Introduction
Knee osteoarthritis (KOA) is a common chronic degenerative joint disease that mainly affects middle-aged and elderly populations, particularly those over 50 years old1. Statistics show that KOA has become one of the leading causes of disability and health impairment, affecting tens of millions of lives globally2. Its incidence continues to rise with population aging, and it is predicted that the burden of this disease will continue to grow in the coming decades3. Beyond its significant impact on individual health, KOA also places a considerable burden on socioeconomic aspects, including health-care resource utilization and diminished work productivity4.
KOA is the primary cause of joint pain and disability in the elderly, which seriously affects the quality of life of the elderly. Identifying the source and mechanism of pain in KOA is important, and understanding the cause of pain may help to better target appropriate treatment to affected patients and may also help to identify alternatives that can help reduce symptoms and improve function. Studies have shown that the peripheral and/or central nervous system plays an important role in the occurrence and development mechanism of KOA-related pain. Peripheral pain mechanisms include direct activation and/or sensitization of nociceptors by stimuli such as joint inflammation and/or structural damage5. The inflammation was mainly synovial inflammation, and the structural damage was mainly the bone marrow lesion and cartilage loss. In KOA, inflammatory lesions, namely synovitis and bone marrow lesions, have always been the main pathological damage related to pain6. Although cartilage loss is an important structural feature, it is not neurogenic and therefore cannot be a direct source of pain in mild-to-moderate disease. Loss, inactivation, or overactivation of nociceptive regulatory mechanisms in the central nervous system can lead to hyperalgesia and hypersensitivity, and their altered sensitivity may explain more persistent pain in KOA7. At present, the treatment of KOA primarily aims to alleviate patients´ pain and improve joint function. Common treatments include non-steroidal anti-inflammatory drugs (NSAIDs), physiotherapy, orthopedic braces, and intra-articular injections8,9. NSAIDs are prescribed when the patient presents with exacerbation of pain and a swollen knee. These agents act by blocking the pro-inflammatory agents such as prostaglandins and leukotrienes by reversibly blocking the cyclooxygenase and lipoxygenase pathway. Long-term use of drugs such as NSAIDs can also cause adverse gastrointestinal reactions and cardiovascular risks, imposing an additional health burden on patients1. Physiotherapy is good quality evidence that muscle strengthening and an aerobic exercise program are beneficial in the management of KOA10. Range-of-motion exercises help to prevent the development of contractures. Periarticular muscle strengthening exercises tend to stabilize the knee and improve symptoms. The aim of an orthosis is to reduce pain and improve function. The ideal candidate for an orthosis is a patient with passively correctable unicompartmental arthritis. A brace may function by improving the biomechanical axis of the deformity thereby unloading the compartment or by improving the perception of instability. Injectable hyaluronate therapy has a theoretical advantage in KOA as a result of its viscoelastic, analgesic, anti-inflammatory, and chondroprotective properties. A review revealed up to 5-13 weeks of improvement in pain and function post-injection following the use of the hyaluronate group of products11. However, although these methods can alleviate patients pain and inflammation to some extent, they cannot fundamentally prevent the progression of the disease and the degradation of the cartilage12. Radiofrequency ablation (RFA) therapy, as an emerging interventional treatment, has received widespread attention in recent years. The principle is to apply radiofrequency energy to the disease site, relieving pain by disrupting nerve endings conduction13. For KOA, RFA is considered a promising treatment option that can improve pain and restore joint function by alleviating inflammatory reactions and abnormal nerve conduction14.
While several studies have delved into the application of RFA in KOA, debates persist regarding its safety and efficacy15. Previous meta-analyses have presented partial evidence, yet they included non-SCI indexed literature of lower methodological quality and incomplete systematic retrieval, while new research findings continue to emerge16,17. Therefore, we conducted this updated systematic review and meta-analysis to more comprehensively assess the efficacy and safety of RFA in the treatment of KOA, citing the latest research evidence to provide a more reliable basis for clinical decision-making.
