INTRODUCTION
Benthic diatoms are one of the main components of microphytobenthos communities (Shannon et al., 2012; Siqueiros-Beltrones, 1996). Depending on the type of substratum where benthic diatom assemblages (BDA) develop they are classified as epiphytes, attached to plant or algal surfaces; epizoic, living on animal surfaces; epipsammic that thrive on sand grains, and epipelic on muddy sediments; and epilithic on rocky substrata (Siqueiros-Beltrones, 2002).
Studies on various Mexican coasts have been carried out concerning the relation between BDA and the surfaces they inhabit (Hernández Almeida & Siqueiros-Beltrones 2012, López-Fuerte et al. 2019), mainly the surfaces of plant and algal hosts (Hernández Almeida & Siqueiros-Beltrones 2008, Ruiz-Fernández et al. 2012, Siqueiros-Beltrones & Martinez, 2017, Siqueiros-Beltrones et al. 2019, 2021). As such, the existence of an interaction between host and diatom taxa has been proposed, inasmuch the epiphytic diatom assemblages may vary according to the macroalgae taxon (Hernández Almeida & Siqueiros Beltrones, 2008) or even between individuals from the same species (Argumedo-Hernández & Siqueiros-Beltrones, 2008), suggesting a possible biochemical interaction (Lee et al., 1975). With respect to artificial surfaces, López-Fuerte et al., (2019) recorded a higher species richness of diatoms attached to fiber glass plates than on sediments, indicating that this type of substratum exerted no influence on the attaching capability of benthic diatoms to it.
Most studies on diatoms along Mexican coasts describe their association structure, including epiphytic forms growing on seagrasses and macroalgae, as well as epipsammic and epipelic forms (López-Fuerte et al., 2020; Martínez et al., 2019; Siqueiros-Beltrones et al., 2016; Siqueiros-Beltrones et al., 2021). However, very few works have compared associations from different substrata in highly variable environments as in coastal lagoons where, besides the differences due to the type of substratum, space-time variations in environmental conditions influence them (Desrosiers et al., 2013; Tornés et al., 2018; Zalat et al., 2021). Thus, the aim of the present study was to describe the BDA that thrive on live (epiphytes) and non-living (sediments) substrata in Terminos lagoon and determine the differences between them based on the parameters that define their structure such as: species composition and richness, species diversity, dominance, and equitability related to type of substratum and environmental variables in each sampling site. In this order, the hypothesis that significant differences were to be observed between sampled BDA, due to, the type of substratum and sampling site, reflecting the within variation that characterizes this type of coastal environment.
MATERIALS AND METHODS
Study area. Terminos lagoon (TL) is located in the coast of Campeche in the southern Gulf of Mexico (GM). With an extension of 706,147 ha, it is the second largest in the country. It interacts constantly with the GM through two openings: del Carmen mouth SW of the system, and Puerto Real mouth to the NE between Isla del Carmen and Isla Aguada (Ruíz-Esparza et al., 2014). Terminos lagoon is a shallow system (<4 m) characterized for high environmental variability both spatial and temporal. A high-fresh water influence is present on the south due, mainly to the Palizada, Chumpán, and Candelaria rivers (Ramos-Miranda et al., 2005).
Three different seasons take place throughout the year: a dry season (February to April) with low precipitation (<60 mm); a wet season (June to September); and “nortes” season (October to January) with predominant northerly winds.
Sample processing. In the wet season at each sampling site, measurements were made for transparency, pH, temperature, salinity, and oxygen concentration (Winkler technique), while water column samples were collected for quantifying nutrients (NH4+, NO3-+NO2-, PO43-, and SiO2) following Strickland & Parsons (1972).
