Vanilla (Vanilla planifolia) is an orchid with important commercial value due to the compound vanillin, that it produces. Vanillin is sought after by various industries for its aroma and flavor. This crop faces a significant threat from the disease known as “root and stem rot”, which is caused by the ascomycete Fusarium oxysporum f. sp. vanillae (Pinaria et al., 2010; Adame-García et al., 2015). However, it has been shown that vanilla is associated with multiple genotypes of F. oxysporum that do not necessarily behave as pathogens (Casillas-Isiordia et al., 2017). This also happens with other plant species, which has led to subdividing them into pathogenic (formae speciales) and beneficial endophytes to avoid losing valuable genetic information that could help understand the nature of this pathogen (Lecomte et al., 2016).
Since there are no resistant vanilla genotypes available to Mexican producers, they seek alternatives to control the disease, the main one being chemical control based on the use of fungicides. The main vanilla-producing region is Papantla, in the state of Veracruz (Hernández-Hernández, 2011), an area considered to be the center of origin, domestication and distribution of vanilla (Lubinsky et al., 2008). The chemical control of Fusarium wilt in vanilla is performed mainly with fungicides of the benzimidazole group (Hernández-Hernández, 2019). These fungicides directly affect the degradation of β-tubulin in microtubules, thus inhibiting fungal cell division (Zhang et al., 2015). Other fungicides suggested for the control of F. oxysporum f. sp. vanillae are broad-spectrum ones, such as chlorothalonil, of the carbamate group, which have multiple sites of action in fungal cells, including the enzyme glycerol-3-phosphate dehydrogenase (GPDH), involved in glycolysis, which, when inactivated, causes the death of the fungus (Long and Siegel, 1975; Baćmaga et al., 2018).
In the case of benzimidazoles, numerous cases of resistance have been reported in various formae speciales of F. oxysporum (Arie, 2019), which have previously been associated with a point mutation at position 200 of the encoding sequence of β-tubulin (Petkar et al., 2017). For this reason, it is crucially important to determine the real susceptibility of F. oxysporum isolates to these fungicides, before using to control these pathogens. The present study aimed to evaluate the effect of three fungicides of the benzimidazole group and chlorothalonil on pathogenic and non-pathogenic strains of F. oxysporum associated with Vanilla planifolia. An additional aim was to determine whether resistance in strains of this forma specialis is related to point mutations in the sequences of the target proteins, β-tubulin and glycerol-3-phosphate dehydrogenase.
Materials and methods
Strains of F. oxysporum. Twelve strains of F. oxysporum isolated from vanilla were used in the present study: nine strains that are pathogenic for V. planifolia and three non-pathogenic ones (Table 1). Except for strains BC1, Chic3 and JF9, the identification (morphological and molecular) and pathogenic capacity of the other strains were reported in the works of Adame-García et al. (2015), Casillas-Isiordia et al. (2017) and Flores-de la Rosa et al. (2018). The morphological and genetic properties of the strains BC1, Chic3 and JF9, and pathogenic capacity against V. planifolia, were evaluated following the protocols described in the aforementioned references (data not shown). All the strains are preserved in the Laboratory of Genetic and Plants-Microorganisms Interactions of the Faculty of Agricultural Sciences of the Universidad Veracruzana. The vanilla plants from which the fungi were obtained were collected from secondary forests (acahuales), jungle or vanilla plantation areas untreated with chemical fungicides.
Table 1 Origin and susceptibility to benzimidazoles and chlorothalonil of strains of F. oxysporum (pathogenic and non-pathogenic) isolated from V. planifolia.
