Hechtioideae is one of eight subfamilies of Bromeliaceae (Givnish et al. 2014); it comprises three genera; Bakerantha L.B. Sm., Hechtia Klotzsch and Mesoamerantha I. Ramírez & K. Romero (Ramírez-Morillo et al. 2018b), and at least 84 species formally recognized (bromeliad.nl/encyclopedia), albeit there are several more yet to be proposed as new. These plants have terrestrial and lithophytic habits, usually growing on volcanic or karst rocks. They all grow as a rosette and have distinctive growth patterns (inflorescence origin, production of new shoots, etc.; Ramírez-Morillo et al. 2014), giving them a wide variety of sizes and shapes (Figure 1). They usually grow in open areas, rocky outcrops, and cliffs, and are often found in xeric shrublands, deciduous tropical forests, and, occasionally, oak-pine forests (López-Ferrari & Espejo-Serna 2014a, Ramírez-Morillo et al. 2014).

Figure 1 Morphological characteristics of Hechtioideae and examples of habitats where it is present. A) Bakerantha purpusii growing on cliff. B) Hechtia schottii. C) H. laevis. D) Mesoamerantha dichroantha growing on rocks. Image credits: A, D by K. Romero-Soler; B, C by I. Ramírez-Morillo.
According to Givnish et al. (2014), this Neotropical lineage may have emerged 16.3 Ma ago, reaching the biogeographic region of Megamexico III, proposed by Rzedowski (1991), where the most recent lineages differentiated 10.3 Ma ago, through long-distance dispersal across the Caribbean or through Central America. Its current range is restricted to this region, stretching across a territorial extension ranging from the deserts of Chihuahua and Sonora south to northern Nicaragua. More than 90 % of the species are found only in Mexico (Ramírez-Morillo et al. 2018a, Espejo-Serna et al. 2020). In the only biogeographic study carried out for this group, Pech-Cárdenas (2015) identified that the biogeographic provinces with the greatest specific richness of Hechtioideae are the Pacific Lowlands, Sierra Madre del Sur, Veracruz, Balsas Basin, and Mexican Highland provinces. Most species have restricted distribution ranges and high endemism rates (Ramírez-Morillo et al. 2018b); accordingly, most of these species have been assessed as under some risk category according to the criteria of the International Union for Conservation of Nature (IUCN) (Pech-Cárdenas 2015, Ramírez-Morillo et al. 2016, Ramírez-Morillo et al. 2020, Romero-Soler et al. 2022).
In recent years, efforts have been made to determine the evolutionary history of this group of plants by analyzing their phylogenetic relationships and the description and delimitation of their species (García-Ruiz et al. 2014, González-Rocha et al. 2014, López-Ferrari & Espejo-Serna 2014a, Ramírez-Morillo et al. 2014, Ramírez-Morillo et al. 2015, Ramírez-Morillo et al. 2016, Flores-Argüelles et al. 2019, Hernández-Cárdenas et al. 2019, Hernández-Cárdenas et al. 2020, Romero-Soler et al. 2022). These advances have made possible to identify the monophyly of Hechtioideae and the presence of three well-supported clades corresponding to three genera; Bakerantha with five species, Hechtia with ca. 76 species, and Mesoamerantha with three species (Ramírez-Morillo et al. 2018b, Romero-Soler et al. 2022). However, there are still evolutionary and biogeographic aspects that have not been fully studied, for example, the diversification process in the lineage and the identification of areas of higher or lower diversity. The answers to these questions may improve our understanding of the events that have shaped the evolutionary history of the group and identify and propose areas for the conservation of these threatened plants.
The analysis of biogeographic patterns at different scales has enabled us to test hypotheses to explore ecological and evolutionary processes influencing the conformation of the biota (Steinbauer et al. 2016). For a long time, studies aimed to identify these patterns by analyzing taxonomic diversity, with the species level as the basic and equivalent unit for measuring biological diversity only, and disregarding the evolutionary, functional, and morphological characteristics that make lineages different (Hillebrand et al. 2018). Thus, phylogenetic diversity (PD) arises as a statistic that allows the analysis of biological diversity considering the evolutionary relationships and cumulative changes in each species over time (Faith 1992). This concept has given rise to multiple indices and methodologies where the phylogenetic distance measurement is used as a tool to identify the historical and environmental processes that give rise to biotic assemblages (Webb et al. 2002).
Although the incorporation of phylogenetic information into the study of spatial diversity is relatively recent, several hypotheses have already been proposed about biogeographic patterns and biotic assemblage mechanisms that consider evolutionary relationships (Faith 1992, Faith et al. 2010, Morlon et al. 2011). Similarly, conservation biology has used this new phylogenetic approach to maximize conservation efforts focused on safeguarding not only as many species as possible, but also the greatest extent of evolutionary history (Purvis et al. 2005, Franke et al. 2020). Previously, the species richness of Hechtioideae had been analyzed across the biogeographic provinces within their geographic range (Pech-Cárdenas 2015). However, multiple species have been described since then, and a fairly robust phylogenetic hypothesis is now available to implement metrics that assess the spatial pattern of lineages by incorporating the evolutionary relationships within the group.
The present research assesses the spatial phylogenetics of the subfamily Hechtioideae across its distributional range. Our goals were to analyze the phylogenetic assemblage processes in the Hechtioideae throughout biogeographic areas and recognize key areas for its conservation.
