Introduction
Colon cancer (CC) is a severe public health problem that occupies the 3rd place in incidence, and is the second type of cancer with the highest mortality worldwide (Bray et al., 2018). Approximately only 10 % of the reported cases are the familial types, whereas 90 % are of the sporadic type, related to the lifestyle, i.e., a diet low in fiber, high in saturated fats and simple sugars, as well as alcohol abuse, sedentary, and intestinal dysbiosis, among others (Sakita et al., 2017).
Carcinogenesis is a multistage process that starts when a normal cell undergoes mutation, without being able to be healed naturally, accumulating several injuries in diverse biological levels, including genetic and biochemical changes among cells that divide until a carcinoma appears (Rowles & Erdman, 2020).
The CC diagnosis is usually delayed, and its treatment is costly for the patient and the medical services of each country (Orangio, 2018). There are several treatments for CC, such as: surgical, radiotherapy, chemotherapy, directed therapy, immunotherapy, and alternative therapies (Mishra et al., 2013), which have shown efficacy depending on the cancer staging and the general conditions of patients. Chemotherapy, radiotherapy, targeted therapy, and immunotherapy are usually treatments in which agents, like 5-fluorouracil, oxaliplatin, irinotecan, capecitabine, bevacizumab, cetuximab, PD-1 inhibitors, among others, are used. These agents can be aggressive because they induce side effects like diarrhea, nausea, hair loss, tiredness, neutropenia, cardiotoxicity, nephrotoxicity, hepatotoxicity, among others (Dienstmann et al., 2015; Hammond et al., 2016; Wu, 2018).
Moreover, the objective of any cancer therapy is its complete eradication, without damaging the rest of the organism. Among the factors to consider in choosing a therapy are the patient’s condition, the location, the stage, and the type of cancer. The notion that most cancers are preceded by premalignant lesions and that their extirpation or suppression is useful to cease the process has hindered the development of the preventing approach and that nutrition is a fundamental part of it (Ricciardiello et al., 2016). Therefore, chemoprevention of cancer is an alternative consisting of the use of non-toxic natural or chemical compounds to prevent, delay, or inhibit carcinogenesis in healthy people or in the early stages (Chhabra et al., 2018). In the diet, diverse compounds can be found in fruits, vegetables, cereals, or legumes, like phenolic and nitrogenated compounds, organosulfur compounds, carotenoids, phytosterols, essential oils, polyunsaturated fatty acids, and fibers, with chemopreventive characteristics, especially for the colon, as they involve diverse action mechanisms, including antioxidant, anti-inflammatory, antimutagenic, inhibitors of uncontrolled cell proliferation, migrations, angiogenesis, apoptosis, and interaction with the intestinal microbiota, among others (Costea et al., 2018). A desirable characteristic for chemopreventive compounds is that they are accessible (available and of low cost). Hence, evaluation of foods is an opportunity to identify and promote the consumption of those with the above-mentioned qualities. To study the CC experimentally in animals, two compounds have been mainly used: azoxymethane (AOM) and the promotor dextran sodium sulfate (DSS) (Chassaing et al., 2014; Venning et al., 2013). DSS has the advantage that it can be administered in drinking water to rodents in one or more cycles to promote a sustained inflammatory stage, exerting a toxic effect on the colon epithelium (Thaker et al., 2012), whereas AOM is a genotoxic agent that starts carcinogenesis by alkylating the DNA bases, fostering mismatches in them (Neufert et al., 2007). Among the disadvantages of this model is that they require a long period for the development of tumors, which, in general, takes between 20 and 30 weeks (Neufert et al., 2007; Tanaka et al., 2003). Therefore, in this work, the number of AOM administrations (1-2) and the used doses (10-15 mg/kg), as well as the DSS concentration (1-2.5 %), the cycles (1-4), and the duration of the cycles (4-7 days) have been modified, in addition to changing the strain of the experimental animals (Cuéllar-Núñez et al., 2018; Elimrani et al., 2017; Ju et al., 2016; Sánchez-Chino et al., 2017). Suzuki et al. (2006) reported that the BALB/c strains depicted a 100 % incidence of adenocarcinomas at the end of 18 experimental weeks.