Materials and methods
We followed the guidelines of the Preferred Reporting Items for Systematic Reviews and Meta-Analyses recommended by the Cochrane Collaboration for this systematic review and meta-analysis18. We searched three electronic databases: PubMed, Embase, and Cochrane Central Register of Controlled Trials (CENTRAL), from their inception to June 26, 2023, and limited the language to English. Our search strategy combined MeSH/Emtree terms and free text, with keywords mainly including "knee," "osteoarthritis," "radiofrequency ablation," "randomized controlled trial," etc., set to search in the title and abstract. Two researchers independently screened electronic records and retrieved publications based on the inclusion and exclusion criteria. During the screening process, any discrepancies were resolved by mutual discussion and full-text review. In cases where a consensus could not be reached, a decision was made by a senior researcher.
In this study, we established the following inclusion criteria: (1) Patients diagnosed with KOA; (2) Patients in the intervention group received RFA treatment; (3) A control group was established, receiving sham surgery or other therapeutic methods such as drugs; (4) Relevant outcomes such as post-operative Visual Analog Scale (VAS), numerical rating scale (NRS), Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC), Oxford Knee Score (OKS), Global Perceived Effect (GPE) scale, adverse reactions, etc.; and (5) Only randomized controlled trials (RCTs) were included. Our exclusion criteria primarily included the following: (1) Duplicate data, extended studies, or the same study; (2) Types of studies irrelevant to the topic, such as animal studies, case reports, literature reviews, or conference abstracts; (3) Studies with incomplete data or unreported established outcomes, such as using a self-control group; and (4) Studies using other interventions or controls.
After excluding irrelevant studies, two researchers independently extracted the features and data of the included studies. In accordance with the suggestions of the Cochrane Reviewers´ Handbook 5.1, two researchers independently assessed the risk of bias in the included studies.
We conducted a meta-analysis using RevMan 5.3 (Nordic Cochrane Centre, Cochrane Collaboration, Copenhagen, Denmark). For continuous variables, we used standardized mean differences (SMD) and 95% confidence intervals (CI) as the statistical analysis indicators of effect size. For categorical variables, we used risk difference (RD) as the statistical analysis indicator of effect size. We used the Cochran Q test in conjunction with the I2 statistic to assess the degree of heterogeneity among the results of the included studies. When the statistical heterogeneity of the results of the included studies was low (p > 0.1 or I2 < 50%), we used a fixed-effect model for analysis; when there was statistical heterogeneity among the results of the included studies (p < 0.1 or I2 ≥ 50%), and we used a random-effects model for meta-analysis. We set the significance level of the meta-analysis at a = 0.05. We evaluated the presence of publication bias by plotting a funnel plot. To assess the impact of individual studies on the overall effect, we conducted a sensitivity analysis, observing the changes in effect size after excluding individual studies. In addition, we also conducted subgroup analyses to examine the changes in the treatment effects of RFA in different situations for patients with KOA.
Results
According to the search strategy, a total of 147 electronic records were retrieved, including 49 from PubMed, 57 from Embase, and 41 from Cochrane. After using Endnote X9 software and manually removing 44 duplicate records, 79 irrelevant papers were excluded by browsing titles and abstracts. By reading the full text, 11 papers with irrelevant outcome indicators, unrelated comparison strategies, incomplete data, or extended similar studies were removed. Finally, 13 papers were included in the meta-analysis, and the results of the literature screening process are shown in figure 1.
This study included 13 articles and 865 patients19-31. The basic characteristics of the included literature in this study are shown in table 1. There were 6 studies conducted in Asia. The majority of the studies´ design (84.6%) were single-center RCTs, 4 studies used a double-blind experiment, 3 studies adopted a single-blind setting, and 6 studies used an open-label setting. The included studies employed various types of RFA procedures, such as pulsed RFA and cooled RFA. The settings of the control groups were diverse, including placebo surgery groups, intra-articular injections of sodium hyaluronate, local anesthetic injections, steroid injections, and oral administration of NSAIDs. The stimulation sites and intervention parameters of RFA varied due to different study designs, but most studies focused on the knee joint nerves as the treatment target. The intervention parameters used were quite varied, and the observed scores were primarily the NRS and VAS for pain, as well as the WOMAC, GPE, and OKS.