To contrast the association structure of the BDA between live and non-living substrata, and as part of a more extensive sampling program in the lagoon (López-Mejía et al., 2022) samples of epiphytic and epipsammic diatoms were collected at four sites, visited only once for this study, where both types of substrates occurred. For the diatoms living on non-living substrata, around 100cc of superficial sediments were collected using 50 mL centrifuge plastic tubes, dragging the tube on the surface of the sediment. While for the live substrate macroalgae thalli (a Chlorophyta at site 21, and a Rhodophyta at site 24); and 2 or 3 bundles of Thalassia testudinium K. D. Koenig seagrass (at site 25) were collected; as well as in situ scraped-of-material from exposed mangrove roots during low tide (site 4), approximately 40cc of material were collected. Three subsamples were taken of each kind to generate a composite sample.
All samples were transported back to the laboratory, fixed in 70% alcohol, where the cleaning of the diatom frustules was performed using nitric acid and commercial alcohol for oxidating organic matter present in the samples (Siqueiros-Beltrones, 2002). Clean frustules were then rinsed repeatedly with distilled water until reaching a neutral pH; then, permanent slides were prepared using synthetic resin Pleurax (IR 1.7). Slides were inspected under a microscope at 1000× for taxonomic identification of diatoms to estimate their relative abundances.
Diatom identification to the lowest category possible based on frustule morphology was done following both classic literature (Peragallo and Peragallo, 1908; Schmidt et al., 1875) and recent works (Aké-Castillo et al., 2018; López-Fuerte et al., 2013; Siqueiros-Beltrones et al., 2021; Siqueiros-Beltrones et al., 2019; Vidal et al., 2017). To estimate the relative abundances of each taxon three permanent slides were inspected for every sample and the results were added to get a total for each kind of sample at every site, with an average of 1070 valves. A minimum of 400 valves per slide were counted. Where valves were scarce all valves found in the slide were counted: the total minimum was 445 for the non-living sample of the site 4.
Using the Olmstead-Tukey test for abundance-frequency data, the identified taxa were classified into dominant (taxa with a relative abundance (RA) higher than 0.39% and a relative frequency (RF) higher than three samples (29%); constant (RA < 0.39% and RF> 29%), occasional (RA > 0.39% and RF < 29%); and rare (RA < 0.39% and RF < 29%).
Alpha diversity was computed using Shannon’s index (H’) as bits/taxon (Siqueiros-Beltrones & Argumedo-Hernández, 2005), Pielou’s equitability (J’), and Simpson’s dominance index (l). Also, the indicator value (IndVal; Formula 1) was estimated, which is based in the degree of specificity (abundance) and fidelity (frequency) of each species for recognizing the indicator taxa for the conditions they belong to.
Nindividualsij: number of individuals of the specie i in all the sites of the group or habitat j
Nindividualsj: number of individuals of the specie i in all habitats
Nsities ij: number of sites of the same habitat where the specie i occurs
Nsitiesj: total number of sites of the same habitat
Beta diversity between substrates was measured with Bray-Curtis and Jaccard indices, which are based on the relative abundances and presence-absence of taxa, respectively.
A multivariate approach was taken for establishing if significant differences existed between the BDA of live and non-living substrates using ANOSIM (analysis of similarity) and DCA (detrended correspondence analysis) to determine sample variation based on species composition. All the analyses were performed using program PAST 4.02 (Hammer, Harper, & Ryan, 2001).
RESULTS
Environmental variables. The lagoon was characterized by a multivariate gradient dominated by salinity, transparency, NO3 - and Chlorophyl a (López-Mejía et al., 2022) (Table 1); areas with low transparency and high chlorophyl a concentration were located near the Palizada river and Xibuja estuary, and del Carmen city. A second gradient running west to east was identified, mainly related to O2%, silicates and pH (López-Mejía et al., 2022).