| ID Cepa | Procedencia | Carácter | Fuente | Benomilo | Carbendazim | Tiofanato metílico | Clorotalonil |
|---|---|---|---|---|---|---|---|
| H5 | Veracruz | Patógeno | 1 | S | S | R | R |
| H9 | Veracruz | Patógeno | 1 | S | S | R | R |
| JF5 | Veracruz | Patógeno | 3 | S | S | R | R |
| A3 | Veracruz | Patógeno | 3 | S | S | R | R |
| V6M2 | Veracruz | Patógeno | 3 | S | S | R | R |
| JF9 | Veracruz | No Patógeno | 4 | S | S | S | S |
| N12 | Nayarit | Patógeno | 2 | S | S | R | R |
| N3 | Nayarit | Patógeno | 2 | S | S | R | R |
| BC1 | Nayarit | No Patógeno | 4 | S | S | R | R |
| Chic 2 | Oaxaca | Patógeno | 3 | S | S | R | R |
| Chic 6 | Oaxaca | No Patógeno | 3 | S | S | R | S |
| Chic 3 | Oaxaca | No Patógeno | 4 | S | S | R | S |
S= susceptible; R= resistant. Source: 1) Adame-Garcia et al. 2015; 2) Casillas-Isiordia et al. 2017; 3) Flores de la Rosa et al. 2018; 4) Present study
Preparation of spore suspension. The strains were activated and subsequently subcultured on PDA for 10 days at 25 °C in a microbiological incubator (Felisa). At the end of the incubation period, 5 mL of sterile distilled water with three drops of Tween 20 were added to the culture medium. A suspension sample of mycelium and conidia was then transferred to 15 mL conical tubes and centrifuged at 1000 rpm for 2 min. Conidia suspensions of 1x109 CFU were prepared by diluting the supernatant with sterile distilled water. The spore count was performed using a Neubauer chamber and microscopic observation.
Fungicide resistance tests. The present study evaluated the susceptibility of twelve strains of F. oxysporum to four commercial fungicides: thiophanate methyl (Cercobin M®), benomyl (Promyl®), carbendazim (Prozycar®), and chlorothalonil (Talonil 75®). The tests were carried out in Petri dishes using the hole-plate method, for which fungal cultures were prepared with 100 μL of the 1x109 CFU spore suspension and 25 mL of PDA. For this, the spore suspension was placed directly on the base of the Petri dish. Subsequently, ⁓ 25 mL of culture medium were poured on top at a temperature of ⁓ 40 °C. The conidia were spread into the agar plate with circular movements before the solidification of the culture medium. After solidification of the agar, a sterile hollow metal rod (a hollow punch) was used to cut out four cylindrical blocks of agar from equidistantly distributed points. Three concentrations of each type of fungicide were then introduced into the remaining holes: one according to the recommendations of the manufacturer (1% P:V), a higher one (2.5% P:V) and a more diluted one (0.5%). Ten microliters of the fungicide solution at the evaluated concentration were placed in each hole. As a control, 10 μL of sterile distilled water were placed in one of the holes. This test was repeated five times for each fungicide and for each strain of F. oxysporum under study. The cultures were incubated for five days at 25-26 °C in a microbiological incubator (Felisa). The diameter of the inhibition zone associated with each treatment was measured with a digital vernier. Resistant strains were identified by the absence of an inhibition halo around the hole into which the fungicide was placed.
Determination of the relationship between the pathogenic/non-pathogenic character of the F. oxysporum strain and its response to fungicides. The possible relationship between the pathogenic or non-pathogenic character of the F. oxysporum strains and their susceptibility or resistance to fungicides, was determined using a Generalized Linear Model (Binomial Dist., Logit Link Function) based on a binary data matrix (0 resistant, 1 susceptible). A hierarchical and complete linkage cluster analysis was performed using Euclidean distance. All analyzes were performed using the statistical analysis software R 2.12.2.
DNA extraction. DNA was extracted, using a protocol based on the use of CTAB proposed by Adame-García et al. (2016a), from ⁓ 50 mg of mycelium of each isolate of Fusarium oxysporum after 10 to 14 days of growth in PDA medium. DNA was quantified with a compact UV-Visible spectrophotometer (Thermo Scientific™ NanoDrop™ One).
Design of primers for the GPDH gene. It was necessary to design primers to amplify the GPDH gene since no reports were found in the literature. The primers were designed by searching the NCBI database for the amino acid sequence of the GPDH protein among those reported for F. oxysporum f. sp. lycopersici. The amino acid sequence was then converted into the corresponding nucleotide sequence using tBLASTn. The resulting sequence was searched for among the reported genomes of F. oxysporum. The sequences found were aligned using the Mega (Kumar et al., 2018), Primer3 Plus (Untergasser et al., 2007) and Primer-BLAST software packages, which yielded a total of 15 possible primer pairs (data not shown). Each primer pair was tested by in silico PCR on the NCBI platform. One of the selected primer pairs was P4f (5’-AAAGTCCACGCACTCCTCTG-3’) and P4r (5’- AGAATGGCGACGGAAACCTT-3’), which was generated with the Primer3 Plus software and resulted in a sequence that extended from base 506 to 1146 of the total size of the gene. The other primer pair selected was P13f (5’-CCTGCACAAGTCAACAAGCC-3’) and P13r (5’-GCGCAACTGTTGTCAACCAT-3’), generated with the Primers BLAST software and resulting in a sequence that extended from base 766 to 1710.