Table 1 Basic Characteristics of Included Studies
| Authors | Year | Country | Design | Blinding | Intervention Group | Control Group | Treatment Target | Intervention Parameters | Observation Score | Longest Follow-up Time (Weeks) |
|---|---|---|---|---|---|---|---|---|---|---|
| Carpenedo et al.20 | 2021 | Italy | Single- center | Double-blind | PRF | Sham | IA | 42°C, 120s | NRS, OKS | 24 |
| Chen et al.21 | 2020 | America | Multi- center | Open-label | CRF | IA HA | IA | 60°C, 150s | GPE, WOMAC | 24 |
| Choi et al.22 | 2011 | Korea | Single- center | Double-blind | RFA | Sham | GN | 70°C, 90s | VAS, GPE, OKS | 12 |
| Davis et al.23 | 2019 | America | Multi- center | Open-label | CRF | IA steroids | GN | 60°C, 150s | NRS, OKS | 24 |
| EI-Hakeim et al.24 | 2018 | Egypt | Single- center | Single-blind | RFA | Oral NSAIDs | GN | 80°C, 270s | VAS, WOMAC | 24 |
| Hong et al.25 | 2020 | China | Single- center | Single-blind | RFT | IA steroids | GN | 70°C, 120s | GPE | 24 |
| Kumaran and Watson26 | 2019 | UK | Single- center | Single-blind | CRMRF | Sham | IA | 15 min | VAS | 12 |
| Qudsi-Sinclair et al.27 | 2018 | Spain | Single- center | Double-blind | RFA | IA steroids | GN | 80°C, 90s | NRS, OKS, KSS, SF-36, PGI-I | 48 |
| Rahimzadeh et al.28 | 2014 | Iran | Single- center | Double-blind | PRF | IA dextrose | IA | 42°C, 15 min | VAS | 12 |
| Sari et al.29 | 2018 | Turkey | Single- center | Open-label | RFA | IA analgesics | GN | 80°C, 90s | VAS, WOMAC | 12 |
| Shen et al.30 | 2017 | China | Single- center | Open-label | RFT | IA PRP+HA | IA | 70°C, 120s | VAS, SF-36, AKSS | 12 |
| Xiao et al.31 | 2018 | China | Single- center | Open-label | RFA | IA HA | GN | 60, 70, and 80°C, 90 s | VAS | 24 |
| Yuan et al.32 | 2016 | China | Single- center | Open-label | PRF | IA analgesics | IA | 42°C, 6 min | VAS, WOMAC | 24 |
PRF: pulsed radiofrequency ablation; NRS: numerical rating scale; OKS: Oxford Knee Scores; CRF: cooled radiofrequency ablation; IA: intra-articular; HA: hyaluronic acid; GPE: global perceived effect; WOMAC: Western Ontario and McMasters Universities Osteoarthritis; GN: genicular nerve; VAS: Visual Analog Score; RFA: radiofrequency ablation; NSAIDs: non-steroidal anti-inflammatory drugs; RFT: radiofrequency thermocoagulation; CRMRF: capacitive resistive monopolar radiofrequency; KSS: knee society score; SF-36: 36-Item Short Form Health Survey; PGI-I: Patient Global Impression Scale of Improvement; AKSS: American K.
In addition, the patient characteristics of the included studies are shown in table 2. The total proportion of males in the RFA group and the control group were 140/434 (32.3%) and 145/431 (33.6%), respectively. The average age range for the RFA group and the control group were 56.5-70.37 years and 56.87-71.08 years, respectively. The average body mass index ranges for the RFA group and the control group were 23.51-32.2 and 25.8-30.5 kg/m2, respectively. The average disease duration for the RFA group and the control group were 5.6-90 months and 4.3-60 months, respectively. The average pain scores for the RFA group and the control group were 5.9-8.25 and 5.6-8, respectively.