Materials and methods
Geographic distribution data and areas for assessment. Georeferenced records of the Hechtioideae were obtained from a comprehensive literature revision that included protologues of all the species in the group, general and specific taxonomic treatments for particular states of Mexico and natural areas (Pulido-Esparza et al. 2004, Espejo-Serna et al. 2005, Espejo-Serna et al. 2007, Espejo-Serna et al. 2010, López-Ferrari & Espejo-Serna 2014a, González-Rocha et al. 2016, Espejo-Serna et al. 2017, Espejo-Serna et al. 2020), and academic theses on taxonomy and phylogenetics of Hechtioideae (Martínez-Correa 2008, Jiménez-Nah 2014, Pech-Cárdenas 2015, Romero-Soler 2017). Besides, online databases such as SEINet (2021) were surveyed through the Herbaria Network of Northwest Mexico (herbanwmex.net), Tropicos v. 3.2.3 (tropicos.org), and the National Autonomous University of Mexico Open Data University Collection website (datosabiertos.unam.mx). Also, multiple herbarium collections have been reviewed, such as (AAU, ARIZ, ASU, B, BH, BIGU, BM, BR, BRH, BRIT, C, CAS, CHAP, CHAPA, CHIP, CICY, CIIDIR, CIMI, CM, CODAGEM, CU, DES, DS, DUKE, EAP, ECON, ENCB, F, FCME, FEZA, G, GH, GOET, GUADA, HAL, HCIB, HEM, HGOM, HNT, HUAA, IBUG, IEB, IND, INEGI, IZTA, K, LAGU, LL, M, MEXU, MHES, MICH, MO, NMC, NY, OAX, P, PH, QMEX, RSA, SEL, SERO, SLPM, TEFH, TEX-LL, TTC, UAMIZ, UAT, UCAM, UJAT, US, USF, USON, VT, W, WIS, WU, XAL, Z, ZEA) (acronyms following Thiers (cont. updated)). Records with no exact coordinates, but with detailed descriptions of the collection site were georeferenced with the program Google Earth Pro v. 7.3.3.7786 (Google 2020). A total of 94 species were considered; of these, five have not yet been described but were included because there is sufficient molecular and morphological information available to consider them in the phylogenetic analysis.
The geographic range of the Hechtioideae was divided into three areas, namely the Nearctic, Neotropical, and Mexican Transition Zone biogeographic regions (sensuMorrone et al. 2017); then, the biogeographic provinces proposed by Morrone (2014) and Morrone et al. (2017) were considered as sub-areas of these regions.
Estimation of divergence times. A phylogenetic hypothesis was constructed for 75 species (out of the 94 species recognized in this study) of Hechtioideae. We included sequences for 50 species of Hechtioideae from chloroplast (the intergenic spacer rpl32-trnL and the ycf1 gene) and nuclear (PRK gene) DNA regions previously generated by Ramírez-Morillo et al. (2018a). We added sequences for other 25 species using the same primers and DNA extraction and amplification protocols as Ramírez-Morillo et al. (2018a). All PCR products were sent to MACROGEN (Seoul, South Korea) for sequencing and were deposited in GenBank (Table S1).
The sequences generated were reviewed and assembled in Geneious v. 7.1.9 (Kearse et al. 2012). The sequences were aligned automatically using MAFFT v. 7.017 (Katoh et al. 2002), implemented in Geneious; manual alignment was performed, as needed, using PhyDe v. 0.9971 (Müller et al. 2010). The species tree and its divergence times were estimated with BEAST v. 1.10.4 (Suchard et al. 2018). The nucleotide substitution model implemented was the one suggested by jModelTest v. 2.1.7 (Darriba et al. 2012), GTR + G for chloroplast regions, and HKI + I + G for the PRK gene. The analysis was performed using a relaxed clock model with an uncorrelated lognormal distribution, with the Yule speciation model. Because of the lack of fossils records for the family Bromeliaceae, we used previously estimated dating times to calibrate the phylogeny, using the crown group age of Hechtioideae, at 10.3 Mya (SD ± 1) as estimated by Givnish et al. (2011). Two runs of 50,000,000 generations were performed, sampling every 5,000 generations. The analyses were run through the CIPRES Science Gateway (Miller et al. 2010). After the BEAST analyses, the log and tree files were pooled using LogCombiner v. 1.10.4 and summarized as the maximum credibility tree with TreeAnnotator v. 1.10.4, with a burn-in of 25 %. The results were reviewed with Tracer v. 1.6 (Rambaut et al. 2014) to verify the convergence and effective sample size (ESS) > 200.
Additionally, 19 species with no information for any molecular sequence were included manually to counter the fact that the lack of lineages underestimated the true phylogenetic pool for each set of species as highlighted by Sandel (2018). To this end, we identified those clades including species with geographic affinity in relation to missing taxa, as the Hechtioideae shows strong patterns of endemism and geographic restriction (Ramírez-Morillo et al. 2018a). The species were linked to the basal node of each respective clade. This addition produced some polytomies, all of them in the terminal parts of the tree. This methodology helps to compensate for the loss of phylogenetic information derived from an incomplete phylogenetic hypothesis of the group.
Diversity metrics and phylogenetic structure. Diversity in the Hechtioideae was calculated using the species richness (Gotelli & Chao 2013), the weighted endemism (Guerin et al. 2015), the phylogenetic diversity (Faith 1992), and the weighted phylogenetic endemism indices (Rosauer et al. 2009, Laity et al. 2015, Laffan et al. 2016), available in Biodiverse v. 3.1 (Laffan et al. 2010).
Phylogenetic structure was calculated using the standardized effect size phylogenetic diversity (Proches et al. 2006, Webb et al. 2008), net relatedness index (NRI), and nearest taxon index (NTI) (Webb et al. 2002, 2008), all available in the package Picante (Kembel et al. 2010), in R language (R Core Team 2020) via the RStudio v. 1.2.1335 interface (www.rstudio.com). These indices consider the average phylogenetic distance between pairs of taxa; therefore, they do not apply for areas with less than two species.
Ancestral area reconstruction. The reconstruction of ancestral areas was carried out with RASP v. 4.2 (Yu et al. 2015). The Nearctic, Neotropical, and Mexican Transitional Zone biogeographic regions were selected as the reconstruction states. The reconstruction model implemented was Bayesian Binary MCMC (BBM) (Ronquist & Huelsenbeck 2003, Ali et al. 2012). The potential ancestral ranges were estimated for each node of the phylogenetic tree. The Hechtioideae chronogram was used as a consensus tree. The number of maximum areas remained at three. Ten MCMC chains were run simultaneously for 5,000,000 generations and the reconstructed state was sampled every 1,000 generations. The fixed model JC + G (Jukes-Cantor + Gamma) was used in BBM analysis with a null root distribution.