Table 1 Measurements for environmental variables in Laguna de Terminos, Campeche, Mexico. MAD: Median Absolute Deviation.
| Site | Depth (m) | Transparency (m) | Temperature (ºC) | Salinity | pH | O2% | NH4+ (µM) | FRS (µM) | NO3- (µM) | SiO2 (µM) | Clor_a |
| 4 | 0.9 | 0.9 | 32 | 25 | 8.3 | 102.21 | 2.59 | 3.23 | 0.59 | 9.63 | 3.57 |
| 21 | 0.7 | 0.7 | 33 | 23 | 8.23 | 46.74 | 0.73 | 3.54 | 0.81 | 11.38 | 0.96 |
| 24 | 0.7 | 0.7 | 31 | 15 | 8.1 | 48.30 | 0.32 | 3.54 | 0.59 | 10.64 | 1.36 |
| 25 | 2.9 | 1.1 | 31 | 25 | 7.96 | 61.11 | 0.17 | 3.52 | 0.54 | 9.57 | 2.26 |
| Median | 0.75 | 0.8 | 31.5 | 24 | 8.165 | 54.70 | 0.53 | 3.53 | 0.59 | 10.14 | 1.81 |
| MAD | 0.05 | 0.1 | 0.5 | 1 | 0.1 | 7.18 | 0.28 | 0.01 | 0.03 | 0.54 | 0.65 |
During this study, the highest O2% was registered on site 4, along with both the highest concentration of ammonium and Chlorophyll a; on the other hand, the lower O2% was on site 21, coinciding with the lower chlorophyll a and the highest concentration of nitrates. The transparency presented high values, allowing the light to reach the lagoon sediments and therefore the microphytobenthic community, reflecting low wave energy and a low sediment resuspension on the sites sampled.
Diatom assemblages. A total of 259 benthic diatom taxa (Supplementary material) were identified belonging to 75 genera. Out of these, 16% of the taxa were classified as dominant, another 16% were classified as constant, 8% as occasional, and the remaining 65% as rare. The taxa classified as dominant represent 89.9% of the total relative abundance.
Floristics by substrate yielded 159 epiphytic diatom taxa, including 59 exclusive taxa for live substrates. Whilst epipsammic assemblages comprising a total species richness of 200 taxa, 50% were exclusive of non-living substrates. Both types of substrata shared 100 taxa.
Computed values of diversity (H’) show a gradual differentiation that corresponds with S values and low dominance values, indicating that the number of dominant/constant taxa are in a relative high proportion among the different diatom assemblages represented in the samples. In general, BDA in samples from live substrata showed lower diversity values than those of non-living substrata from the same sites (Table 2).
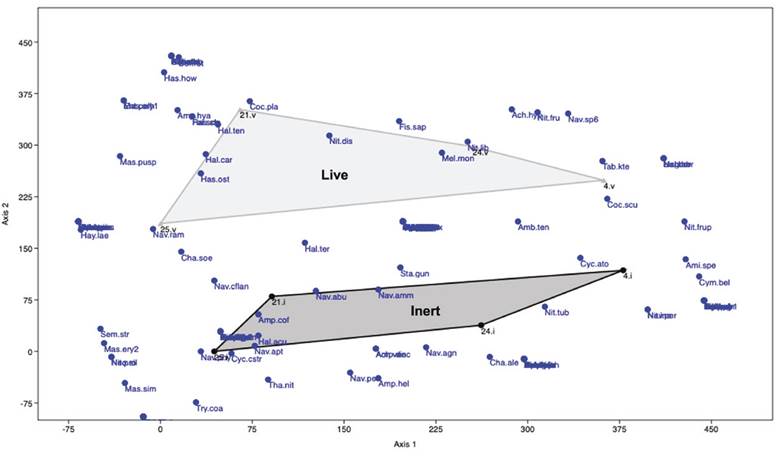
Figure 2 Site ordination DCA scattergram based on the BDA. Polygons show assemblages of samples for live (Light gray polygon) and non-living substrates (dark gray polygon).