Table 2 Basic characteristics of the included population
| Authors | Sample size (RF/Con) | Male count (RF/Con) | Intervention group age (years) | Control group age (years) | Average BMI (RF/Con) | Average disease duration (months) (RF/Con) | Baseline pain score of intervention group | Baseline pain score of control group |
|---|---|---|---|---|---|---|---|---|
| Carpenedo et al.20 | 8/8 | 2/3 | 70.37 ± 7.36 | 70.87 ± 11.81 | 29.48/29.62 | 9.62/10.37 | 8.25 ± 0.70 | 8 ± 1.19 |
| Chen et al.21 | 89/88 | 37/34 | 63.3 ± 10.7 | 63.1 ± 9.7 | 32.2/30.5 | 90/106 | NA | NA |
| Choi et al.22 | 17/18 | 2/3 | 67.9 ± 7.1 | 66.5 ± 4.8 | 26.2/26.5 | 75.6/88.8 | 7.82 ± 1.38 | 7.72 ± 0.75 |
| Davis et al.23 | 76/75 | 26/26 | 63 ± 12 | 66 ± 13 | 30.6/30.4 | 10.7/8.6 | 7.3 ± 1.2 | 7.2 ± 1.0 |
| EI-Hakeim et al.24 | 30/30 | 9/12 | 62 ± 7.37 | 56.87 ± 6.53 | 32.02/30.21 | 7.6/5.7 | 7.07 ± 0.2 | 7.07 ± 0.2 |
| Hong et al.25 | 26/27 | 10/12 | 59.46 ± 5.81 | 60.93 ± 7.50 | 24.6/25.8 | 32.54/34.67 | 6.46 ± 1.14 | 6.37 ± 0.93 |
| Kumaran and Watson26 | 15/15 | 6/6 | 63 ± 10 | 63 ± 10 | 31/31 | 5.6/4.3 | 6.3 ± 1.2 | 5.8 ± 1.2 |
| Qudsi-Sinclair et al.27 | 14/14 | 4/3 | 67.4 ± 7.2 | 71.08 ± 9.4 | NA | 42/31 | 7.07 ± 1.06 | 6.43 ± 1.56 |
| Rahimzadeh et al.28 | 24/26 | 11/10 | 56.95 ± 8.31 | 60.57 ± 7.47 | NA | NA | 7.08 ± 1.41 | 7.11 ± 1.03 |
| Sari et al.29 | 37/36 | 7/9 | 64 ± 8 | 64 ± 10 | 23.51/22.89 | 60/60 | NA | NA |
| Shen et al.30 | 27/27 | 7/9 | 62.24 ± 10.35 | 62.35 ± 9.70 | NA | 60.12/59.52 | 7.12 ± 1.08 | 7.14 ± 1.03 |
| Xiao et al.31 | 49/47 | 12/11 | 56.5 ± 9.5 | 61.5 ± 8.5 | NA | 36.5/35.5 | 7.48 ± 1.24 | 7.53 ± 1.27 |
| Yuan et al.32 | 22/20 | 7/7 | 69.9 ± 11.1 | 67.4 ± 10.3 | NA | 41.6/38.3 | 5.9 ± 1.1 | 5.6 ± 1.4 |
RF: radiofrequency group; Con: control group; BMI: body mass index; NA: non-applicable.
Detailed information about the risk of bias is shown in figure 2. Quality assessment of the literature was conducted using the cochrane collaboration tool. All studies clearly reported methods of random sequence generation, and most studies (61.5%) described allocation concealment methods. Some trials obtained unclear or high-risk bias due to open-label or single-blind measures for participants and executors, only four studies explicitly mentioned conducting double-blind research, and many studies did not provide explicit descriptions for outcome indicator blinding. All RCTs did not have incomplete outcome data, apparent selective reporting, or other biases.
All 13 studies reported post-treatment pain scores. Among them, 9 studies reported pain scores 1-2 weeks after treatment, 7 studies reported pain scores 4 weeks after treatment, 10 studies reported pain scores 12 weeks after treatment, and 6 studies reported pain scores 24 weeks after treatment. Compared with the control group, the pain scores of the patients in the RFA group significantly reduced at 1-2 weeks, 4 weeks, 12 weeks, and 24 weeks after treatment, with SMDs of −1.24 (95% CI: −1.99-−0.49; p = 0.001; I2 = 91%), −0.76 (95% CI: −1.27-−0.26; p = 0.003; I2 = 76%), −1.70 (95% CI: −2.56-−0.83; p = 0.0001; I2 = 94%), and −2.26 (95% CI: −3.49-−1.04; p = 0.0003; I2 = 95%), respectively (Fig. 3).