Amplification and sequencing of the β-tubulin and GPDH genes. The amplification of the β-tubulin gene was carried out with the primer set Tub1 (5’-AACATGCGTGAGATTGTAAGT-3’) and Tub22 (5’-TCTGGATGTTGTTGGGAATCC-3’) (Nosratabadi et al., 2018) under the following thermal conditions: initial denaturation at 94 °C for 5 min; 35 cycles of denaturation for 30 s at 94 °C, alignment for 45 s at 60 °C, extension for 80 s at 72 °C; and final extension at 72 °C for 7 min. The primer pairs P13f - P13r and P4f - P4r, designed in this study, were used for the amplification of the GPDH gene under the following thermal conditions: initial denaturation at 94 °C for 2 min; 35 cycles of denaturation at 94 °C for 2 min, alignment at 60 °C, extension at 72 °C for 60 s; final extension at 72 °C for 7 min. The amplification products were analyzed by horizontal agarose gel (1.8%) electrophoresis and ethidium bromide staining, which confirmed that the amplification products of the β-tubulin and GPDH genes corresponded to 950 bp and 450 bp, respectively. After electrophoresis, the amplification products were purified following the Wizard® SV Gel and PCR Clean-Up System kit protocol and sent to the Institute of Biotechnology (UNAM) for sequencing.
Bioinformatic analysis. The sequences were edited using the MEGA X software and the consensus sequences of each gene were obtained from the sequences generated with the sense and antisense primers for each strain under study. Each resulting sequence was analyzed using the BLAST algorithm. In order to make the 3D designs of the β-tubulin protein molecule and identify the motifs, the sequences were aligned and compared using the BioEdit software. To search for conserved domains of the β-tubulin gene in strains susceptible and resistant to thiophanate methyl, the sequences were translated into proteins and aligned with the MEGA X software (Kumar et al., 2018). The sequences of F. oxysporum f. sp. niveum Fon12105 (accession KX369333) and Fon53 (accession KX369332), used in the study by Petkar et al. (2017), were used for this purpose. Mutations were manually identified in the alignment, and the sequences of three resistant strains of F. oxysporum and the susceptible strain Fon12105 were compared. The EzMol software was used for the identification of protein motifs, and the Phyre2 software was used for the 3D modeling of the molecules.
Results
Fungicide resistance tests. The cluster analysis identified three groups, according to the response of F. oxysporum strains from vanilla to the fungicides under study: A) strains susceptible to benomyl and carbendazim, but resistant to thiophanate methyl and chlorothalonil; B) strains susceptible to benomyl, carbendazim and chlorothalonil, and resistant to thiophanate methyl; C) strains susceptible to the four fungicides under study (Figure 1). Group A comprised nine strains from Veracruz, Nayarit and Oaxaca; group B comprised two strains from Oaxaca; group C comprised one strain from Veracruz (Table 1). No relationship was found between the pathogenic character or the provenance of the strains and their susceptibility to fungicides (P > 0.05), according to the tests of the generalized linear model susceptibility ~ pathogenicity + provenance. It is important to note that no differences were observed in the response of the fungi due to the different concentrations of the fungicide used.