Table 2 Computed values of diversity for each sample. L: live substrate, I: non-living substrate. S=Species richness; H’=Shannon’s diversity; J’=Equitability; l=Simpson’s dominance. Ind=Individuals
| Samples | Average | ||||||||||
| 4.L | 4.i | 21.L | 21.I | 24.L | 24.I | 25.L | 25.I | General | Live | Non-living | |
| Ind | 1183 | 529 | 1244 | 1292 | 1336 | 1330 | 1159 | 1247 | 1165 | 1230 | 1099 |
| S | 44 | 54 | 78 | 69 | 61 | 84 | 96 | 116 | 75 | 70 | 81 |
| H’ | 3.12 | 3.25 | 3.82 | 4.46 | 3.42 | 4.08 | 4.34 | 5.01 | 3.94 | 3.68 | 4.20 |
| J’ | 0.65 | 0.64 | 0.69 | 0.83 | 0.65 | 0.72 | 0.75 | 0.83 | 0.72 | 0.69 | 0.76 |
| l | 0.13 | 0.23 | 0.11 | 0.04 | 0.13 | 0.09 | 0.09 | 0.03 | 0.11 | 0.12 | 0.10 |
Average diversity (H’) reached a relatively high value of 3.93 bits/ taxon, with the higher value computed for the sediment sample of site 25.i (5.01 bits/taxon) and the higher values of S (116 taxa) and equitability (J’= 0.83), and the lowest dominance (l =0.03). In contrast, the lowest diversity (H’=3.12) was recorded for the live substratum sample at site 4.L, along with the lowest S (44 taxa). Whilst the highest dominance (l=0.23) and lowest equitability was recorded for the BDA of the sediment sample at site 4.I.
Forty-three taxa show IndVal values over 50%; 19 correspond to live substrates and 24 to non-living substrates (Table 2). Species best representing live substrates were: Nitzschia frustulum (Kützing) Grunow (95.4%), Navicula sp.6 (100%) and Tabularia ktenoeoides M. Kuylenstierna (100%). While, in the case of non-living substrates other taxa stood out: Halamphora acutiuscula (Kützing) Levkov (91.2%), Navicula agnita Hustedt (94.6%) and Tabularia fasciculata (C. Agardh) D. M. Williams & Round (100%) with higher values of the IndVal (Table 3). No coincidence occurred in the taxa IndVal values for the two compared substrates, indicating the occurrence of different dominating taxa.
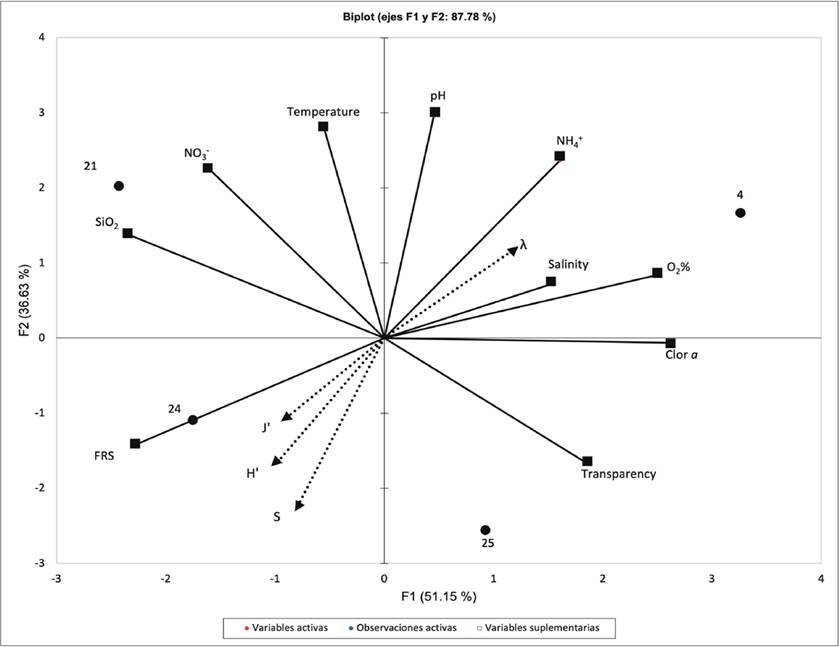
Figure 3 PCA plot with diversity index as supplementary variables (triangles). Active variables (squares); Sites (points)
Table 3 Results of indicator values (IndVal). *Taxa exclusive of non-living substrata; **Taxa exclusive of live substrata.