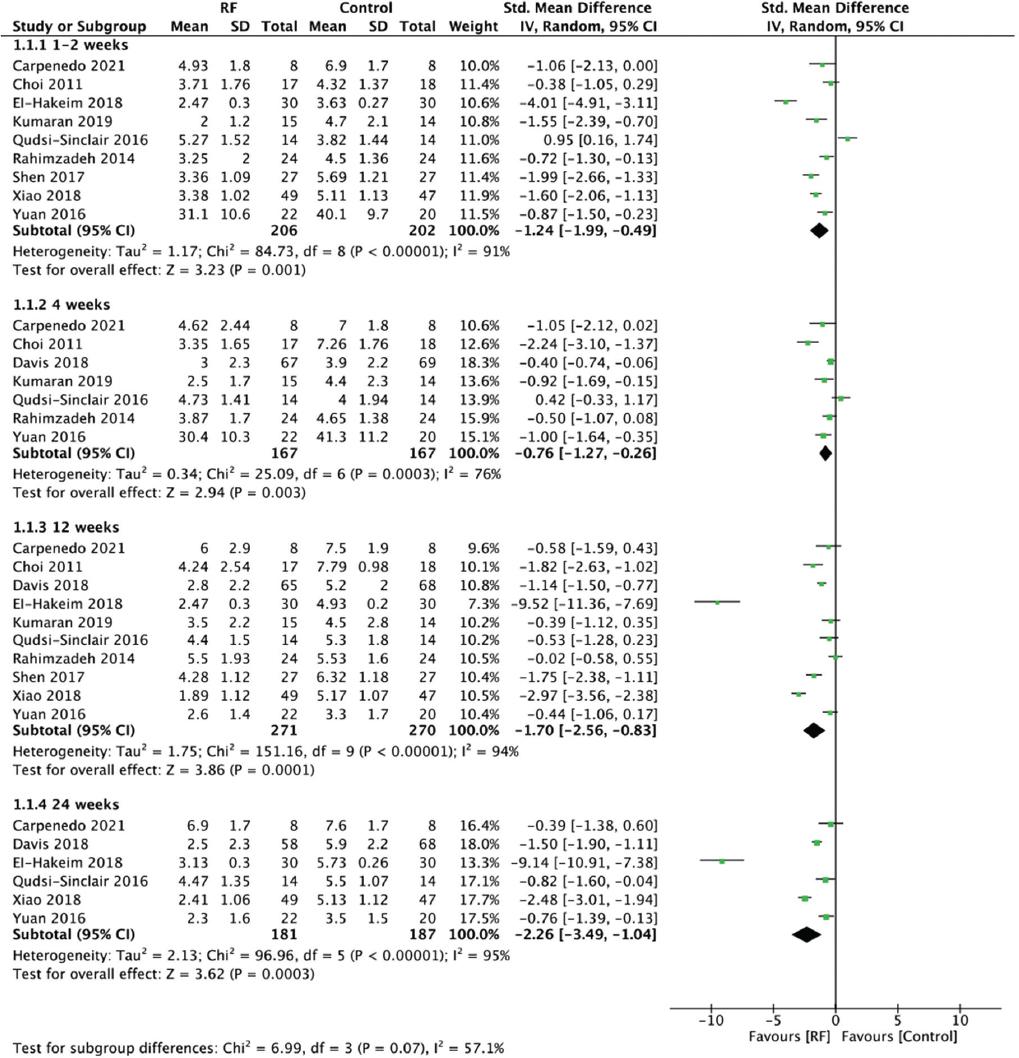
Figure 3 Forest plot comparing pain scores between the RF group and the control group. RF: radiofrequency; M-H: Mantel-Haenszel; SD: standard deviation; IV: inverse variance.
Three, four, and three studies, respectively, evaluated the changes in the WOMAC index at 4 weeks, 12 weeks, and 24 weeks after treatment. The results showed that compared with the control group, the WOMAC index of the RFA group was lower. The pooled SMDs were −0.65 (95% CI: −1.07-−0.23; p = 0.002; I2 = 60%), −1.26 (95% CI: −2.33-−0.19; p = 0.02; I2 = 94%), and −1.58 (95% CI: −2.89-−0.26; p = 0.02; I2 = 94%), respectively (Fig. 4).

Figure 4 Forest plot comparing WOMAC index between the RF group and the control group. WOMAC: Western Ontario and McMasters Universities Osteoarthritis; RF: radiofrequency; M-H: Mantel-Haenszel; SD: standard deviation; IV: inverse variance.
Three studies each reported the comparison of the GPE scores of the two groups of patients after treatment. Compared with the control group, RFA significantly improved patient satisfaction 12 weeks after treatment, but there was no significant difference 4 weeks after treatment. The pooled SMDs were 1.29 (95% CI: 0.52-2.06; p = 0.001; I2 = 82%) and 0.66 (95% CI: −0.20-1.52; p = 0.13; I2 = 88%), respectively (Fig. 5).

