Introduction
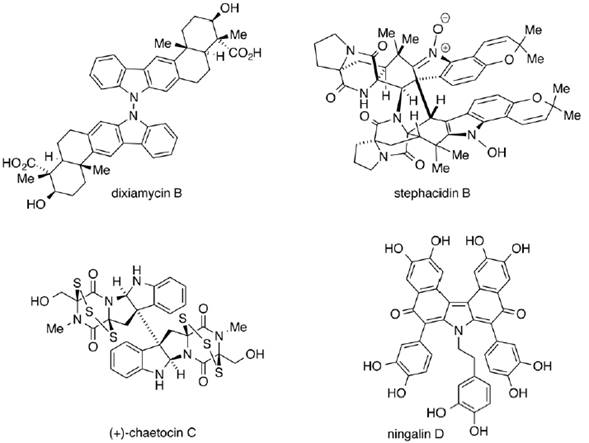
Oxidation processes on organic molecules are important transformations. Several examples of organic oxidations can be pointed out (Minisci, Citterio, Vsaimara, Fontana & De Bernardinis, 1989); however, those implying the homo-coupling of a single molecular unit to generate symmetric dimers are of special relevance. In this sense, it is possible to highlight only few relevant C2-symmetric naturally occurring compounds (He et al., 2012) (figure 1).
Source: Author’s own elaboration.
Figure 1. Relevance of the C2-symmetry highlighted by some naturally occurring organic structures.
As illustrated in figure 1, very important compounds are identified as organic dimers, which attract the synthetic chemistry industry attention for developing oxidative homo-dimerization procedures.
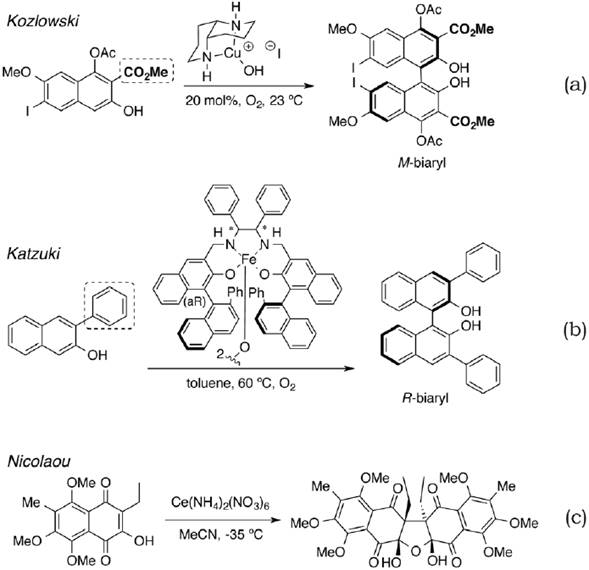
Among the great amount of oxidative homo-dimerization protocols (Dohi, Takenaga, Goto, Maruyama & Kita, 2007) described up to date (Li et al., 2010), the transition metal-catalyzed procedures have been continuously used in different publications. Among them, we can mention methodologies (Monguchi, Yamamura, Fujiwara, Somete & Mori, 2010) using Cu(I) (Sabbasani & Lee, 2015), Fe(III) (Smith, Nawrat & Moody, 2011), Mo (V) (Moritz & Siegfried, 2016) or Ce(IV) (Shukla, Rani & Tewari, 2012) as relevant examples (figure 2).
Source: Author’s own elaboration.
Figure 2. Representative examples for oxidative homo-coupling of aryl using different metals.
Copper-catalyzed enantioselective dimerization of the 2-acetoxy-3-hydroxynaphthalene derivative takes place under oxygen atmosphere conditions (figure 2a). Another assay in similar conditions was developed using Fe(III) as a catalyst and atmospheric oxygen (figure 2b); in this case, a 2-phenyl group is present in the starting material. Finally, an exquisite application of ammonium cerium(IV) nitrate in the naturally occurring total synthesis of hybocarpone is illustrated (figure 2c).
As revised, the target homodimers can successfully be synthesized. Specifically, in naphthalenes, an hydroxyl group adjacent to de reaction site was necessary.