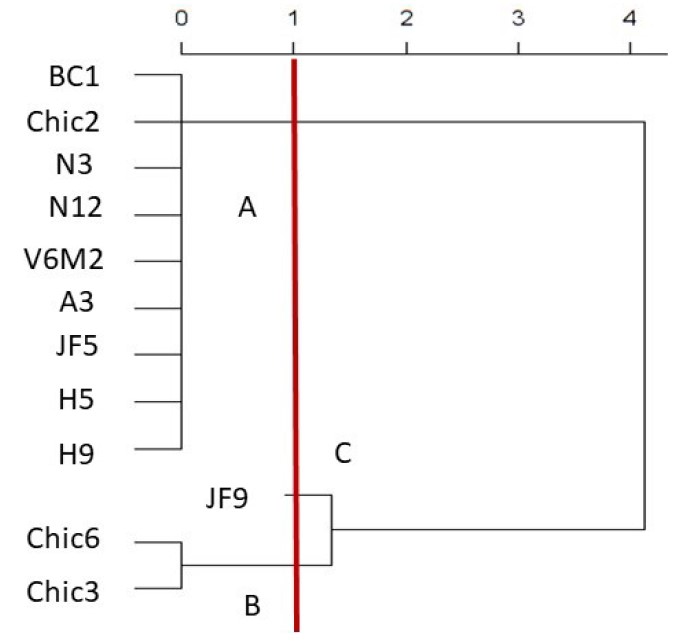
Figure 1 Grouping of F. oxysporum strains according to their susceptibility to benzimidazoles and chlorothalonil. A) susceptibility to benomyl and carbendazim, resistance to thiophanate methyl and chlorothalonil; B) susceptibility to benomyl, carbendazim and chlorothalonil, resistance to thiophanate methyl; C) susceptibility to all fungicides.
Amplification of β-tubulin and GPDH genes. In the 12 strains evaluated, the amplification produced a 950 bp fragment of the β-tubulin gene using the primers previously reported in the literature for this gene, and a 450 bp fragment of the GPDH gene using the P4F primers (Figure 3). No amplification product of the GPDH gene was obtained with the P13 primers.
Bioinformatic analysis. BLAST analysis confirmed that all the sequences obtained corresponded to the β-tubulin and GPDH genes, respectively (Table 2). The sequences of the pathogenic strains V6M2 and N3, and the non-pathogenic strains Chic6 and BC1, were used to find differences in the β-tubulin gene between the susceptible and resistant strains to the fungicides thiophanate methyl and chlorothalonil. The alignment analysis showed that the strain Fon12105 susceptible to thiophanate methyl (Petkar et al., 2017) and the resistant strain Chic6 shared the thymine base at position 200, while adenine was found in the rest of the sequences (Figure 2). For the analysis of the GPDH gene, six sequences of strains resistant to chlorothalonil (N3, JF5, A3, V6M2, Chic2 and N12) and one susceptible strain (JF9) were considered. No mutations of the GPDH gene were found in these sequences, which suggests that the susceptibility or resistance response is not related to variations in the sequence of the analyzed region of this gene.
Table 2 Sequences of Fusarium oxysporum strains registered in the GenBank-NCBI database that had significant alignments by BLAST to the β-tubulin and GPDH gene sequences of Fusarium oxysporum strains isolated from Vanilla planifolia.
| Secuencias de F. oxysporum en GenBank-NCBI | Porcentaje de similitud | ID cepa de F. oxysporum |
|---|---|---|
| F. oxysporum f. sp. vasinfectum strain ATCC 16612 beta-tubulin (BTU) gene, complete cds (Clave de acceso KT323825.1) | 98.72 | BC1 |
| F. oxysporum isolate TX85 beta-tubulin (BTU) gene, complete cds (Clave de acceso MH827996.1) | 97.85 | V6M2 |
| F. oxysporum isolate MS15 beta-tubulin (BTU) gene, complete cds (Clave de acceso MH827993.1) | 96.91 | N3 |
| F. solani strain MICMW-30.1 beta-tubulin gene, partial cds (Clave de acceso KX912242.1) | 94.34 | Chic6 |
| F. oxysporum f. sp. lycopersici 4287 glycerol-3-phosphate dehydrogenase Mrna (Clave de acceso XM_018394006.1) | 97.84 | N3 |
| F. oxysporum f. sp. lycopersici 4287 glycerol-3-phosphate dehydrogenase mRNA (Clave de acceso XM_018394006.1) | 95.60 | JF5 |
| F. oxysporum f. sp. lycopersici 4287 glycerol-3-phosphate dehydrogenase mRNA (Clave de acceso XM_018394006.1) | 95.32 | JF9 |
| F. oxysporum f. sp. lycopersici 4287 glycerol-3-phosphate dehydrogenase mRNA (Clave de acceso XM_018394006.1) | 99.29 | A3 |
| F. oxysporum f. sp. lycopersici 4287 glycerol-3-phosphate dehydrogenase mRNA (Clave de acceso XM_018394006.1) | 99.64 | V6M2 |
| F. oxysporum f. sp. lycopersici 4287 glycerol-3-phosphate dehydrogenase mRNA (Clave de acceso XM_018394006.1) | 100 | Chic2 |
| F. oxysporum f. sp. lycopersici 4287 glycerol-3-phosphate dehydrogenase mRNA (Clave de acceso XM_018394006.1) | 99.82 | N12 |
Identification of motifs. It was found that the sequences of the β-tubulin protein in the strains N3, BC1 and Fon53, which were resistant to thiophanate methyl, and the susceptible strain Fon 12105, share motifs 237537 and 335235. Strain N3 differs, as it has motifs 236152 and 279881, while the Fon 12105 strain was the only one to have the 237844 motif (Figure 3).