Figure 5 Forest Plot Comparing GPE Scores between the RF Group and the Control Group; GPE: Global Perceived Effect; RF: radiofrequency. M-H: Mantel-Haenszel; SD: standard deviation; IV: inverse variance.
Ten RCTs reported on side effects after using RFA. Compared to the control group, the risk of adverse events in patients using RFA did not change. The pooled RD was 0.01 (95% CI: −0.02-0.04; p = 0.52; I2 = 0%) (Fig. 6).

Figure 6 Forest plot comparing adverse reactions between the RF group and the control group; RF: radiofrequency; M-H: Mantel-Haenszel; SD: standard deviation; IV: inverse variance.
We also conducted a subgroup analysis to assess the impact of different factors on the pooled results and heterogeneity of pain scores at the 12-week follow-up, as shown in Table 3. The results show that whether the studies were conducted in Asia or other regions, RFA is indicated to improve patient pain scores. The effect is better when targeting the nerves of the knee joint, whereas the intra-articular approach has achieved a marginal effect (p = 0.05). In addition, both traditional RFA and other RFA methods have achieved improvements. It is worth noting that heterogeneity did not significantly change in the subgroup analysis, suggesting that it may come from other sources.
Table 3 Subgroup analysis of patient pain score at 12 weeks post-treatment
| Group | Number of studies | Pooled SMD (95% CI) | Z-value | p-value | Heterogeneity | |
|---|---|---|---|---|---|---|
| I2 (%) | p-value | |||||
| Geographic Location | ||||||
| Asia | 5 | −1.40 (−2.51-−0.29) | 2.46 | 0.01 | 94 | < 0.001 |
| Other | 5 | −2.16 (−3.75-−0.57) | 2.66 | 0.008 | 95 | 0.008 |
| RFA Target | ||||||
| GN | 5 | −2.93 (−4.50-−1.36) | 3.66 | < 0.001 | 96 | < 0.001 |
| IA | 5 | −0.63 (−1.27-0.00) | 1.95 | 0.05 | 77 | 0.002 |
| Type of RFA | ||||||
| RFA | 4 | −3.53 (−5.79-−1.26) | 3.05 | 0.002 | 97 | < 0.001 |
| Other | 6 | −0.74 (−1.26-−0.22) | 2.78 | 0.005 | 77 | < 0.001 |
SMD: standard mean difference; CI: confidence interval; GN: genicular nerve; IA: intra-articular; RFA: radiofrequency ablation.
We performed a funnel plot analysis on post-operative pain scores. The funnel plots show that the results are approximately symmetrically distributed at any follow-up period, indicating no apparent publication bias (Fig. S1). Moreover, we performed a sensitivity analysis on the post-operative pain scores. The results did not significantly change after excluding each study, suggesting that individual studies have a limited impact on the overall results, but the heterogeneity among studies remains high.
Discussion
This meta-analysis systematically evaluates the efficacy and safety of RFA as a treatment for patients with KOA, and a methodological quality assessment was carried out on the included studies. The primary findings of this study are as follows: (1) Compared to the control group, patients undergoing RFA showed significant decreases in pain scores at 1-2 weeks, 4 weeks, 12 weeks, and 24 weeks post-treatment, although no significant differences were observed in the VAS scores at 48 h post-operation between the two groups; (2) RFA helps to reduce the WOMAC scores of patients at 4 weeks, 12 weeks, and 24 weeks; (3) RFA significantly improves patient satisfaction at 12 weeks post-treatment, but no significant difference was noted at 4-week post-treatment; and (4) Compared to the control group, RFA does not increase the risk of adverse events in patients. This study hopes to provide evidence-based medical justification for the clinical use of RFA as a pain relief method in treating patients with KOA and offer a reference for improving patient satisfaction and preventing adverse events.