The objective of this research was to obtain a protocol that, in the least time, will allow the development of tumors for the assessment of chemopreventive agents.
Methods
Animals
The animals used for the experimental protocol were BALB/c male mice (from the animal facilities of the FES Iztacala of the Universidad Nacional Autónoma de México, Tlalnepantla, Estado de México, México) with the following characteristics: 6-8 weeks of age and weight in the range of 20-25 g. The conditioning period for all mice was seven days with 12 h light/darkness cycles at 23 °C, free of pathogens. During this period, they were supplied with standard laboratory animal feed (Rodent Laboratory Chow 5001; LabDiet, USA) and purified water, both ad libitum. All animal studies were conducted with the ethical approval of the ethics committee of Escuela Nacional de Ciencias Biológicas/Instituto Politécnico Nacional (ENCB/IPN) approved the experimental protocol carried out in this research (Approval No. CEI-ENCB-011-2017) on June 14, 2017, and in accordance with the internationally accepted principles for laboratory animal use and care (NOM-062-ZOO-1999, 1999).
Induction of colon cancer
Colon cancer was induced with AOM (A5486, Sigma-Aldrich, USA) and DSS (36,000-50,000 M.W., CAS 9011-18-1, MP Biomedicals, CA) as promoter. After a 7-days adaptation period, animals were divided in four groups of seven mice each; three protocols were designed (Figure 1) and one group corresponded to the control animals, based on reports by Tanaka et al. (2003), but making changes in the doses, concentrations, and times of administration of carcinogens according to the following scheme: P0 (control group), one intraperitoneal injection of saline solution and water ad libitum; P1: two intraperitoneal injections of 10 mg/kg of AOM, one each five days, and two cycles of 1.5 % DSS in the water ad libitum for five days, and three resting days between each cycle; P2: one intraperitoneal injection of AOM (10 mg/kg), then five days passed and were administered two cycles of 2 % DSS in the water ad libitum during seven days for each cycle, and 5-day resting period between each cycle. Finally, P3: one intraperitoneal injection of AOM (10 mg/kg), then five days passed and continued with two DSS cycles at 2 % in the water provided ad libitum for four days for each cycle and four resting days between each cycle. Once carcinogenesis had been induced, animals were maintained in 12-h dark/light cycle at 23 °C with food and water for 14 weeks; at the end of the experimental period, animals were euthanized by cervical dislocation.
General observations
The weight and disease activity index (DAI) of each animal were monitored weekly. The DAI was calculated as follows: 0 = normal, 1 = soft stools without visible blood (diarrhea), 2 = visible blood in stools, 3 = soft stools with blood (Shi et al., 2015). Besides, the survival of animals in each protocol was assessed along the 14 experimental weeks.
Macroscopic appearance and incidence of colon tumors
After euthanizing animals, the colon was excised, washed with Phosphate-buffered saline (PBS) pH 7.4, at 4 °C, and fixed with 4 % formaldehyde in PBS, pH 7.4, for 24 h in a Petri dish with solid paraffin in the bottom. The whole colon was weighed and measured for further analysis. The presence of tumors was observed macroscopically according to the definition of the National Cancer Institute of US (NCI, 2021, T): “abnormal mass of tissue that appears when cells multiply more than needed”. Incidence of tumors in whole colon was calculated for each group (Cuellar-Nuñez et al., 2018).
Quantification of oxidized proteins in colon homogenates
To quantify oxidized proteins, 0.35-0.4 g of whole colon were weighed and homogenized in an ULTRA-TURRAX® in PBS (1:10), pH 7.4, at 4º C for 30 s. For this test, 200 µL of the homogenate was mixed with 500 µL of 2,4-Dinitrophenylhydrazine (DNFH, D199303, Sigma, USA), incubated for 1 h at room temperature in darkness and supplemented with 500 µL of Trichloroacetic acid (TCA, T6399, Sigma Aldrich, USA); to precipitate the hydrazones, this mixture was homogenized and centrifuged at 13,600 g for 10 min. The pellet was recovered and washed three times with 1 mL of an ethyl-ethanol (1:1) solution, the pellet was resuspended with 1 mL of guanidine hydrochloride (G4505, Sigma Aldrich, USA), incubated at 37 °C/15 min and centrifuged at 13,600 g for 10 min. The supernatant was read at 361 nm in a UV-VIS spectrophotometer (Thermo Spectronic, Genesys 20, CA, USA). The concentration of oxidized carbonyls (OC) was calculated with a molar extinction coefficient of 21,000 M and expressed as nanograms per microgram of protein (ng/µg protein) (Sánchez-Chino et al., 2017).