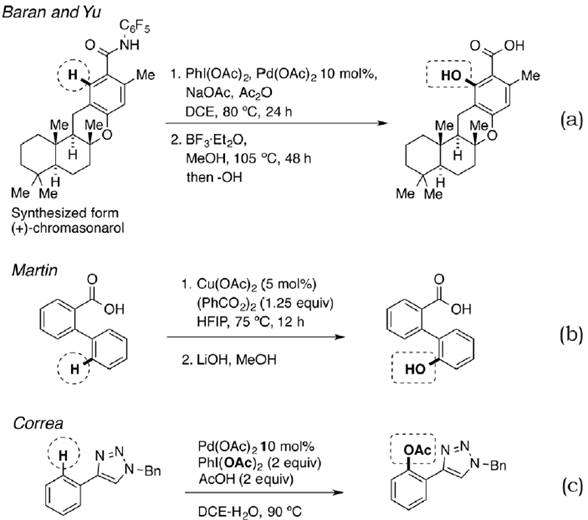
On the other hand, for the ‘Discussion’ section in this manuscript, it is important to mention some procedures regarding the direct C-O bond formation in aryls. In this sense, we can cite the work of Rosen et al. (2013), as well as Gallardo-Donaire & Martin (2013) and Novák, Correa, Gallardo-Donaire & Martin (2011) as significant representatives (figure 3).
Source: Author’s own elaboration.
Figure 3. Direct Csp2-O bond formation by palladium- and copper-catalyzed re actions.
Palladium-catalyzed acetoxylation of (+)-methylchromasonarol derivative followed by Lewis acid mediated amide hydrolysis give rise to a hydroxylation product (figure 3a). Moreover, the copper-catalyzed 6Hbenzo[c]chromen-6-one formation, from 2-arylbenzoic acids and concomitant hydrolysis, yield the corresponding hydroxylation (figure 3b). Finally, the direct acetoxylation of benzene derivatives, using triazoles as directing groups, lets to the direct C-O bond formation.
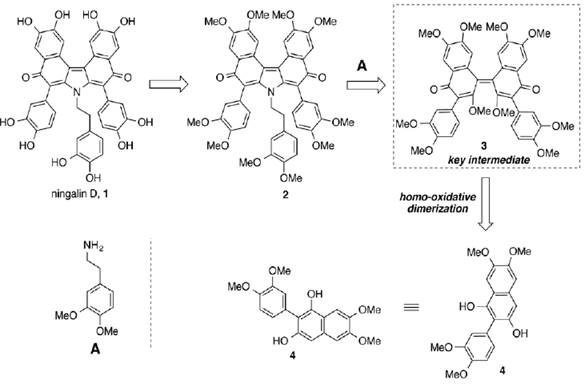
As part of this program towards the synthesis of molecules in cancer chemotherapy, the naturally occurring alkaloid ningalin D (Hamasaki, Zimpleman, Hwang & Boger, 2005) (figure 1) was identified as an excellent candidate in the treatment against multidrug resistance (MDR) cancer. The total synthesis of the alkaloid implies an oxidative aryl homo-dimerization as the key step, based upon the logical chemistry of its symmetrical architecture. The retrosynthetic analysis is outlined as follows:
This synthetic plan involves the synthesis of 1,3-naphthalenediol 4, with the properly functionalization, which would undergo oxidative homo-dimerization, yielding the key advanced intermediate 3. Finally, double 1,4-addition-elimination 2 and hydroxy group deprotection would give rise to the total synthesis of ningalin D, 1.
Materials and methods
Complete details on the synthesis of obtained compounds, as well as analytic and spectroscopic characterization data, are given in this section.
The glass material was dried with a heat gun previous to use, according to the Schlenk technique. The solvents (PhMe, MeCN, EtOH, MeOH) were anhydrous and deoxygenated.
Thin layer chromatography was carried out on silica gel supported on aluminum foil (0.25 mm). Adimensional retention factor (R f ) is reported. Purification of crude of reactions was carried out using column chromatography with a stationary phase in which silica gel (60-200 mesh size) is used as adsorbent.
The 1H and 13C NMR spectra were recorded in a pair of Bruker's AscendTM spectrometers (400 MHz and 500 MHz) using CDCl3. The chemical shifts (δ) for acquired spectra were reported relative to 0.0 ppm considering the TMS signal, or to 7.27 ppm by the residual signal in CDCl3.