3D structures of target proteins. The three-dimensional structure of the β-tubulin protein showed differences between the susceptible strain and the strains resistant to thiophanate methyl. It was found that the resistant strains V6M2, BC1 and Fon53 have the amino acid arrangement histidine-glycine-serine, while strain Fon12105 has, at the same position, cysteine-cysteine-proline (Figure 4a). The GPDH proteins of the JF5 (resistant to chlorothalonil) and JF9 (susceptible to chlorothalonil) strains showed a different structure than the proteins of the resistant strains A3, V6M2, Chic2 and N12 (Figure 4b).
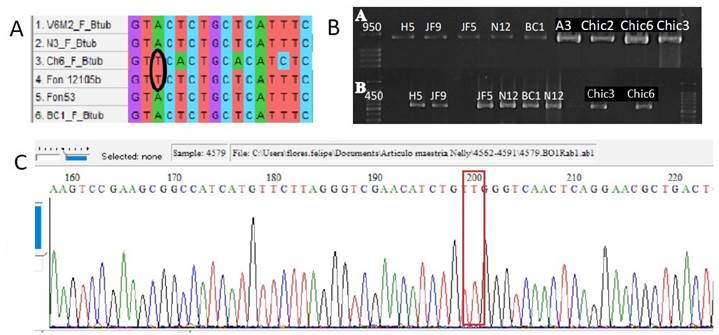
Figure 2 A) Fragment of the alignment of the β-tubulin gene sequences where it can be observed that the strains Ch6 (Chic6, present study) and Fon12105b (F. oxysporum f. sp. niveum, Petkar et al., 2017) share the thymine nucleotide at position 200 (framed in the rectangle). Chic6 is resistant to thiophanate methyl, while Fon12105b is susceptible. B) Amplification of PCR products in agarose gel of the β-tubulin gene (A) and GPDH gene (B) of some Fusarium strains under study. C) Electropherogram of the β-tubulin gene sequence of the Chic6 strain, resistant to thiophanate methyl, showing thymine at position 200.
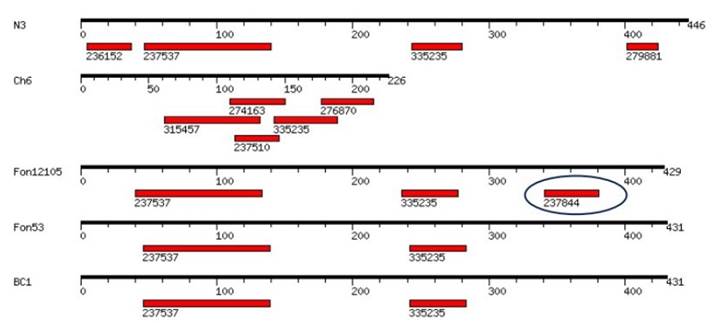
Figure 3 Search for conserved domains in the β-tubulin gene of strains of F. oxysporum that are susceptible and resistant to thiophanate methyl, isolated from V. planifolia. The 237844 motif in the susceptible strain Fon12105 (Petkar et al., 2017) was found not to be present in the resistant strains Fon53 (Petkar et al., 2017), N3 and BC1.
Discussion
The limited genetic variability of vanilla (V. planifolia) makes it highly vulnerable to various pathogens (Koyyappurath et al., 2015). The fungus F. oxysporum f. sp. vanillae, causal agent of root and stem rot disease, poses an important phytosanitary problem for vanilla producers worldwide (Pinaria et al., 2010; Adame-García et al., 2016b; Casillas-Isiordia et al., 2017).