Osteoarthritis is a chronic degenerative joint disease, the progression of which involves several pathological changes32,33. First, the damage and degeneration of articular cartilage are the core features of osteoarthritis. The degeneration of cartilage leads to irregularities on the joint surface, resulting in joint friction and wear. Second, the inflammatory response around the joint and changes in synovial fluid are also important characteristics of osteoarthritis. The inflammatory response leads to synovial membrane thickening and an increase in joint fluid production, further exacerbating the pathological changes of the disease. Finally, osteophyte formation is a late-stage manifestation of osteoarthritis. It may represent the body´s self-repair mechanism in response to joint damage, but it may also cause joint deformity and functional impairment. Pain is one of the most common and primary symptoms among osteoarthritis patients. The occurrence of pain is related to several factors34. First, the destruction and degeneration of articular cartilage cause irregularities on the joint surface, increasing joint friction and pressure, and leading to inflammation and pain. Second, the inflammatory response around the joint and changes in the synovial fluid lead to congestion of the synovial membrane and increased sensitivity of nerve endings, further triggering pain. Furthermore, a decrease in joint stability and a decline in muscle strength can also increase joint load and the perception of pain. At present, the therapeutic management of osteoarthritic pain mainly includes two aspects: Pharmacological and non-pharmacological treatments. Commonly used pharmacological treatments include NSAIDs and corticosteroids. NSAIDs have anti-inflammatory and analgesic effects and can effectively alleviate the pain and inflammatory response of osteoarthritis. In addition, topical NSAIDs also offer a choice for local pain relief6. The OARSI guidelines recommend that NSAIDs should be given in conservative doses and durations, as there is concern regarding an increasing risk of gastrointestinal disturbance and multi-organ failure35. So, caution and attention must be focused on avoiding excessive use of these medications. In addition, consideration of all known safety information and individual patient comorbidities is imperative when the health-care practitioner is selecting any of these medications for a patient. Non-pharmacological treatments include physical therapies (such as hot compress, cold compress, and rehabilitative exercise) and rehabilitation therapies36. These therapeutic methods aim to improve joint function, alleviate pain, and enhance the patients quality of life.
RFA is an interventional treatment method that uses the effects of radiofrequency current to destroy disease-related tissue or nerve conduction pathways to achieve pain relief. This technique is based on the high-frequency oscillation and thermal effects of radiofrequency current, which can precisely target specific areas for tissue ablation37. The principle of RFA is based on the resistive heating effect of tissues. Under the influence of radiofrequency current, friction between positive and negative charges within tissues generates heat. This high-temperature effect can destroy nerve conduction pathways in the diseased tissue, thus blocking the transmission of pain signals38. RFA has adjustable power and time settings, allowing for personalized treatment according to specific conditions. The application of RFA in disease treatment has a multi-year developmental trajectory. Initially, RFA was primarily used in the field of cardiology, for treating diseases such as arrhythmias39. With continuous technological advancement and accumulated clinical practice, RFA has gradually found applications in other areas, such as tumor treatment, pain management, and more40. RFA has become one of the major means in the field of interventional treatment. In disease therapy, significant advancements have been made in the pain relief applications of RFA. This technique is extensively utilized to treat chronic pain conditions, such as back pain, neck pain, and arthritis41. Compared to traditional pharmacological treatments, RFA provides durable analgesic effects and can reduce drug usage, thus lowering the occurrence of adverse reactions42. Therefore, RFA is widely recognized as a safe and effective pain management method. The application of RFA in the treatment of osteoarthritis has also received much attention. As a minimally invasive interventional treatment modality with quick recovery, RFA demonstrates the potential in relieving osteoarthritic pain. It can improve patients´ symptoms and quality of life by precisely destroying pain sources, thereby alleviating arthritic inflammation and transmission of pain signals43.