Results
Geographic distribution. A total of 2,515 records of Hechtioideae were gathered, of which 220 (8.7 %) belonged to the genus Bakerantha, 2,040 (81.1 %) to the genus Hechtia, and 255 (10.2 %) to the genus Mesoamerantha. These records identified the distribution range of Hechtioideae, which is restricted to the Megamexico III biogeographic region (Figure 2). Hechtia is the genus with the widest distribution within the Hechtioideae, spreading from the south of the USA throughout the Mexican territory south to at least Honduras, whereas the genera Bakerantha and Mesoamerantha have more restricted distributions, covering certain areas in central Mexico and Central America, respectively.
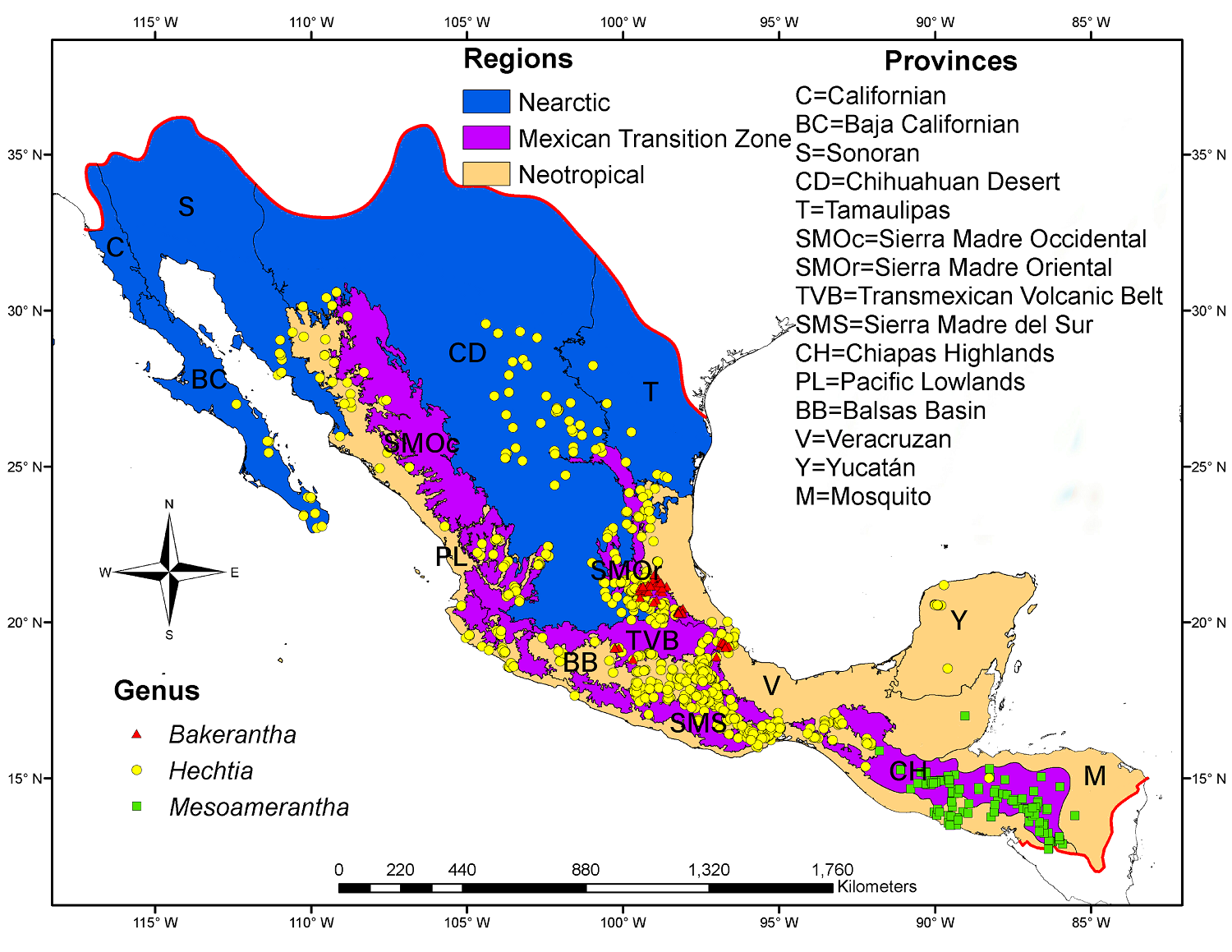
Figure 2 Territorial extension of Megamexico III (demarcated by red lines) and the different provinces that make up the Nearctic and Neotropical biogeographic regions, as well as the Mexican Transition Zone within the area. The dots on the map correspond to records considered to determine the distribution range of each species.
Estimation of divergence times. The chronogram generated for Hechtioideae includes 94 of the 95 species in the group (Figure 3). The only species that was not included in the phylogenetic analysis was Hechtia reticulata L.B. Sm. because the identity of this species is still uncertain due to the fragmentary type material; this makes it difficult to relate it to any other species.