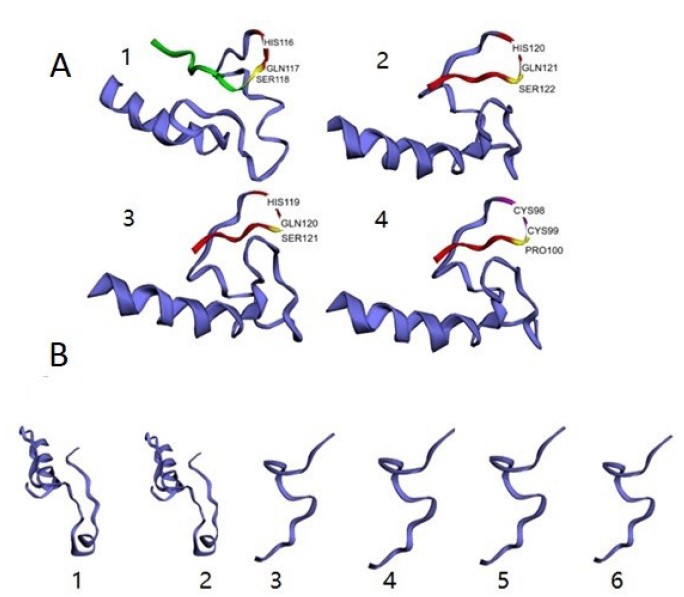
Figure 4. 3D models of the target proteins of: A) thiophanate methyl (1, strain V6M2; 2, strain Fon 53; 3, strain BC1; 4, strain Fon12105); B) chlorothalonil (1, strain JF5; 2, strain JF9; 3, strain A3; 4, strain V6M2; 5, strain Chic2; 6, strain N12). Structural differences are observed in both cases associated with resistance to fungicides.
Different types of control have been researched to combat this disease: genetic, cultural, biological and chemical. The latter is the most widely used by producers since it is cheap, effective and easily accessible. However, the excessive use of fungicides can lead to development of resistance to their active principles in fungi, as has happened with pathogens of other crops (Hobbelen et al., 2011; Steinberg and Gurr, 2020). Some species of the genus Fusarium, such as F. fujikuroi (Chen et al., 2014), F. graminearum (Duan et al., 2014; Zhou et al., 2016; Zhu et al., 2018), F. asiaticum (Suga et al., 2011), among others, have been reported as resistant to some fungicides such as benomyl, thiophanate methyl and carbendazim. In the case of F. oxysporum f. sp. vanillae, there are no previous reports of its resistance or susceptibility to fungicides.
In vanilla cultivation, the most used fungicides are from the benzimidazole group (Hernández-Hernández, 2019), such as benomyl, carbendazim and thiophanate methyl, which have systemic activity. Broad-spectrum multi-site contact fungicides such as chlorothalonil are widely recommended. When in contact with the fungus, benzimidazoles inhibit the assembly of microtubules through the degradation of β-tubulin, preventing the generation of the mitotic spindle, thus interrupting cell division (Chung et al., 2009). Chlorothalonil inhibits the activity of the enzyme glycerol-3-phosphate-dehydrogenase (GPDH) that is part of the glycolysis pathway of the fungus (Long and Siegel, 1975).
Benzimidazoles are widely used in agricultural production, although the 2019 FRAC list describes resistance to benzimidazoles as a common feature among fungal species, including some special forms of F. oxysporum, including lilii, gladioli (Chung et al., 2009), lycopersici (Amini and Sidovich, 2010) and niveum (Petkar et al., 2017). In the present study, all strains of F. oxysporum (pathogenic and non-pathogenic to vanilla) were susceptible to benomyl and carbendazim, while nine strains were resistant to thiophanate methyl regardless of their pathogenic character.