Quantification of oxidized lipids in colon homogenates
For this, 500 µL of the homogenized whole colon and 2 mL of TCA-TBA-HCl (15 % w/w - 0.375 w/v - 0.25N) were used. The mixture was boiled for 15 min, cooled in an ice bath for 10 min, centrifuged at 4,000 rpm for 10 min; the supernatant was recovered and read at 532 nm in a UV-VIS spectrophotometer (Thermo Spectronic, Genesys 20). The result was expressed in nanograms (ng) of malondialdehyde (MDA) per microgram (µg) of total protein with a molar extinction coefficient of 156,000 M (Sánchez-Chino et al., 2017).
Statistical analyses
All results were processed through descriptive statistics, using mean central tendency measures ± standard error (SE). With the Minitab 17.0 statistical software, a one-way variance analysis (ANOVA) and a Tukey-Kramer comparison test were performed to identify significant differences among groups.
Results
General observations
The toxicity signs induced by the carcinogens are the weight loss, DAI, and survival, which were monitored during the 14 experimental weeks. Figure 2A shows that the three protocols, studied from induction and up to week 3, presented a lower weight gain with respect to P0. During DSS application, P2 presented a 2 g reduction compared to the initial weight. Starting with week 3, P1 maintained a constant weight until the end of the experiment, and this weight was kept below P0 along the whole experiment. Regarding P2, along the experiment, the weight depicted an irregular behavior perhaps due to the low survival percentage (43 %), however, at the end of the experiment, it showed the same weight as P1, in both cases, the weights were 10 % lower and statistically different (p < 0.05) from P0. Whereas P3, which during the first six weeks maintained its weight below the other used protocols, its weight gain was sustained and, starting at week 8, the weight was similar to that of P0, without significant difference (p < 0.05). Thus, the applied carcinogens will influence weight loss during induction and at the end of the experiment due to disease development.
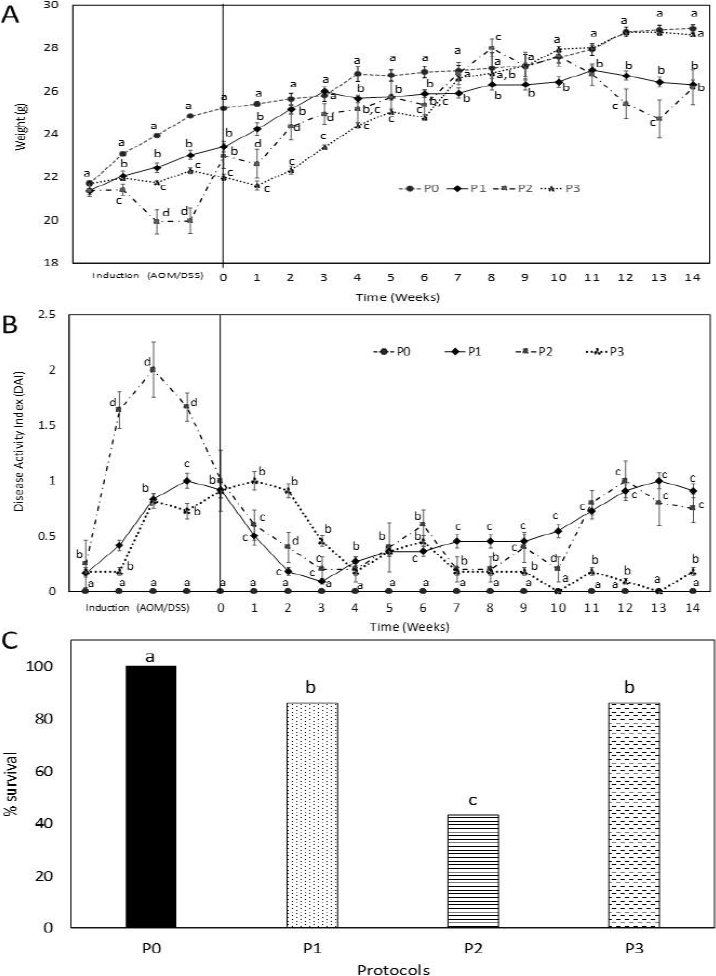
Note: A) Weight, B) DAI, C) Percentage of survival during the 14 experimental weeks (n = 7). P1) Two AOM injections and two DSS cycles at 1.5 % for five days, with three resting days; P2) One AOM injection and two DSS cycles at 2 % for seven days with five resting days, and P3) One AOM injection and two DSS cycles at 2 % for four days, with three resting days. Different letters in the groups represent significant differences among them. Unifactorial ANOVA (p < 0.05), Tukey-Kramer´s test.