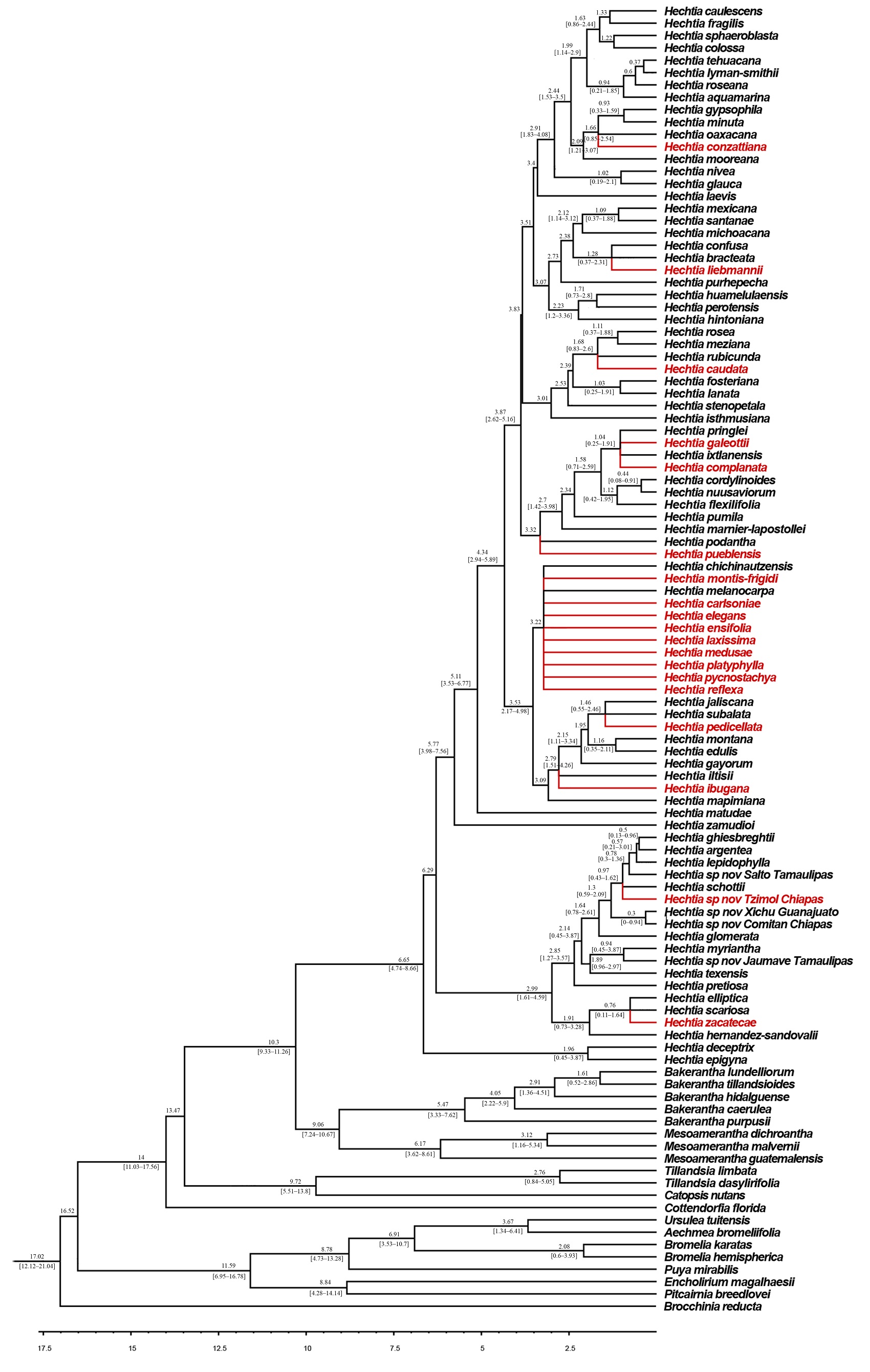
Figure 3 Chronogram for Hechtioideae derived from the Bayesian analysis of chloroplast (ycf1 and rpl32 - trnL) and nuclear (PRK) regions; the species included manually are marked in red. Values above branches indicate the average age of each node; values below branches indicate the 95 % highest posterior density intervals.
According to the chronogram recovered from the phylogenetic analyses, the ancestral lineage of Hechtioideae underwent a diversification process during the Mid-Miocene, with a crown age of 10.29 Ma, which gave rise to two large lineages. One corresponds to the ancestral lineage of Hechtia, which has a crown age of 6.65 Ma; its diversification process started in the late Miocene with a peak diversification rate during the Pliocene and Pleistocene (5-1 Ma). Currently, this lineage includes 87 species clustered in various clades. The other large lineage corresponds to the ancestor of the Bakerantha-Mesoamerantha clade, which has a crown age of 9.05 Ma and whose diversification occurred during the late Miocene. The Mesoamerantha lineage has a crown age of 6.16 Ma and its diversification occurred during the late Miocene-Pliocene; it currently includes three species. Finally, the Bakerantha lineage has a crown age of 5.47 Ma and its diversification occurred during the Pliocene-Pleistocene; it currently includes five species.
Diversity metrics and phylogenetic structure. At the regional level, species richness (SR = 59) and weighted endemism (WE = 44.50) are higher in the Neotropical region (NT) (Table 1). The Mexican Transition Zone (MTZ) includes 56 species and has the highest values of phylogenetic diversity (PD = 166.35) and phylogenetic weighted endemism (PWE = 110.94) (Table 1). The Nearctic region (NA) includes 13 species and has the lowest values of weighted endemism (WE = 9), phylogenetic diversity (PD = 37.72), and phylogenetic weighted endemism (PWE = 19.85) (Table 1).
Table 1 Species richness (SR), weighted endemism (WE), phylogenetic diversity (PD), phylogenetic weighted endemism (PWE), standard effect size of phylogenetic diversity (SES.PD), net relatedness index (NRI), and nearest taxon index (NTI) of each area assessed (regions and provinces). For SES.PD, NRI, and NTI, the asterisk indicates statistical significance, where P < 0.05 (clustering) or P > 0.95 (overdispersion).