| Taxa | Substrate | |
| Live | Non-living | |
| Achnanthes ventralis (Krasske) Lange-Bertalot | 5.0 | 67.5 |
| Amphicocconeis discrepans (A. W. F. Schmidt) Riaux-Gobin, Witkowski, Ector & Igersheim* | 0.0 | 75.0 |
| Amphora coffaeiformis (C. Agardh) Kützing | 14.3 | 85.7 |
| Amphora graeffeana Hendey* | 0.0 | 75.0 |
| Amphora helenensis M. H. Giffen | 2.4 | 67.9 |
| Amphora hyalina Kützing var. inpalpabilis Proshkina-Lavrenko | 69.6 | 7.6 |
| Amphora laevissima var. perminuta Grunow | 2.1 | 68.8 |
| Berkeleya rutilans (Trentepohl ex Roth) Grunow | 74.2 | 0.3 |
| Chamaepinnularia alexandrowiczii Witkowski, Lange-Bertalot & Metzeltin* | 0.0 | 50.0 |
| Chamaepinnularia soehrensis (Krasske) Lange-Bertalot & Krammer | 4.7 | 81.3 |
| Cocconeis placentula Ehrenberg | 67.9 | 7.1 |
| Cocconeis scutellum var. scutellum Ehrenberg | 65.3 | 6.5 |
| Cyclotella cf. striata var. mucronulata Chi & Chang | 17.8 | 76.3 |
| Eunotogramma laeve Grunow* | 0.0 | 75.0 |
| Halamphora acutiuscula (Kützing) Levkov | 6.6 | 91.2 |
| Halamphora aponina (Kützing) Levkov | 67.5 | 2.5 |
| Halamphora caribaea (Wachnicka & E. E. Gaiser) Rimet & R. Jahn | 66.2 | 2.9 |
| Halamphora tenerrima (Aleem & Hustedt) Levkov | 60.5 | 14.5 |
| Halamphora terroris (Ehrenberg) P. Wang | 51.2 | 36.6 |
| Haslea howeana (Hagelstein) Giffen | 66.4 | 8.6 |
| Haslea stundlii (Hustedt) Blanco, Borrego-Ramos & Olenici* | 0.0 | 75.0 |
| Hyalosynedra laevigata (Grunow) D. M. Williams & Round | 72.7 | 1.5 |
| Karayevia amoena (Hustedt) Bukhtiyarova** | 75.0 | 0.0 |
| Melosira cf. moniliformis C. Agardh | 57.8 | 11.5 |
| Navicula agnita Hustedt | 2.7 | 94.6 |
| Navicula ammophila Grunow | 33.1 | 66.9 |
| Navicula apta Hustedt | 9.8 | 87.0 |
| Navicula directa (W. Smith) Brébisson | 21.0 | 54.0 |
| Navicula phyllepta Kützing | 0.3 | 74.1 |
| Navicula ramosissima (C. Agardh) Cleve | 82.4 | 8.8 |
| Navicula sp.6** | 100 | 0.0 |
| Nitzschia cf. dissipata (Kützing) Rabenhorst* | 0.0 | 50.0 |
| Nitzschia dissipata (Kützing) Rabenhorst | 59.4 | 5.2 |
| Nitzschia frustulum (Kützing) Grunow | 95.4 | 4.6 |
| Nitzschia incognita Lagler & Krasske | 15.4 | 79.4 |
| Nitzschia laevis Frenguelli | 5.0 | 60.0 |
| Nitzschia libertruthii Rabenhorst | 88.8 | 5.6 |
| Staurosira leptostauron var. dubia (Grunow) Edlund | 55.0 | 6.7 |
| Staurosirella gunter-grassi (Witkowski & Lange-Bertalot) E. A. Morales, C. E. Wetzel & Ector | 25.5 | 66.0 |
| Tabularia fasiculata (C. Agardh) D. M. Williams & Round* | 0.0 | 100 |
| Tabularia ktenoeoides M. Kuylenstierna ** | 100 | 0.0 |