Figure 2 General observations on the colon carcinogenesis induction protocols.
The DAI indicates the toxic clinical effects caused by the administration of DSS. The stools were observed macroscopically revealing that P0 did not present any toxicity sign. During the induction period, P2 revealed a higher DAI than the other protocols, once the induction finished, a recovery period started. This parameter fluctuates irregularly along the experiment and, in week 10, an increase in the toxicity signs was observed anew. P1, during induction, showed an increase in DAI, during this period maximal damage was reached. Once the induction finished, a recovery period was observed that was lost at week 3, in which the damage increased, and from this period to the end of the experiment it increases constantly, reaching a similar damage to that shown during induction. P3, from induction until week 2, presented the highest damage, after this week a recovery started that was maintained until the end of the experiment; however, at week 6, there is a peak that was related with the death of one of the experimental animals (Figure 2B). All P0 animals (100 %) survived the whole experiment, whereas P1 and P3 had an 86 % survival each one (Figure 2C). The protocol with the highest mortality was P2 with 57 %, due to the toxicity presented by the administered AOM/DSS doses, which discards this model as induction system.
Weight/length (mg/cm) ratio and incidence of colon tumors
At the end of the 14 experimental weeks, animals were euthanized, the tissue was extracted and processed as described in the Methods section. The weight/length ratio was assessed as one of the colon inflammations signs of the experimental animals. In the P0, the observed relation was lower than that observed in the other protocols: P1 was significantly different (p < 0.05) from P0, but presented less damage than P2 and P3, the three protocols were significantly different among them (p < 0.05); in P2, the damage was larger than in the other protocols. In P3, a 47 % increase in the weight/length ratio was observed with respect to P0 (Table 1).
Table 1 Effect of the colon cancer induction protocols on the weight, length of colon, and on the incidence of tumors in male BALB/c mice.
| Group | Weight/length (mg/cm) ratio | Incidence of tumors (%) |
|---|---|---|
| P0 | 43.92 ± 1.31a | 0 |
| P1 | 58.11 ± 1.00b | 83 |
| *P2 | 101.68 ± 2.99d | 100 |
| P3 | 62.92 ± 1.26c | 33 |
Note: Results present the mean ± SE (p < 0.05) (n = 5 and *P2 n = 3). Different letters in the columns indicate significant differences among groups. Unifactorial ANOVA (p < 0.05), Tukey test. Incidence represents number of mice with tumors per group.
In the control group (P0), none of the animals presented tumors; of the tested protocols, the one with the lowest response was P3, because only 33 % of the animals presented tumors. In P1, the incidence was of 83 %, whereas 100 % of the survived animals of P2 presented tumors. The three protocols presented a significant difference (p > 0.05).
Lipids and proteins oxidation
Assessment of oxidation markers is shown in Figure 3. P0 shows the basal concentration of MDA and OC, which were 0.298 and 0.418 ng/µg protein, respectively. The highest oxidation was observed in P2, for both lipids and proteins, being 1.9- and 2.2-times higher for each one with respect to P0. P1 presented 0.507 and 0.650 ng/µg protein, for MDA and OC, respectively. P3 was the one with the least damage, with only 1.5- and 1.4-times the MDA and OC concentrations as compared to P0, revealing a significant difference among the assessed protocols (p > 0.05). Proteins were the most oxidized molecules.