| Regions | Provinces | SR | WE | PD | PWE | SES.PD | NRI | NTI |
|---|---|---|---|---|---|---|---|---|
| Nearctic | 13 | 9 | 37.72 | 19.85 | -3.15* | 1.79* | 2.78* | |
| Californian | 0 | 0 | 0 | 0 | - | - | - | |
| Baja Californian | 2 | 1.20 | 12.44 | 3.37 | -1.43* | 1.46* | 1.40* | |
| Sonoran | 1 | 0.20 | 10.29 | 1.22 | - | - | - | |
| Chihuahuan Desert | 11 | 6.06 | 34.78 | 11.70 | -2.86* | 1.69* | 2.62* | |
| Tamaulipas | 3 | 1.83 | 15.42 | 4.04 | -1.61* | 1.62* | 1.56* | |
| Mexican Transition Zone | 56 | 39.50 | 166.35 | 110.94 | 0.33 | -1.87* | 0.63 | |
| Sierra Madre Occidental | 3 | 0.90 | 16.59 | 2.91 | -1.26* | 1.13 | 1.43* | |
| Sierra Madre Oriental | 17 | 11.36 | 63.51 | 33.57 | -0.88 | -1.19 | 2.14* | |
| Transmexican Volcanic Belt | 11 | 7.53 | 53.02 | 25.86 | 0.34 | 0.41 | -1.09 | |
| Sierra Madre del Sur | 23 | 15.50 | 58.10 | 30.54 | -4.01* | 4.22* | 3.00* | |
| Chiapas Highlands | 5 | 3.58 | 36.17 | 14.80 | 1.63 | -2.08* | -0.86 | |
| Neotropical | 59 | 44.50 | 159.12 | 106.71 | -1.45 | 2.23* | 0.82 | |
| Pacific Lowlands | 24 | 15.98 | 72.51 | 34.35 | -1.90* | 2.48* | 1.31 | |
| Balsas Basin | 29 | 20.83 | 84.99 | 50.48 | -2.34* | 3.21* | 1.16 | |
| Veracruzan | 12 | 6.75 | 54.57 | 20.01 | 0.10 | -1.71* | 0.72 | |
| Yucatán | 1 | 1 | 10.29 | 2.17 | - | - | - | |
| Mosquito | 1 | 0.25 | 10.29 | 2.44 | - | - | - |
At the biogeographic province level, the California province (C) does not harbor any species of Hechtioideae, while the Sonoras (S), Yucatan (Y), and El Mosquito (M) provinces each have a single species. The Balsas Basin (BB) is the province with the highest values of species richness (SR = 29), weighted endemism (WE = 20.83), phylogenetic diversity (PD = 84.99), and phylogenetic weighted endemism (PWE = 50.48) (Table 1). The Pacific Lowlands (PL) and Sierra Madre del Sur (SMS) provinces have high diversity values and include 24 and 23 species, respectively (Table 1). The Sierra Madre Oriental (SMOr) province has 17 species and has high phylogenetic diversity values (PD = 63.51). At the regional scale, NA has significant positive values for NRI (1.79, P = 0.01) and NTI (2.78, P = 0.0009) (Table 1). MTZ has a significant negative NRI (-1.87, P = 0.98) and a non-significant positive NTI (0.63, P = 0.24) (Table 1). Finally, NT has a significant positive NRI (2.23, P = 0.02) and a non-significant positive NTI (0.82, P = 0.20) (Table 1).
Considering biogeographic provinces as sub-areas of regions, the NRI/NTI values for the NA region are all positive and with minor variations (Figure 4A, 4C); the Baja Californian (BC) province has significant positive NRI (1.46, P = 0.03) and NTI (1.40, P = 0.04), as well as the Chihuahuan Desert (CD) (NRI = 1.69, P = 0.02; NTI = 2.62, P = 0.0009), and Tamaulipas (T) (NRI = 1.62, P = 0.01; NTI = 1.56, P = 0.02) (Table 1; Figure 4B, 4D). In addition, the MTZ provinces show a higher variation of NRI/NTI values (Figure 4A, 4C); the Transmexican Volcanic Belt (TVB) has a non-significant positive NRI (0.41, P = 0.35) and a non-significant negative NTI (-1.09, P = 0.85), Sierra Madre Occidental (SMOc) has a non-significant positive NRI (1.13, P = 0.07) and a significant positive NTI (1.43, P = 0.04), Sierra Madre Oriental (SMOr) has a non-significant negative NRI (-1.19, P = 0.88) and a significant positive NTI (2.14, P = 0.01), Sierra Madre del Sur (SMS) has significant positive NRI (4.22, P = 0.0009) and NTI (3, P = 0.0009), and Chiapas Highlands (CH) has a significant negative NRI (-2.08, P = 0.98) and a non-significant negative NTI (-0.86, P = 0.79) (Table 1; Figure 4B, 4D). Finally, the NRI values of the NT provinces show a considerable variation (Figure 4A, 4C), while NTI values are very similar (Figure 4C); the Balsas Basin (BB) has a significant positive NRI (3.21, P = 0.0009) and a non-significant positive NTI (1.16, P = 0.12), Pacific Lowlands (PL) has a significant positive NRI (2.48, P = 0.005) and a non-significant positive NTI (1.31, P = 0.11), finally, Veracruz (V) has a significant negative NRI (-1.71, P = 0.95) and a non-significant positive NTI (0.72, P = 0.25) (Table 1; Figure 4B, 4D).
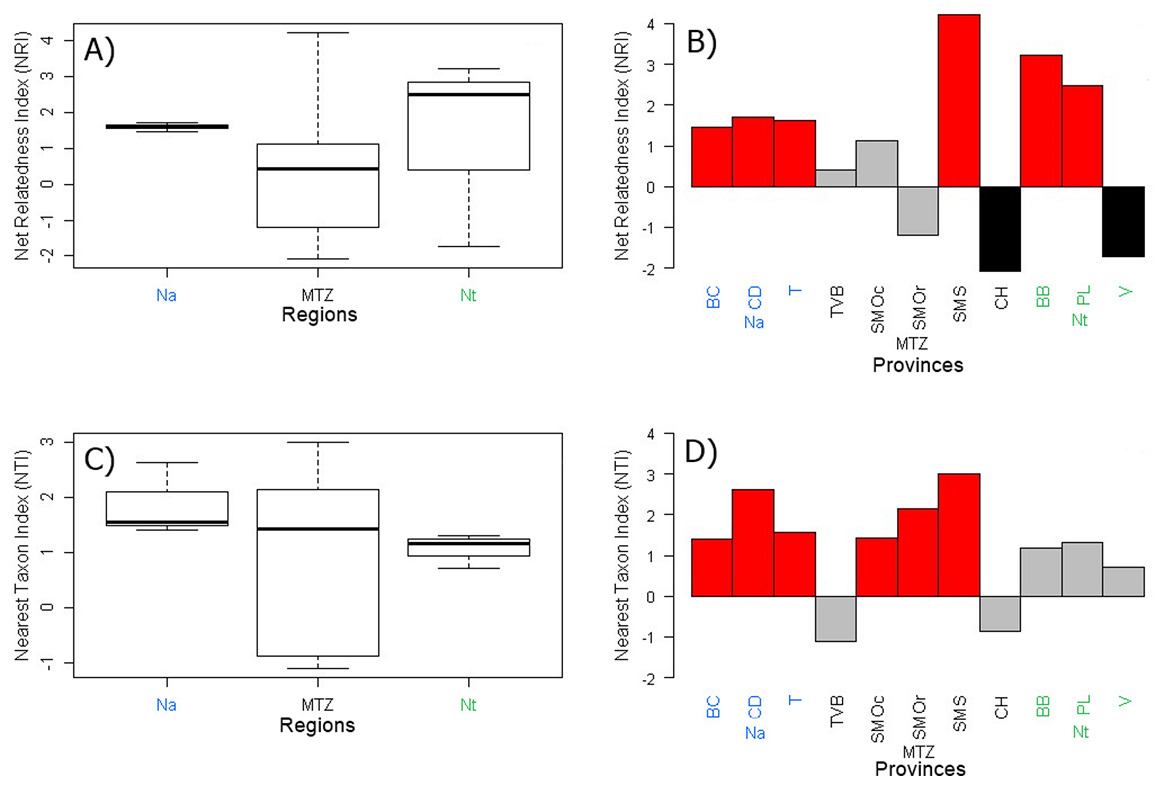
Figure 4 NRI/NTI values for each area. A) NRI values by biogeographic regions. B) NRI values by biogeographic provinces. C) NTI values by biogeographic regions. D) NTI values by biogeographic provinces. The values in the boxplot correspond to the results of each province belonging to the different regions. Positive values suggest phylogenetic clustering; negative values suggest phylogenetic overdispersion. Statistical significance is indicated by colors in bars: P < 0.05 = red bars, P > 0.95 = black bars.