| Thalassionema nitzschioides (Grunow) Mereschkowsky | 9.8 | 60.3 |
| Tryblionella coarctata (Grunow) D. G. Mann* | 0.0 | 75.0 |
Similarity values, both presence/absence of taxa and with relative abundances, between samples were low (Table 4). In the first case, Jaccard index indicated that the highest similarity was between the live and non-living substrata at site 25, that also yielded a high value with the Bray-Curtis index (0.50). However, the highest similarity (0.54) was recorded between samples 21.L and 24.L, in accordance with the rest of the pairwise comparisons that yielded average values between live substrate samples of 0.43 and 0.42 for Jaccard and Bray-Curtis indices, respectively. On the other hand, non-living substrate samples showed average similarity values of 0.38 for Jaccard and 0.35 for Bray-Curtis.
Table 4 Similarity between samples. a) Jaccard index, b) Bray-Curtis index.
| a) | 4.L | 4.I | 21.L | 21.I | 24.L | 24.I | 25.L | 25.I | b) | 4.L | 4.I | 21.L | 21.I | 24.L | 24.I | 25.L | 25.I | |
| 4.L | 1 | 4.L | 1 | |||||||||||||||
| 4.i | 0.39 | 1 | 4.i | 0.43 | 1 | |||||||||||||
| 21.L | 0.33 | 0.32 | 1 | 21.L | 0.33 | 0.30 | 1 | |||||||||||
| 21.i | 0.21 | 0.32 | 0.44 | 1 | 21.i | 0.20 | 0.28 | 0.41 | 1 | |||||||||
| 24.L | 0.38 | 0.26 | 0.53 | 0.34 | 1 | 24.L | 0.45 | 0.27 | 0.54 | 0.31 | 1 | |||||||
| 24.i | 0.31 | 0.32 | 0.39 | 0.50 | 0.46 | 1 | 24.i | 0.29 | 0.30 | 0.34 | 0.44 | 0.42 | 1 | |||||
| 25.v | 0.42 | 0.36 | 0.51 | 0.31 | 0.36 | 0.31 | 1 | 25.v | 0.33 | 0.34 | 0.47 | 0.31 | 0.38 | 0.26 | 1 | |||
| 25.i | 0.29 | 0.34 | 0.44 | 0.34 | 0.34 | 0.45 | 0.57 | 1 | 25.i | 0.23 | 0.28 | 0.37 | 0.36 | 0.29 | 0.42 | 0.50 | 1 |
However, despite the low similarity values in general, the ANOSIM test indicates that these differences are not significant to be considered distinct assemblages (p>0.05), which rejects the initial hypothesis.
Ordination of sites based on the DCA represents a gradient in species composition of the BDA in which samples from live and non-living substrates are segregated, as well as a gradient (from left to right; Axis 1) that follows the spatial location of the sampling sites (Figure 2).
This gradient also can be observed with the diversity index, with the highest value on site 25 located by the mouth of Puerto Real, followed by site 24 on the eastern side, site 21 on the southern coast of the lagoon, and site 4 located by del Carmen mouth presented the lower diversity. The result of the ANOSIM, with the sites as groups, shows significant differences between them (p < 0.05).