Spectroscopic data are reported in the following order: chemical shift in ppm (δ), multiplicity, coupling constant in Hz (J) and integration. The multiplicities are reported as s (singlet), d (doublet), dd (doublet of doublets), t (triplet), c (quadruplet) or m (multiplet).
The melting points were determined using a FisherJones apparatus and they are reported in Celsius degrees (°C).
Results
In accordance with figure 4, the first step towards the synthesis of ningalin D is the preparation of the 1,3-naphthalenediol 4. A strongly recommended strategy, in total synthesis, is to develop a model system. The model systems are molecules structurally close related to the target, whose synthesis is easier and allows the evaluation of a promising or uncertain result in the complete route. Considering this idea, we decided to carry out the synthesis of a model molecule towards the preparation of ningalin (figure 4).
Source: Author’s own elaboration.
Figure 4. This synthetic strategy for the synthesis of ningalin D, sticking out the oxidative homo-dimerization as key step.
Thus, ningalin D analogue 5, would be prepared by double Michael addition-elimination of phenethylamine B in 6. Methylation of 7 will activate the free hydroxyl groups as better leaving groups to the aforementioned 1,4-addition-elimination reaction. As a consequence, 7 could be prepared via oxidative homo-dimerization of 8. Clearly it is possible to realize that 8 is a closely related to 4 (figure 5).
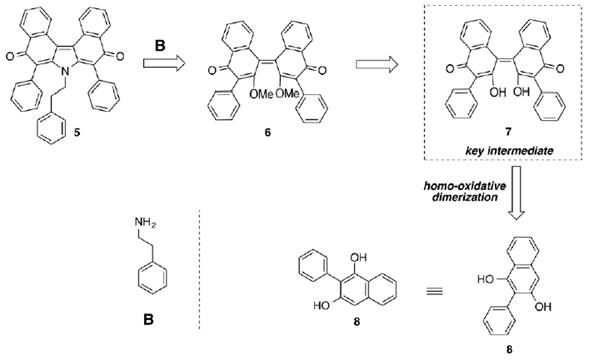
Source: Author’s own elaboration.
Figure 5. Model system strategy towards this proposed total synthesis of ningalin D.
Thereby, everything started synthesizing 8 (figure 6).
The route begins with the synthesis of 9 by reacting commercial inexpensive benzyl chloride with potassium cyanide in EtOH-H2O solvent system, under refluxing conditions. Remarkably, the step occurs on a hundred-gram scale and 79% yield. Nitrile hydrolysis and concomitant esterification give rise to the compound 10 in 84%. Claisen reaction between two single units of 10 in presence of sodium hydride produces 11 which is not purified and directly reacted with concentrated sulfuric acid yielding 8 in 42% yield. This reaction takes place on at least a five-gram scale allowing the preparation of several batches. It is worthy to highlight that this first route to 8 was carried out in multigram scale. This is an important feature to embark on a total synthesis program.
Once naphthol 8 synthesized, the oxidative homodimerization was the next step according to this strategy (figure 5). It was decided to explore transition metals as well as known organic oxidants (table 1).
Table 1 Conditions explored toward the synthesis of dimer 7, via oxidative homo-dimerization.
|
| |||
| Entry | Oxidant | mol%, Solvent, T (ºC) | Result |
| 1 | PIDA | 20, MeCN, 23a,b | Complex mixture |
| 2 | PIDA | 50, MeCN, 23 | Complex mixture |
| 3 | FeCl3 | 50, MeCN, 23 | Complex mixture |
| 4 | FeCl3 | 20, H2O, 50 | Complex mixture |
| 5 | FeCl3 | 20, DCE, 23a,b | Complex mixture |
| 6 | CAN | 50, MeCN, 23 | Complex mixture |
| 7 | CAN | 20, MeCN, 23a,b | Complex mixture |
| 8 | CuI | 50, MeCN, 23 | 12 in complex mixture |
| 9 | CuI | 20, MeCN, 23a,b | 12, observed in 30 min |
a Oxygen atmosphere was used. bThe progress of the reaction was followed by TLC along four days.
Source: Author’s own elaboration.