Ancestral area reconstruction. The results of the Bayesian Binary Analysis MCMC (BBM) suggest that the biogeographical history of Hechtioideae has been complex, since the reconstruction of the ancestral area for many of the nodes (including the ancestor of all Hechtioideae) does not correspond to a single region but to combined areas (multi-areas). According to our results, the most likely ancestral area of the Hechtioideae lineage corresponds to a multi-area (BC) formed by the Mexican Transition Zone (B) and the Neotropical region (C) (abbreviations for each area indicated in Figure 5; BC = 66 %, B = 25.61 %, C = 8.39 %). It reached its current range through 60 dispersal events, 13 vicariance events, and 2 extinction events (Figure 5A). The most likely ancestral areas of the Mesoamerantha and Bakerantha lineages, also correspond to the multi-area BC (Mesoamerantha: BC = 68.68 %, B = 30.18 %, C = 1.14%; Bakerantha: BC = 74.11 %, C = 15.89 %, B = 10 %). On the other hand, the most likely ancestral area of the Hechtia lineage is MTZ (B = 93.11 %, BC = 6.89 %) and shows several dispersal events to different regions within the same MTZ, others to the NT region and, a few others to the NA region (Figure 5A). Thus, the ancestral area of most lineages within the genus Hechtia show shared ancestral areas between MTZ and NT. The reconstruction analysis reveals that the largest diversification in the group occurred around 3.5 Ma with an increase in the dispersal and vicariance rates, followed by a second increase about 1 Ma (Figure 5B).
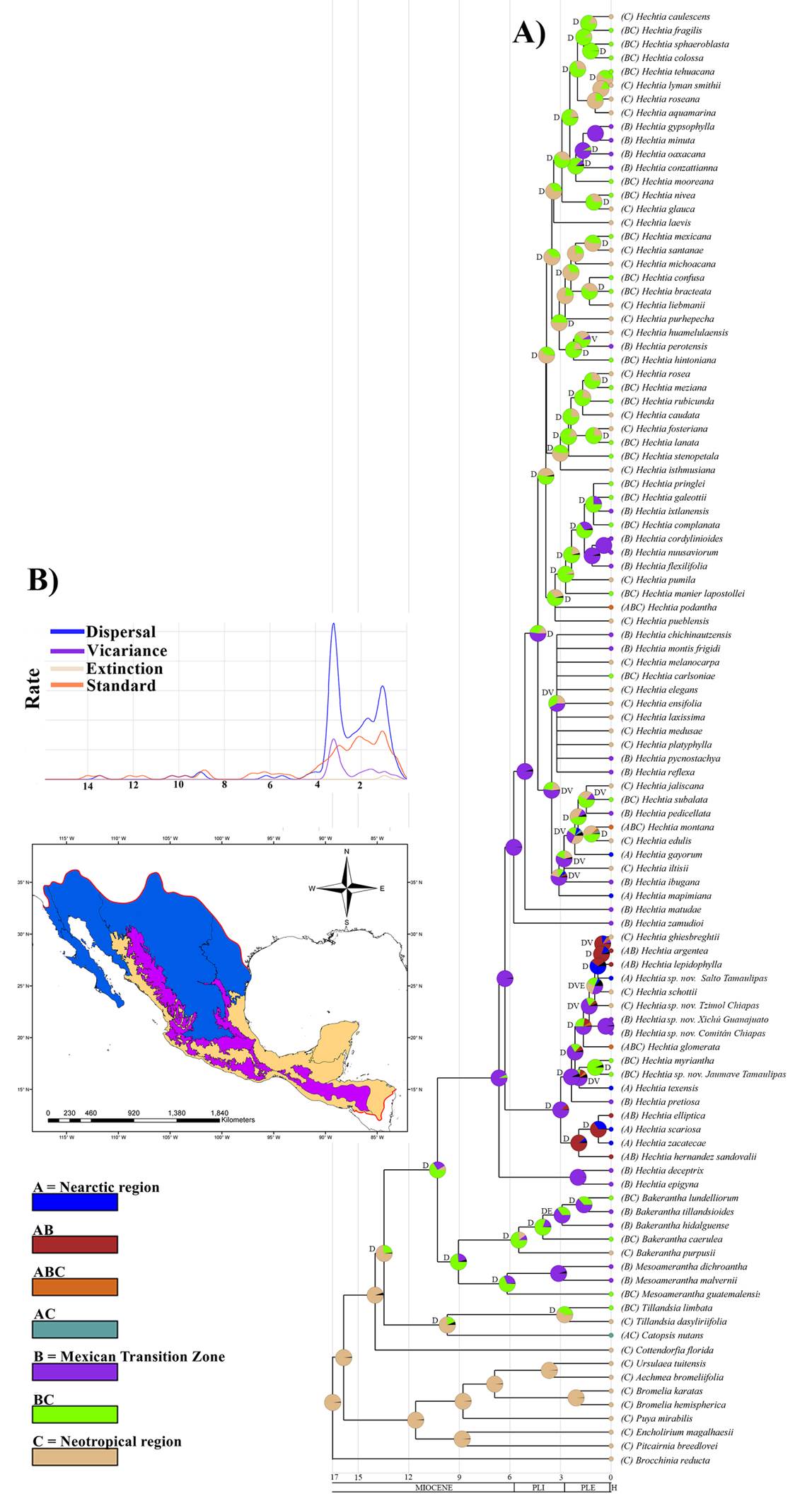
Figure 5 Ancestral area reconstruction of Hechtioideae with BBM in RASP. A) Reconstruction of ancestral areas by node; colors at the nodes of each pie chart indicate the percentage of reconstruction for each area. Letters in each node indicate: D = dispersion, V = vicariance, and E = extinction. B) Rate of dispersal, vicariance, and extinction events over time.