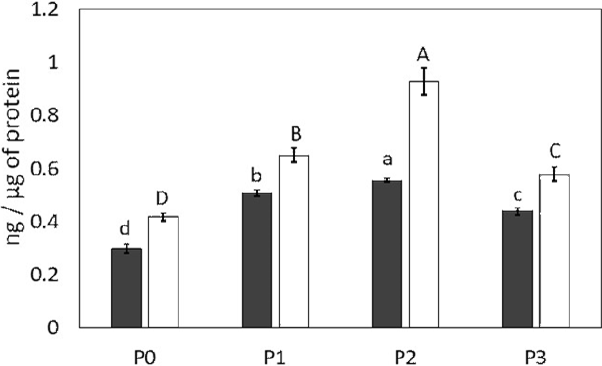
Note: Gray color: Lipids oxidation expressed in ng of MDA/ μg of protein. White color: Oxidation of proteins expressed in ng of OC/μg of protein. Upper case letters represent OC. Different letters in the groups indicate significant difference. Unifactorial ANOVA (p < 0.05), Tukey-Kramer´s test.
Figure 3 Quantification of lipid and protein oxidation products during colon carcinogenesis (n = 3).
Proteins by being abundant in biological systems are more susceptible to oxidative stress; in this way, carbonyl groups were assessed to measure the oxidation of proteins (Davies, 2016). Results of OC quantification revealed that P1 and P3 presented a higher concentration of OC, being statistically significant (p < 0.05) compared to P0 (Figure 3).
Discussion
One of the main issues when attempting to study chemopreventive agents found in foods is that the models usually take a long time to develop tumors (An et al., 2014; Shi et al., 2015; Elimrani et al., 2017; Sánchez-Chino et al., 2017; Cheng et al., 2018). Besides, it is also ideal to assess in different stages, both early and late, to determine the efficacy of the chemopreventive agent. Given that the sporadic colon cancer is caused mainly by the chronic exposure to small amounts of mutagens/carcinogens and promotors present in the environment, models have been proposed that used genotoxic and inflammatory agents. Among the most used are those in which an inducer is combined with a promoter (Tanaka et al., 2003).
In this research, a protocol was obtained that in a lesser time allowed for the development of tumors to assess natural products as chemopreventive agents. The combination of AOM administered at 10 mg/kg of weight (1-2 doses) and DSS at 1.5-2 % (4-7 days) in the drinking water allowed observing tumors in 14 weeks in the experimental animals (male BALB/c mice). Ju et al. (2016) and Lin et al. (2020) developed models that in a short time gave rise to tumors, but using genetically modified animals, which does not provide an actual knowledge of the environment or of the environmental factors that influence the sporadic cancer and, besides, they increase the research cost.
One of the observed clinical signs, during the development of the protocols, was the weight loss. Byun et al. (2015) observed this same effect and reported weight loss in groups treated with AOM/DSS one week after the administration of DSS (2 and 1 %), with a weight recovery between each cycle of DSS administration (five days), this may be due because the resting period applied was of 15 days among them. In another study, Elimrani et al. (2017) reported a 5 to 20 % weight loss in all mice treated with AOM/DSS; in the present study, the highest weight loss of experimental animals was observed during induction, showing a recovery period in weeks 3 to 10 of the experiment.
In cancer patients, there is, generally, weight loss, which has been attributed normally to the diminution of skeletal muscle as a response to the overactivation of proteolysis together with the reduction in proteins synthesis (Tan & Fearon, 2008). This phenomenon is known as cachexia due to cancer, a term that refers to a multifactorial syndrome characterized by a loss of skeletal muscle mass (with or without fatty mass), which cannot be reversed completely with the conventional nutritional support and leads to a progressive functional deterioration. Its physiopathology is characterized by a negative balance of proteins and energy, triggered by a variable combination of reduced food consumption and abnormal metabolism. The diagnostic criterion for cachexia is a weight loss over 5 %, which is also associated with a reduction in life quality and duration. In oncological patients, a mortality of 20 to 30 % has been estimated due to this syndrome (Bonetto et al., 2016; Fearon et al., 2011). Bonetto et al. (2016) reported that CD2F1 adult mice carriers of the C26 carcinoma lost body weight, which they associated with wear off the skeletal muscle and reduction in the muscular fiber size, demonstrated through morphometric assessment. Besides, this was associated with the presence of proinflammatory molecules, especially interleukin 6 (IL-6), as well as with the activation of proteolysis.
