species (1.87-2.04 vs. 2.97; 71-91 vs. 94 µm; 97-124 vs. 134 µm, respectively). The absence of lateral alae in males also distinguishes the new species from C. acanthurum and C. parva, the other 2 species of Cosmocerca that have been recorded in Mexico. Body length is another characteristic that differentiates C. colimense n. sp. from C. acanthurum, being shorter in the new species (1.87-2.04 vs. 2.18-2.38); also, the spicules and gubernaculum are shorter in C. acanthurum (51-66 vs. 71-91 µm, and 75-90 vs 97-124 µm, respectively) (Table 2). On the other hand, C. colimense n. sp. differs from C. parva mainly in the length of the spicules, which is shorter in the new species (71-91 vs. 90 -100 µm) (Table 2); also, they differ in the shape of the gubernaculum, which is y-shaped in C. parva, and conical in C. colimense n. sp. (Mordeglia & Digiani, 1998).
Discussion
It is interesting to note that the helminth species richness found in this study was low compared with previous studies on these host species. Mata-López et al. (2013) recorded 8 helminth species (3 adult and 2 larval digeneans, 2 adult and 1 larval nematodes) in L. melanonotus in several localities of Colima, while we only found 1 digenean and 3 nematode species. In the case of I. marmoreus, previous record in Jalisco (Galicia-Guerrero et al., 2000) was similar in species richness (4 species vs. 3 in this study), but a study in Oaxaca resulted in a 10 taxa record. This could be the result of several factors, including the massive growth of urban and agricultural areas in recent years in Colima, with the consequent pollution of streams and rivers (Arceo et al., 2023; Villaseñor-Cortés et al, 2014). This condition affects mostly those helminths with complex life cycles; this could be the case of digeneans of the genus Gorgoderina (reported by Mata-López et al. [2013], and absent in this study) that use aquatic intermediate hosts and present free swimming larval stages (Velázquez-Urrieta & Pérez-Ponce de León, 2021). Also, in the Summer of 2021, Colima registered the highest volumes of precipitation in decades, 2,280 mm, the highest cumulative annual amount of all entities in Mexico during that year (Zavala-Fajardo, 2022), which resulted in high water levels and speed in the streams and rivers, washing away entire populations of amphibians. This also may have affected the intermediate hosts populations, such as molluscs or aquatic insects, and consequently, the recruitment of helminths in that season. On the other hand, nematode species, which have a direct life cycle as Cosmocerca colimense n. sp. (Kirillova & Kirillov, 2021), and which larval stages penetrate the amphibians´ skin directly, were widely distributed across the collecting sites.











 nueva página del texto (beta)
nueva página del texto (beta)