Discussion
This is the first biogeographic study of Hechtioideae from an evolutionary framework that contributes to understand the history and diversification of this lineage. Our results strongly support the hypothesis that the group is endemic to the Megamexico III biogeographic region, through the analysis of the spatial distribution of its species, as previously noted (Ramírez-Morillo et al. 2018a, Espejo-Serna et al. 2020). Distributional patterns of Hechtioideae through Megamexico III are not homogeneous as our results indicate that species are concentrated in certain areas and absent in many others.
Another methodological consideration regards the manual addition of species to the phylogenetic tree. Munguía-Rosas et al. (2011) point out that the manual addition of species to the phylogenetic analysis helps to compensate for the loss of underlying information in an incomplete phylogeny. In this study, we did not follow any previously reported protocols. Instead, we implemented a methodology of our own consisting of the insertion of each missing lineage at a specific location in the phylogenetic tree based on our best estimate of relationships and using geography and morphology as affinity indicators. This criterion is supported by documented knowledge that most Hechtioideae species have narrow distribution ranges, and the supported clades in the phylogeny have high levels of endemism (Ramírez-Morillo et al. 2018a). Altogether, this strongly suggests that species that are geographically close and morphologically similar most likely belong to the same lineage. We consider that this methodology overestimates the phylogenetic clustering values for some areas. However, when comparing the results of diversity and phylogenetic structure obtained in this analysis with those emerging from an analysis lacking these “added” species, we find similar, consistent patterns. In fact, the addition of species resulted in higher statistical support of probability values in the phylogenetic structure analyses. Of the 94 species considered in this study, only 19 (20 %) were added manually. The effects of this methodology in the analysis of spatial phylogenetics should be evaluated within a broader framework.
Late-Miocene speciation by dispersal. In general, the greatest diversification of Hechtioideae took place during the late Miocene and the Pleistocene (6.5-1 Ma) (Figure 3). This is consistent with the analysis of the dispersal and vicariance rates that increased during the late Pliocene (3.5 Ma), followed by a second peak during the Pleistocene (1 Ma) (Figure 5A). These periods were characterized by broad climatic fluctuations, mainly driven by the glacial and interglacial events of the Pleistocene, which led to the emergence of arid biomes in North America (Mastretta-Yanes et al. 2015), as well as a dynamic and complex geological activity. The latter was particularly evident in central and southern Megamexico III, e.g., the uprising of the Chiapas Highlands (CH) in the late Miocene and the Pliocene (Witt et al. 2012), the partial depression between the Sierra Madre del Sur (SMS) and CH in the late Miocene and early Pliocene (Barrier et al. 1998), and the great tectonic and volcanic activity that led to the formation of the Transmexican Volcanic Belt (TVB) from the Miocene to the present, with the greatest activity during the Pleistocene (Ferrari et al. 2012). These events modified the environmental conditions in the region and gave rise to different new habitats, promoting the expansion and reduction of the distribution of many species (Halffter et al. 2008). In turn, these boosted the diversification of multiple lineages, such as the Agave sensu lato group (Asparagaceae), with a speciation rate that increased in the late Miocene (6.18 Ma), most likely associated with the appearance of arid biomes in North America (Jiménez-Barrón et al. 2020), as well as the Fouqueriaceae lineage, whose origin is ancient (ca. 75.54 Ma), but its highest rate of diversification occurred during the Mio-Pliocene, associated with the origin of the deserts of North America (De-Nova et al. 2018); or the Milla clade (Brodiaeoideae, Asparagaceae), whose radiation and divergence has also been associated with the climatic fluctuations of the Mio-Pleistocene (Gándara et al. 2014).
Nearctic region. The Nearctic region (NA), located in the northernmost part of Megamexico III, is characterized by aridity and a mild-cold climate (Morrone 2019) and is home to only a few Hechtioideae species (SR = 13) (Table 1). This is because Hechtioideae, a group of Neotropical and MTZ origin, has no morphological adaptations to tolerate cold climates or severe frost events (Givnish et al. 2007, 2011, 2014). In addition, the lower-than expected PD values in all provinces in NA (Table 1) and the phylogenetic clustering pattern (NRI/NTI) (Figure 4) support the tropical conservatism hypothesis (Wiens & Donoghue 2004), which suggests that the standardized effect size of phylogenetic diversity (SES.PD) value tends to be negative as latitude increases (Kerkhoff et al. 2014). This pattern reflects the fact that climatic conditions at high latitudes have been unstable over time, favoring particular environmental conditions at different times; accordingly, species tend to exhibit phylogenetic clustering patterns resulting from an environmental filtering process (Webb et al. 2002). On the other hand, it has been documented that certain evolutionary processes, such as in-situ speciation, isolation of lineages, and recent divergence, also reflect phylogenetic clustering structures (Webb et al. 2002, Morlon et al. 2011). According to the phylogenetic analyses (Figure 3) and the reconstruction of ancestral areas (Figure 5A), Hechtioideae lineages in the NA region reached their highest diversification rates during the second half of Pleistocene (3.5 Ma), derived from dispersal and vicariance processes. It is highly likely that certain geological events, such as the formation of the Transmexican Volcanic Belt (TVB), isolated some lineages of central Megamexico III, and that these lineages settled in the NA region, where they subsequently diversified. Evidence supporting this hypothesis is the clustering of certain lineages in the NA region, as in the case of Hechtia argentea Baker, H. lepidophylla I. Ramírez and Hechtia sp. nov. from El Salto del Tigre, Tamaulipas, grouped in the same clade together with H. ghiesbreghtii Lem., H. schottii Baker and Hechtia sp. nov. from Tzimol, Chiapas, which are distributed in the NT region. Our results suggest that this clade underwent vicariance events at the end of the Pliocene (2 Ma). A similar pattern also occurs in the clade of H. gayorum L.W. Lenz, H. mapimiana López-Ferr. & Espejo and H. montana Brandegee, which are clustered together in the phylogenetic tree along with species distributed in NT and MTZ. Similarly, the results suggest that this clade underwent vicariance and dispersal events at the end of the Pliocene (3 Ma). In addition, there are other lineages whose current species are completely restricted to the NA region, such as the one formed by H. elliptica L.B. Sm., H. hernandez-sandovalii I. Ramírez, C.F. Jiménez & J. Treviño, H. scariosa L.B. Sm., and H. zacatecae L.B. Sm., whose diversification occurred in the mid-Pleistocene (1 Ma) and for which our results suggest that they reached their current areas of distribution through dispersal, i.e., they are lineages that diversified in the NA region. The times of differentiation and diversification of these lineages coincide with the last period of high geological activity of the TVB, characterized by the rise of hundreds of volcanoes during the late Pliocene and Pleistocene (3-1 Ma) (Mastretta-Yanes et al. 2015).