DSS is used in a model to study intestinal inflammatory diseases, because it is 100 % reproducible, as it produces chronic colitis, which is manifested by diarrhea, bleeding, prolapse, weight loss, shortening of the colon, ulceration of the mucosa, and neutrophilic infiltration (Sasaki et al., 2003). Chronic DSS ingestion is associated with epithelial dysplasia that progresses to an invasive adenocarcinoma (Sasaki et al., 2003; Elson et al., 1995). In this study, the toxicity signs were manifested from the first days of DSS administration, diminishing during the resting periods and up to week 4 after induction. Jeon et al. (2018) showed similar results, mentioning that the DAI increased daily during the first week of DSS treatment and diminished in the resting periods. Elimrani et al. (2017) reported the appearance of toxic clinical signs related to colitis starting on the 3rd day of DSS administration, which increases the severity of colitis, inducing a progressive weight loss, bleeding, and diarrhea. Shi et al. (2015) observed bleeding and soft stools one week after administering DSS, and the signs increased five days after induction with AOM/DSS. Notably, DAI is a recognized marker to measure chronic inflammation in colon since it combines various signs such as: weight loss, diarrhea, blood stool, and it has been better predictor of colon carcinogenesis, providing the degree of severity of the inflammatory process that it can be correlated with damage to intestinal mucosa that the expression of some molecular markers such as interleukin 1β (IL-1β) is an inflammatory cytokine produced predominantly by macrophages and activated monocytes or histological damage to tissues (Oliveira et al., 2014; Park et al., 2015). Moreover, Park et al. (2015) reported in their DSS induction model that mice treated with different doses and exposure times the best marker was DAI. That is why this type of marker was used as an approach to evaluate chemopreventive agents in this trial.
Mortality is another clinical sign of CC, however, it is important to count upon a representative sample, hence, it is advisable to have at least six animals per experimental group (n = 6) at the end of the experiment to detect statistical differences (Bonetto et al., 2016). Jeon et al. (2018) reported that the increase of the toxicity signs like weight loss, diarrhea, and bleeding, which reflect an increase of the DAI, led to mortality among AOM/DSS-treated animals. P2 of this study had an increased DAI during induction and an irregular behavior along the experiment, with fluctuations in toxicity signs, which induced a higher mortality among the animals of this protocol; hence, although signs developed successfully, this protocol was discarded due to its effect on mortality. Elimrani et al. (2017) reported that in the positive control with AOM/DSS less than 50 % of animals survived; therefore, analyses were performed only in three of the ten animals, which, according to Bonetto et al. (2016) would not represent a convenient statistical analysis. Groups P1 and P3 complied with an adequate number of live animals (n = 6) at the end of the experiment to perform an adequate statistical analysis with three animals for each determination, hence, they are to be considered appropriate protocols for assays with foods, according to the pursued objective.
The weight/length ratio of the colon is used as a marker of hyperplasia of the mucosa and of the severity of chronic colitis; because DSS induces a shortening of the colon, this can be used as a visual index and has been used for the study of foods with anti-inflammatory potential (Cuéllar-Núñez et al., 2018; Elimrani et al., 2017). Furthermore, a characteristic of intestinal inflammation is the thickening and shortening of the large bowel which can be measured in mice as an increase in the colon weight/length ratio, which is and indirect indicator of colonic edema and inflammation (Sydora et al., 2012; Daniluk et al., 2017).