The PCA, with the diversity index as supplementary variable, shows an association between diversity and high FRS, and low Ammonium and Salinity concentration. The ordination of the sites based on the environmental conditions coincide with the DCA ordination. On the other hand, the result of CCA shows a correlation between the diversity index and the environmental conditions (p < 0.05).
In our study, dominant taxa exhibited distinct patterns of distribution: 1) By site, showing higher abundances such as the case for Amicula specululum (Witkowski) Witkowski in site E4; Berkeleya rutilans (Trentepohl ex Roth) Grunow at E21; Nitzschia tubicula Grunow at E24; and Hyalosynedra laevigata (Grunow) D. M. Williams & Round at E25; 2) by zone, as with Cymatosira belgica Grunow being the most abundant taxon in the West zone and Halamphora terroris (Ehrenberg) P. Wang in the East; 3) by substrate, with Navicula sp.6 and Nitzschia frustulum (Kützing) Grunow having the higher abundances on live substratum; and Navicula agnita Hustedt and Cyclotella striata (Kützing) Grunow that dominated non-living substrata; 4) homogeneous distribution without any preference as Navicula ammophila Grunow and Navicula apta Hustedt.
DISCUSSION
The benthic diatom assemblages from Terminos lagoon, exhibited high species richness and diversity that indicate favorable environmental conditions for their development, as suggested by Siqueiros et al. (2017), who propose that S higher than 50 taxa and species diversity H’ higher than 3.89 H’ for BDA are normal within undisturbed ecosystems, although displaced to the higher part of the modal values for most BDA (Siqueiros-Beltrones & Argumedo-Hernández, 2005).
The observed gradient in diversity depicts values becoming greater toward the area of marine influence (site 25 to 21), and the lowest on site 4, mostly influenced by the Palizada river discharge and the Pom-Atasta lagoon system (Carvalho et al., 2009).
The fact that the BDA from non-living substrates had higher species richness and diversity values in general seems to be somewhat a constant inasmuch in similar studies the same difference has been observed (Hernández-Almeida & Siqueiros-Beltrones, 2012), whilst numerous colonial species that thrive on living substrates may affect the estimated values of diversity (Siqueiros-Beltrones et al., 2016). Taxa dominating the epiphytic diatom assemblages belong to genera known for the efficiency in colonizing live substrates, such as Cocconeis, Navicula, Nitzschia and Amphora (Siqueiros-Beltrones & Argumedo-Hernández, 2005).
Those taxa having higher abundances in a specific zone or substrate occurred homogeneously in the correspondent samples, thus contributing in general to the low dominance detected. However, many other taxa were also abundant in a single site, generating low values of similarity between samples. Most computed values of similarity were <50% for indices (Jaccard and Bray-Curtis), which was also evident in the DCA scattergram where, based on the BDA, two groups were segregated, i.e., epiphytes and epipsammic. Thus, a certain influence can be observed, albeit low, of the substrate type on the supported BDA.
For both groups the depicted BDA for each sample were ordered according to their sampling site and their actual spatial location. This may reflect the influence of environmental variables at each site as it has been observed elsewhere (Frankovich et al., 2006; Kelly et al., 2009), and whose variability is very high in coastal ecosystems (Day et al., 2013). Terminos lagoon exhibits high temporal and spatial variations (Fichez et al., 2019; Guerra-Santos & Kahl, 2018) and usually is divided in zones related to the influence of surrounding systems. However it has been observed that these zones are not constant and that the lagoon presents a series of environmental gradients which vary spatially and temporally (López-Mejía et al., 2022). During the sampling, Terminos Lagoon presented multivariate gradients that shows the influences of the marine environment, the freshwater inputs and the del Carmen City; these gradients were related with transparency, salinity, nitrates, O2%, silicates and pH (López-Mejía et al., 2022). Some of these gradients coincide with the stations sampled in this study, therefore explaining the differences detected in the samples. The site 4 was related with a high O2%, which corresponds to the conditions observed for the zone, this is, under the influence of Pom-Atasta system at the West of the lagoon. On the other hand, the East sites (21, 24, and 25) have a lower O2%, but higher concentration of nitrates and silicates (López-Mejía et al., 2022). Moreover, the above description agrees with the typical patchy distribution proposed for BDA along environmental gradients (McIntire & Overton, 1971; Siqueiros-Beltrones, 2002).