Neotropical region and Mexican Transition Zone. The central and southern regions of Megamexico III, corresponding to the Neotropical (NT) and Mexican Transition Zone (MTZ), are areas with a high diversity of Hechtioideae. These regions display a great variety of climates and environments, giving rise to different types of vegetation, some of these being the habitats where the Hechtioideae thrive, such as deciduous tropical forests and xeric shrublands (Espejo-Serna et al. 2005, Rzedowski 2006, Espejo-Serna et al. 2010, López-Ferrari & Espejo-Serna 2014a). Hechtioideae show a high species richness in NT and MTZ, with 59 and 56 species, respectively, and high endemism rates, with WE values of 44.5 and 39.5, respectively (Table 1). Similarly, NT and MTZ show high phylogenetic diversity values of PD = 159.12 and 166.35, respectively (Table 1). Four of the five Bakerantha species are found in MTZ, as well as all Mesoamerantha species and closely related species from all the large Hechtia clades (Figure 5B). This representation of lineages is reflected in higher-than-expected phylogenetic diversity values (SES.PD = 0.33) and overdispersion patterns in the inner parts of the phylogenetic tree (NRI = -1.87, P = 0.98) and clustering in the outer parts (NTI = 0.63, P = 0.24) (Table 1). Besides, although the NT region has a high number of species, its phylogenetic diversity is lower than expected (SES.PD = -1.45). The three genera are also present in this region, although represented by a single species of the genus Mesoamerantha, three species of the genus Bakerantha and multiple phylogenetically grouped Hechtia species; to note, contrasting with the representation of lineages in MTZ, not all the ancestral Hechtia lineages are represented in the NT region (Figure 5B). The pattern in the phylogenetic structure of MTZ, also known as “overdispersion of clusters”, facilitates the identification of processes that influence the phylogenetic assemblage over different time scales (Mazel et al. 2016). The pattern observed in the phylogenetic structure of the Hechtioideae in the MTZ is likely due to the fact that this zone is intermediate between NA and NT, characterized by a highly heterogeneous landscape that has served as a route of exchange between Neotropical and Nearctic lineages (Halffter 1987, Morrone & Márquez 2001, Morrone 2019), such as terrestrial invertebrates (Morrone & Márquez 2001) and mammals (Ortega & Arita 1998, Escalante et al. 2004). Ancestral Hechtioideae lineages have been established in this region since very early periods and have undergone diversification within the MTZ through in-situ speciation events and dispersal events, probably influenced by historical and climatic events. Altogether, they have boosted diversification and colonization in Neotropical and Nearctic areas.
Biogeographic provinces in Megamexico III. The analysis of the diversity and phylogenetic structure of Hechtioideae according to biogeographic provinces revealed biogeographic patterns on a finer scale. In general, no significant phylogenetic overdispersion patterns were found with the NTI index; instead, most provinces show phylogenetic clustering patterns. As this index is more susceptible to identifying phylogenetic structure patterns in the terminal parts of the tree (Webb et al. 2002, Mazel et al. 2016), we associate this result, together with the high dispersal rates during the Pliocene and Pleistocene, with the high rates of endemism previously reported for the Hechtioideae (Ramírez-Morillo et al. 2018a, Espejo-Serna et al. 2020). From this, we suggest that this pattern may be influenced by two processes: in-situ speciation events within each province and lineages dispersal. In addition, considering the times of diversification of this group, we related these processes to the geological, historical, and climatic events that took place during the Late Miocene and Pleistocene, which modified the environmental conditions and promoted the diversification of multiple lineages.
This study reconstructed the evolutionary history of the Hechtioideae, which was used to analyze the biogeographic patterns by incorporating geographic and phylogenetic information. The results of this study revealed important biogeographic patterns. The highest diversification rate of the Hechtioideae occurred from the mid-Miocene to the Pleistocene. The ancestral area of this lineage corresponds to the NT and MTZ regions, and dispersal events were extremely frequent during the diversification of the group. Certain climatic and geological events, such as the emergence of volcanoes and mountain ranges in central and southern Mexico, have influenced the evolutionary history of the Hechtioideae. The provinces of central Megamexico III harbor the highest diversity of the group, with the Balsas Basin province having the highest number of species and phylogenetic diversity, and the Veracruz province including representatives of the three genera of Hechtioideae. Conservation studies should focus on these areas, to preserve both the evolutionary potential and maintain the geographic range of the group.
Supplementary material
Supplemental data for this article can be accessed here: https://doi.org/10.17129/botsci.2975











 nueva página del texto (beta)
nueva página del texto (beta)