Petkar et al. (2017) proposed that the substitution of thymine (T) for adenine (A) in position 200 of the β-tubulin gene encodes two different amino acids, phenylalanine (TTC) and tyrosine (TAC), which are probably what determines the quality of susceptibility or resistance to thiophanate methyl, respectively. The comparison of the sequences of the pathogenic and non-pathogenic F. oxysporum strains of vanilla that are resistant to thiophanate methyl against the sequence of the susceptible (Fon12105) and resistant (Fon53) strains of F. oxysporum f. sp. niveum used in the study by Petkar et al. (2017) showed that the strains Fon12105 (susceptible strain) and Chic6 (resistant strain of F. oxysporum f. sp. vanillae) have a thymine (T) at position 200, while the rest of the resistant strains have an adenine (A) at the same position. The hypothesis that resistance to this fungicide is explained by a substitution in this specific position of the coding sequence of this gene is ruled out. It has previously been proposed that in Fusarium fujikuroi (Chen et al., 2014), as well as in other formae speciales of F. oxysporum (Chung et al., 2009; Amini and Sidovich, 2010), phenylalanine is found at position 200 of the gene, independently of the resistance or susceptibility to thiophanate methyl. Other studies attribute the resistance to benzimidazoles of Sclerotinia homoeocarpa to the mutation from phenylalanine to tyrosine (Dowling et al., 2017), and indicate that the reverse mutation tyrosine-phenylalanine is responsible for resistance in Colletotrichum musae (dos Santos-Vieira et al., 2017).
Due to the controversy regarding the mutation in codon 200 as the one responsible for the resistance or susceptibility of the fungus to the fungicide, the present study searched for conserved motifs in the gene sequences. It was found that the motif 237844 (Pfam) was only present in the susceptible strain, which suggests that in strains that are resistance to thiophanate methyl, the site of binding and action of the fungicide does not include the codon residue, as proposed by Vela-Corcía et al. (2018) in a study of the coupling between the action site of benzimidazoles and the protein β-tubulin. No further biological information was found for motif 237844 (Pfam).
Benzimidazoles interfere with certain sites associated with the polymerization of α1-tubulin, α2-tubulin and β2-tubulin monomers. However, once microtubule polymerization is complete, it is can no longer be reversed (Zhou et al., 2016). Polymerization inhibition depends on the active ingredient; thus, benomyl and carbendazim act on the α1/β2 and α2/β2 subunits, while thiabendazole shows a lower capacity to inhibit α2/β2 (Zhou et al., 2016), which could be related to a greater probability of resistance. Regarding thiophanate methyl, there are no data on how it affects these polymerization processes, and it may behave in a similar way to thiabendazole. Further studies are needed on the mode of action of thiophanate methyl to determine if this is the resistance mechanism of F. oxysporum f. sp. vanillae.
Chlorothalonil inhibits the fungal enzymatic activity of dehydrogenases, catalases, and acid phosphatases (Baćmaga et al., 2018); it specifically inhibits the activity of the GPDH enzyme (Long and Siegel, 1975). The high levels of resistance to chlorothalonil found in pathogenic and non-pathogenic strains of F. oxysporum associated with the root of V. planifolia contradict the annotations of FRAC (2019), which considers chlorothalonil as part of a group of fungicides with a low level of risk in the generation of resistance due to its multisite action. However, this fungicide has shown high effectiveness against other formae speciales of F. oxysporum such as F. oxysporum f. sp. lentis (Dahal and Shrestha, 2018).
Although the strains of F. oxysporum associated with vanilla did not present variations in the GPDH gene that explain the difference in the response to chlorothalonil, some characteristics of the conserved motifs of the enzyme could be associated with susceptibility. To determine the resistance mechanism, it is necessary to study other target enzyme systems for this and other fungicides, since Fusarium strains associated with vanilla have shown a high variation in enzymatic activity (Adame-García et al., 2011) as well as high genetic variability (Flores-de la Rosa et al., 2018), which explains the variability in their response to the fungicides under study. Regarding other multisite fungicides, such as mancozeb, a substantial reduction in susceptibility has been recorded in many pathogen species. This has been associated with genes involved in the formation of transcriptional machinery, regulation of cellular pH, and transporters of multiple drugs (Yang et al. 2019).
Conclusions
Pathogenic and non-pathogenic F. oxysporum strains of V. planifolia were highly susceptible to the fungicides benomyl and carbendazim, and highly resistant to thiophanate methyl and chlorothalonil. The point mutation from adenine to thymine in codon 200 of the β-tubulin gene, encoding phenylalanine/tyrosine, respectively, is not responsible for resistance to thiophanate methyl, as has been proposed by other authors. The presence of conserved motifs may be related to the susceptibility of the fungus to this fungicide. No evidence was found that the variability in the analyzed sequence of the GPDH gene is related to the response of susceptibility or resistance to chlorothalonil.











 texto en
texto en 