Despite the low values of similarity, ANOSIM indicated that differences between samples of epiphytic and epipsammic BDA were non-significant. Nevertheless, significant differences between sites were observed. The above refutes the hypothesis that significant differences were to be observed between sampled BDA due to the type of substratum; but supports it with the differences of the sampling site, reflecting the within-site variation that characterizes this type of coastal environments plus an exchange of species between epiphytic and epipsammic substrata as observed in other coastal areas (Hernández-Almeida and Siqueiros-Beltrones, 2008). Actually, the epiphytic diatom assemblage in Terminos lagoon shared over 60% of its floristics with the epipsammic assemblage in contrast with further observations by Hernández-Almeida & Siqueiros-Beltrones (2012), who recorded differences between epiphytic and epilithic BDA from other marine environments. This can be related to the high environmental variation that characterizes coastal lagoons (Day et al., 2013; Escavarage et al., 2004), unlike the marine zones with more stable conditions (Fisher, 1977) where the low variability on the water column would allow that the community and its host interact for a longer time, reflecting on the BDA.
The diatom assemblages from the several live substrata sampled did not show differences between them either. Although the structure of the epiphytic diatom assemblages was somewhat typical of a live substrate, a particular specificity between host and BDA was not detected as proposed in other studies (Lee et al., 1975). In the same way, the high environmental variability may be reducing the importance of host and BDA interaction, taking a second place on the list of conditions that both influence and determined the structure of the community, mainly showing the environmental conditions on the BDA.
For some species, like B. rutilans, N. frustulum, and N. agnita no conclusive information was found to explain their patterns, but is known, for N. frustulum, that the environmental conditions determine its morphology (Trobajo et al., 2004). On the other hand, H. laevigata has been reported on individuals of Thalassia testudinum K. D. Koenig (Frankovich et al., 2006; López-Fuerte et al., 2013; López-Mejia, 2016), consistent with what was observed in this study finding its highest abundances at site 25 were an important patch of seagrass is located. An interesting case was N. tubicula, this taxon presented its greatest abundance at site 24, where stromatolite like formations are abundant and can be related with this taxon as Brake et al. (2004) reported for a system on Indiana, USA. These distribution patterns showing by dominant taxa could be reflecting the influences of the environmental condition and the different ways each taxon respond.
The results in this study may be indicating that the influence that the type of substrate may have on the diatom assemblages in coastal areas apparently not determine significant differences between assemblages from different substrata within the same ecosystem.
Apparently, the influence the substrate may have on the diatom assemblage could be similar to that of the spatial location from where the samples are collected, thus showing the importance of the environmental conditions (variables) overall on the BDA in Terminos lagoon.
The multivariate approach allowed us to compare BDA from two substrata based on both presence/absence and relative abundance of taxa with a better in-depth perspective of their structure. This has an advantage over studies based on classical single variable approach.
We thus conclude that in lagoon ecosystems like Terminos, where an environmental zonation exists due to differential combinations of physicochemical factors, BDA present fragmented or heterogeneous distributions that affect the forms colonizing a particular substrate. In this way BDA from similar substrata, either live or non-living, may show gradual variations as observed in this study. These may be representative of certain localities with distinctive species composition and particular parameter values, even though having similar overall structures.
The above gives way to the hypothesis that in coastal ecosystems characterized by highly variable environmental conditions, the structure of BDA will be influenced more by environmental variables rather than by the substrate that harbors them.











 nueva página del texto (beta)
nueva página del texto (beta)