When the homo-dimerization reaction assays were carried out with PIDA (entries 1-2), only complex mixtures were observed. The use of FeCl3 (entries 3-5) or CAN (entries 6-7) only gave rise to decomposition of the starting material or complex mixture reactions. The use of CuI in either 20 mol% or 50 mol% clearly allowed the observation of a new formed spot.
After a complete and careful characterization, it was determined that compound 12 is formed instead of 7. The best conditions were found to be 20 mol% of CuI in presence of oxygen atmosphere.
To fully confirm the synthesis of compound 12, at least two large scale preparation on 1 g and 2 g were accomplished by using the previously developed con ditions (equation 1).
In that way, and also by comparison, the scalable preparation of compound 12 was confirmed, since the quinone in question had been already synthesized.
Discussion
The previously described results represent a totally difSource: Author’s own elaboration.ferent transformation to the desired one. The synthesis of this 2-hydroxy-3-phenyl 1,4-naphthoquinone implies the direct formation of a new Csp2-O bond and subsequent oxidation to generate the 1,4-naphthoquinoid system. To the best of this knowledge, this is the first example of a scalable oxidation on electron-rich naphthalenes using only Cu(I) and O2 as oxidants, and generating 1,4-naphthoquinones. This novel transformation has no precedent in copper-catalyzed direct C-O bond formation reactions. Even though similar protocols have been reported, in those cases cobalt-complexes with salen ligands were described as catalysts.
While an experimental evidence for an unequivocal mechanism requires additional studies, here in it is proposed a plausible reaction pathway, based upon the known chemistry of copper (Didziulis, Butcher, Cohen & Solomon, 1989) (figure 7).
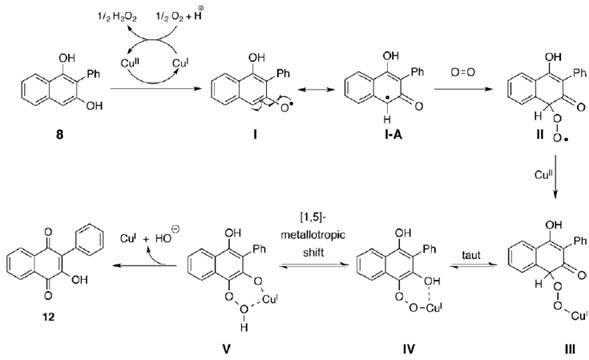
Source: Author’s own elaboration.
Figure 7. Proposed reaction pathway in the Cu(I)/O2-mediated oxidation of 1,3-na phthalendiol to 2-hydroxy-1,4-naphthoquinone.
The mechanism proposes the CuI to CuII oxidation by atmospheric oxygen (Didziulis et al., 1989). Thus copper (II) generates radical I/I-A which traps molecular oxygen giving rise to the radical specie II. Then reduction of CuII to CuI produces the copper(I) peroxide III. The following tautomerization produces IV in [1,5] metallotropic equilibrium with V. Finally the peroxide V, promote another redox cycle CuI to CuII which oxidize the hydroquinone to observed 1,4-naphthoquinone releasing CuI that get into another catalytic cycle.
Even though a [4+2] cycloaddition between 8 and O2 can be plausible, at this point we ruled it out on account of two reasons: 1) This is not a reaction with an atmosphere rich in O2, but molecular oxygen comes only from air which is a mixture of some other gases. 2) Naphthoquinones are not totally good dienes in a [4+2] cycloaddition, which imply that for achieving success in such a reaction under this pathway, additionally to Cu used as a catalyst, an abundant source of molecular oxygen would be necessary; nevertheless, this oxygen source is not present.
Conclusions
In conclusion, here in it was described the first example in a novel oxidation of a 1,3-naphtalenediol using only Cu(I) and molecular O2 from air. The procedure was successfully applied to the gram scale synthesis of the 2-hydroxy-3-phenyl-1,4-naphthoquinone. In regards to synthetic chemistry, the developed reaction features low cost, easy manipulation, efficiency and no high atmospheric pressures. Studies on the future scope are currently on going in this laboratory.











 nueva página del texto (beta)
nueva página del texto (beta)