Okayasu et al. (1990) proposed a model for the induction of chronic ulcerative colitis with DSS and found that the colon of DSS-treated animals was significantly shorter than in non-treated animals. Cuéllar-Núñez et al. (2018), in a carcinogenesis model of one AOM injection (10 mg/kg) and three cycles of DSS at 2 % in the drinking water, reported that the colon of these animals was 35 % shorter than in non-treated animals. Ju et al. (2016) observed that the weight/length ratio of the colon increases 4-times with the administration of AOM/DSS respect to a non-treated group.
Cuéllar-Núñez et al. (2018) reported a 100 % incidence of tumors in mice treated with AOM/DSS (DSS at 3 % in the drinking water). Zhang et al. (2018) observed 100 % of neoplasms in C57BL/6 mice at the end of 63 experimental days with a model consisting of one injection of AOM (10 mg/kg) and three cycles of DSS at 1 % in the drinking water with 15 resting days among cycles, whereas Shi et al. (2015) reported 100 % of tumors in ICR mice at the end of 20 experimental weeks with one injection of AOM (10 mg/kg) and one cycle of DSS at 2 % for seven days in the group with AOM/DSS. The DSS cycles differed in the three mentioned models in which 100 % of tumors were obtained, because the concentration, time of exposure, and the mouse strain differed and indicate that modifications can be made regarding the DSS cycles to obtain positive results. In the assay performed here, mice were BALB/c strain and a 100 % of tumors was obtained in the P2 group in which mortality was extremely high, but PI had 83 % tumors and more than 50 % of animals survived.
The development of carcinogenesis induces oxidative stress, which affects molecules like proteins that are found in a higher proportions and fatty acids are important targets of lipid peroxidation (Hawkins & Davies, 2019; Ramana et al., 2017).
Lipids oxidation is considered a relevant mechanism in the destruction of cell membranes, as well as for deteriorations of enzymes and receptors bound to those membranes (Betteridge, 2000). MDA is considered the main aldehyde from lipoperoxidation, hence, its determination through spectrophotometry by quantifying the adducts formed between the thiobarbituric acid (TBA) with alkenes, alkadienes; thus, MDA is considered a reliable marker of oxidative stress (Shichiri, 2014). Regarding proteins oxidation, the oxidized carbonyl groups are the product of this oxidative process in which the conformation, activity, and function of the proteins have been altered, so that they become resistant to proteolysis and affect the functionality of disease-challenged cells (Hecker & Wagner, 2018). Carbonyl groups are formed by the interaction of the amine group with the products of lipid peroxidation, like MDA and 4-hydroxy-2-nonenal (Stadtman, 2001).
The quantification of MDA and OC confirmed that the three protocols oxidized lipids and proteins significantly along the experiment. A higher proteins oxidation was obtained with the three protocols, similarly to the report by Sánchez-Chino et al. (2017). With this model, at the end of the experiment, it is also possible to assess the oxidation of these macromolecules.
Based on the above mentioned, this model could also be useful in the study of foods with antioxidant activity and to evaluate, in in vivo studies, the capacity of inhibiting the oxidation of key carcinogenesis biomolecules, as well as to assess the impact that foods can have on the tumor microenvironment, which is highly oxidant (Costea et al., 2018; Prasad et al., 2017). Blocking the free radicals allows inhibiting cancer progression, because the first manifestation of the disease starts with an oxidation and inflammation caused by an oxidant environment induced by AOM and the inflammatory state induced by the DSS.
Conclusion
The P1 and P3 induction protocols presented less mortality, weight loss, and acceptable DAI, a weight/length ratio higher than the negative control and presence of tumors, as well as oxidation of molecules like lipids and proteins as markers of oxidative stress present in the carcinogenic process. Hence, these protocols are considered adequate to be used for chemoprevention assays in male BALB/c mice.
It is a challenge to identify models that can attain tumors in a reasonable time and which are reproducible, therefore, the implementation of a CC induction protocol that will allow for the assessment of tumors in a relatively short time span, is a useful tool for the analysis of foods with a protecting effect, where prevention or reversion of the CC is possible. In addition, it is important to analyze the food to be able to observe the action mechanisms and the synergism presented by the compounds that grant their chemoprotective capacity.











 nueva página del texto (beta)
nueva página del texto (beta)




