Introduction
The term «twiddler syndrome», coined in 1968, is used to describe the dysfunction of a cardiac implantable electronic devices (CIED) lead due to direct manipulation or spontaneous rotation of the generator.1 Although it involves less than 0.1% of all CIED in most cases lead replacement or repositioning is required.2 Currently, three different device rotation syndromes have been described (twiddler, reel and ratchet), each one of them associated with a unique pattern of lead coiling and dysfunction. We describe a case reel syndrome involving de left ventricular (LV) lead of a cardiac resynchronization-defibrillator (CRTD) device.
Case presentation
A 68-year-old man with ischemic cardiomyopathy and a left ventricular ejection fraction of 30% was seen in our institution for worsening heart failure symptoms despite optimal medical treatment (carvedilol 12.5 mg bid, furosemide 40 mg P.O., spironolactone 25 mg P.O., enalapril 20 mg orally bid). Atrial flutter with severe left and right atrial enlargement and persistently high ventricular response (heart rate: 120 bpm), despite the addition of digoxin to maximum tolerated doses of beta blockers, was considered responsible for the patient’s deteriorating health status, and AV node ablation with cardiac resynchronization-defibrillator (CRTD) implant was undertaken to control his heart rate.
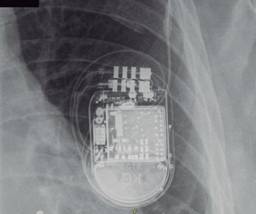
Both procedures were carried out uneventfully, and the patient was discharged the next day. One week after the procedure, the patient presented with a device pocket hematoma which developed after initiation of rivaroxaban 20 mg P.O.; transient suspension of his anticoagulant treatment and physical measures (ice packs and compression) were sufficient to achieve hematoma resolution. Adequate device function was confirmed with significant improvement of his heart failure symptoms. During his second follow up visit six months after device implant, loss of capture of the left ventricular (LV) lead was seen. Chest X ray (Figure 1) revealed a slightly pulled right ventricular electrode and a missing LV lead. Detailed image review (Figure 2) revealed the LV lead located behind the device generator, compatible with lead dislodgement due to «reel» syndrome. The patient underwent LV lead repositioning, during which a large device pocket was found and pocket remodeling (using absorbable sutures) was undertaken. During follow up, no further events of lead dislodgement have been found.

Figure 1: Chest X-ray of the patient. The device is in a left subcutaneous pocket, with two leads connected, but only one within the vascular space. An empty fixation sleeve is seen. The right ventricular lead is slightly pulled. The round radio-opaque images are pellets from a gunshot wound the patient suffered several years earlier.
Discussion
The first description of pacemaker lead malfunction due to generator rotation was made in 1968 by Bayliss et al. in patients with pacemakers.1 Termed «twiddler syndrome», it is associated with device rotation on its long axis (either by patient manipulation of the generator or spontaneous rotation) which produces coiling (and generally fracture) of involved leads. Since this initial description, multiple reports in patients with single and dual lead pacemakers, implantable defibrillators, resynchronization devices, and even abdominal devices have been published.3-6 Currently three different syndromes have been described (Figure 3):
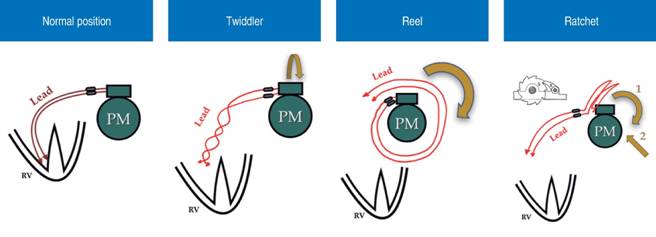
Figure 3: Differences between the three mechanisms of device rotation and lead displacement syndromes. PM, pacemaker; RV, right ventricle.
• Twiddler’s syndrome, produced by device rotation along its long axis, causing twisting and twining of the leads resulting in lead damage and displacement (Figure 4). In most cases, leads must be replaced due to extensive damage, including total lead rupture.7
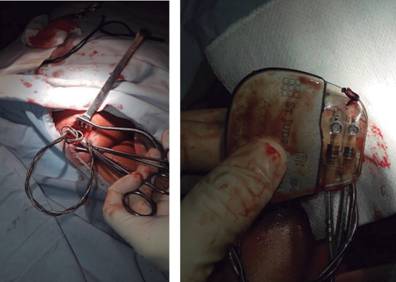
Figure 4: Lead damage in a twiddler syndrome. Twisting of the leads (left image) produces structural damage and dislocation. In this case, generator fixation with a non-absorbable suture (right image) did not prevent rotation of the device in a large pocket.
Reel syndrome, produced by device rotation along its short axis, causing lead coiling around the device generator. Lead damage is unusual, and repositioning of the involved leads is usually sufficient.
Ratchet mechanism, produced by initial lead retraction followed by a ratchet (cogwheel) mechanism, causing progressive retraction of the leads without twisting or coiling around the device.8
Risk factors include female gender, obesity, advanced age (these three possibly associated with increased tissue laxity); nonetheless, the most important (and preventable) risk factor is the creation of -a large generator pocket. In our case, the presence of a hematoma could have produced an enlargement of the pocket, allowing for device rotation (despite the elongated device shape). Other risk factors include participating in sports which include wide and repetitive movements of the arms, such as golfing and swimming.9 Lead dysfunction usually presents during the first weeks, but have been described as early as 17 hours and as late as one year after device implant.2,9 Interestingly, in reel syndrome not all leads are affected equally, and «selective» cases, as in our case, have been described.10,11
Several strategies have been proposed to avoid device rotation, including the creation of a small pocket; sub pectoral implant of the device; use of non-absorbable suture for fixation or polyester cases; use of active fixation leads and even immobilization of the upper extremity in the first week post implantation. However, none of these will be completely effective in preventing device rotation.2,12-14 Most patients will remain asymptomatic until lead dysfunction occurs, and presenting symptoms may be related to loss of capture (dyspnea, dizziness, exercise intolerance or syncope) or extra-cardiac stimulation (dysphonia, hiccups,; phrenic nerve, thoracic muscle or brachial plexus stimulation).14,15
Once diagnosed, lead replacement or repositioning, along with generator pocket remodeling, must be undertaken. If the generator pocket is very large, a new (smaller) sub pectoral pocket should be created to reduce movement of the device.
Conclusion
Lead malfunction due to device rotation is an uncommon complication associated with conditions that increase tissue laxity (advanced age, female gender, obesity) and the presence of a large implantation pocket. In the «reel syndrome», coiling of the leads around the device due to rotation of the generator along its short axis generally produce dislodgement without structural damage of the involved lead, thus allowing for lead repositioning.











 nueva página del texto (beta)
nueva página del texto (beta)