The results of this study are consistent with previous meta-analyses and relevant research, supporting the efficacy and safety of RFA in the treatment of pain in patients with KOA17,18. This meta-analysis has several advantages, highlighting the importance of updated clinical evidence, the inclusion of more studies, and the exclusion of low-quality research. First, a crucial advantage of this meta-analysis lies in its updated clinical evidence. The latest research outcomes were included in this meta-analysis to provide more accurate and reliable conclusions. By including the latest studies, we can better understand the safety and efficacy of RFA therapy in treating KOA. Second, this meta-analysis incorporated more studies. By extensively searching multiple databases and academic journals, we endeavored to access as many relevant studies as possible and included them in the analysis. The advantage of doing this is the increase in the sample size, thereby enhancing the statistical power of the analysis, which allows for a more accurate assessment of the effects of RFA therapy. Including more studies can also enhance the consistency and stability of the results, making the conclusions more universally applicable and can be generated for other studies. Compared to previous meta-analyses, we also searched for studies that had been overlooked before and incorporated them into this analysis. Third, this meta-analysis excluded low-quality research. Through a rigorous screening and evaluation process, we excluded lower-quality non-SCI included studies previously incorporated by Liu et al.18. By doing so, we intend to ensure the reliability and accuracy of the analysis, avoiding the introduction of bias from low-quality research that could adversely affect the results. By excluding low-quality research, we can draw more reliable and trustworthy conclusions, providing more meaningful guidance for clinical practice. Due to the low incidence rate, and for a more systematic evaluation of the effects of RFA, this study combined all the reported data on the incidence of adverse reactions from all the studies and used RD for analysis, instead of classifying adverse reactions for quantitative analysis. The results found that the use of RFA did not increase the risk of adverse reactions, which is also consistent with previous research. Subgroup analysis found that the geographical area of the study, the target location, and the type of RFA did not significantly affect the consolidated results after 12 weeks, to some extent supporting the therapeutic effect of RFA for pain relief in KOA. However, it is worth noting that the source of heterogeneity is not yet determined; this might come from the design of the control group therapy, different blind method settings, etc., suggesting the need for more high-quality evidence in the future, and the strengthening of the classification and screening of the included research data.
RFA has recently gained popularity as an intervention for chronic knee pain in patients. Long-term efficacy and adverse events are still largely unknown. Although vascular injuries after genicular nerve RFA have not been reported, genicular vascular complications are well documented in the surgical literature. The systematic review of RFA showed that among the 27 patients analyzed, the superior lateral genicular artery was involved in 25.9% (7/27), the superior medial genicular artery was involved in 40.7% (11/27), and the inferior medial genicular artery was involved in 33.3% (9/27)44. Most often, these vascular injuries result in the formation of a pseudoaneurysm, arteriovenous fistula (AVF), hemarthrosis, and/or osteonecrosis of the patella. Based on the detailed dissections and review of the literature, our investigation suggests that vascular injury is a possible risk of genicular RFA. Therefore, the interventionist must exercise great care while performing RFA of genicular nerves to avoid inadvertently injuring nearby structures, especially vascular structures, leading to iatrogenic complications. We should also consider the sink effect of blood vessels in proximity to the RFA targets. Due to constant blood flow, the temperature of the targeted area is attenuated45. Perhaps, this reduction in temperature may lead to a better coagulation effect than if it were by direct needle trauma, and thus, vascular injury can be avoided. The longest follow-up period of the 13 included studies was only 48 weeks, and none of them involved adverse events of osteonecrosis in RFA, so our study did not address the long-term theoretical risks associated with RFA in the knee, including the possibility of vascular injury leading to osteonecrosis. However, these potential complications have not been observed in long-term RFA studies46,47, and our subjects did not develop any early symptoms of these complications. We conclude that RFA is unlikely to result in these types of complications when performed by a fully trained and experienced physician. In the future, we will pay attention to studies with long-term follow-up results to analyze whether there are adverse reactions such as osteonecrosis in the treatment of KOA with RFA.
There are some limitations to this study that requires discussion. First, even though this analysis only included RCTs, significant heterogeneity could lead to biased results. Therefore, more high-quality RCTs are required in the future to further investigate this issue. Second, the current studies mainly focus on the short-term impact of RFA on patients, with a lack of research into long-term follow-up results, and the indicators of attention to adverse reactions from RFA are not sufficiently detailed. Furthermore, the results of this study rely solely on data reported in published studies. For some critical details or specific subgroup analyses, there may be situations where data are incomplete or unobtainable. This may impact the reliability and accuracy of certain conclusions. Lastly, despite excluding low-quality studies, some of the included studies still demonstrate poor research quality. This might have some effect on the final results. In addition, due to potential variances in methodologies and standards across different studies, heterogeneity might present certain challenges.
Conclusions
In summary, RFA, as a surgical approach, when compared to conventional treatment or sham surgery, helps enhance analgesic effects, improves joint symptoms, and increases patient satisfaction, without increasing the incidence rate of side effects. It has the potential to become a new therapeutic strategy for pain management in patients with KOA. However, due to the rather significant heterogeneity and the lack of studies on long-term follow-up results in this analysis, more high-quality research is needed in the future to delve deeper into these aspects of the results.











 nueva página del texto (beta)
nueva página del texto (beta)




