Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Revista mexicana de fitopatología
versión On-line ISSN 2007-8080versión impresa ISSN 0185-3309
Rev. mex. fitopatol vol.35 no.2 Texcoco may. 2017
https://doi.org/10.18781/r.mex.fit.1610-1
Scientific articles
Effects of climatological factors on fluctuation of spores in mango trees cv. Ataulfo, in Guerrero, México
1Instituto Nacional de Investigaciones Forestales, Agrícolas y Pecuarias. Campo Experimental Iguala, Carretera Iguala-Tuxpan km. 2.5 Iguala, Guerrero. CP 40000.
2Universidad Autónoma de Guerrero. Unidad Académica de Ciencias Agropecuarias y Ambientales, Carretera Iguala-Teloloapan, Iguala, Guerrero. CP 40040.
3Instituto Nacional de Investigaciones Forestales, Agrícolas y Pecuarias. Campo Experimental Centro de Chiapas, Km 3.0 carretera Ocozocoautla-Cintalapa, Ocozocoautla, Chiapas. CP 29140.
The seasonal fluctuation of spores of the principal pathogens of mango was determined in San Jerónimo, Guerrero, Mexico. During the period October 2011 to December 2012 quantifications of total spores of Colletotrichum sp., Cladosporium sp. and Lasiodiplodia sp., were made at tree canopy level. Captured total spores occurred throughout the study period, with monthly variation; detecting peaks in September and October during the preflowering stage, with 1,844 total spores/week. Other peaks are in the second week of November, the third week of January and April, flowering and fruit growth, with 531, 648 and 1,139 total spores/week respectively. Low populations occur in May, June, July and august, during vegetative growth. The largest populations of Cladosporium sp., spores (1,533 spores/week), were in October coinciding with Colletotrichum sp., (160 spores/week). To Lasiodiplodia sp. the highest concentration was in September with 92 spores/week. The relative humidity was correlated with populations of Colletotrichum sp. (α<0.011), Lasiodiplodia sp. (α<0.009) and total spores (α<0.035). The dew point was correlated with Colletotrichum sp. (α<0.008) and total spores (α<0.059). The rain rate was positively correlated with Colletotrichum sp. (α<0.002) and Lasiodiplodia sp. (α<0.031). Using this information, modifications are proposed to the conventional management of diseases such as anthracnose and peduncle blight and rot.
Key words: Colletotrichum; Cladosporium; Lasiodiplodia; weather
Se determinó la fluctuación estacional de esporas de los principales hongos patógenos del mango en San Jerónimo, Guerrero, México. Durante el período de octubre 2011 a diciembre 2012 se cuantificaron las esporas totales de Colletotrichum sp., Cladosporium sp. y Lasiodiplodia sp. a nivel de copa de los árboles de mango. Las esporas totales capturadas se presentaron durante todo el periodo de estudio, con variación mensual; detectando picos poblacionales en septiembre y octubre, durante la etapa de prefloración, con 1,844 esporas totales/semana. Otros picos fueron en la segunda semana de noviembre, la tercera semana de enero y abril, en floración y crecimiento de frutos, con 531, 648 y 1,139 esporas totales/semana respectivamente. Las bajas poblaciones ocurrieron en mayo, junio, julio y agosto, durante el crecimiento vegetativo. La mayor población de esporas de Cladosporium sp. (1,533 esporas/semana) y Colletotrichum sp. (160 esporas/semana) fue en octubre, mientras que para Lasiodiplodia sp. fue en septiembre (92 esporas/semana). La humedad relativa fue correlacionada con poblaciones de Colletotrichum sp. (α<0.011), Lasiodiplodia sp. (α<0.009) y esporas totales (α<0.035). El Punto de rocío fue correlacionado con Colletotrichum sp. (α<0.008) y esporas totales (α<0.059). La tasa de lluvia mostró una correlación positiva con Colletotrichum sp. (α<0.002) y Lasiodiplodia sp. (α<0.031). Utilizando esta información se proponen modificaciones al manejo convencional de enfermedades como antracnosis, atizonamiento y pudrición del pedicelo.
Palabras clave: Colletotrichum; Cladosporium; Lasiodiplodia; clima
Mexico is the country with the fifth largest production of mango worldwide, and the second largest exporter, with 19.6 % (232,643 ton), after India with 24 % (286,775 ton) (FAO, 2016). Nationwide, the state of Guerrero provides 20 % (356,291 ton) of the country’s production, placing it as the first producer of mango, followed by Sinaloa (302,091 ton), Nayarit (252,394 ton), Chiapas (215,008 ton), Michoacán (155,360 ton), Oaxaca (146,029 ton) and Veracruz (118,544 ton). The substantial collaboration in the national production implies that the production of mango has an economic and social importance for Guerrero, since rural producers depend directly on this activity, and suppliers and people who can use the workforce depend on it indirectly. The region with the highest production and surface planted in the state of Guerrero is the Coast (SAGARPA, 2016); 33 % of its surface is covered with the variety Manila, followed by Ataulfo and Haden, and 30 and 16 %, respectively. The presence of mango diseases caused by pathogenic fungi and their impact on the production and quality of fruits has been widely studied by several authors (Ploetz, 2003; Acosta-Ramos et al., 2003; Guillén-Sánchez et al., 2007; Huerta-Palacios et al., 2009; Noriega-Cantú et al., 1999).
Colletotrichum gloesporioides (Penz.) Penz. and Sacc. is the causal agent of anthracnose in mango, and it causes severe economic losses to farmers of this and other crops in tropical and subtropical regions of the world. It is reported as a pathogen that affects leaves, inflorescences and fruits in the producing regions in Mexico, with the most important damage in postharvest (Ploetz, 2003; Acosta-Ramos et al., 2003). Fitzell and Peak (1984) determined that spores are the most important source of inoculant in Australia, and are produced on terminal branches, mummified inflorescences, flower bracts, and leaves. Under laboratory conditions, spores are produced in a wide range of temperatures (10-30 °C) and relative humidity of 95-97 %. In the Soconusco, Chiapas, the greatest damage was the blutting of inflorescences in the cv. Ataulfo, which takes place between November and February, after the rainy season has ended (Benítez-Camilo et al., 2003).
Cladosporium tenuissimum Cooke is reported as the causal agent of necrosis in flowers, peduncles and small fruits in the inoculated panicle of cv. Haden mango. This pathogen was reported for the first time along the coast of the states of Guerrero and Michoacán, Mexico. The organs affected were covered with a gray cotton-like mycelium, and a grayish-olive green sporulation. After its identification, the molecular characteristics represented C. tenuissimum (Guillén-Sánchez et al., 2007).
Lasiodiplodia theobromae Pat. is reported as the main causal agent of peduncular rotting in Costa Rica, and is considered the second most important disease after anthracnose; the fungus colonizes branch tissues endophytically before the establishment of inflorescences and weeks after flowering, it can reach the fruit’s peduncle, despite indications that these infections remain latent until the fruit ripens and the symptoms of the disease begin (González et al., 1999). Acosta-Ramos et al. (2003) report it is the pathogen that causes the rotting of the peduncle, one of the main diseases of mango fruit in postharvest in Mexico. Likewise, Ali et al. (2004) report it as the causal agent of the declination or descending death of branches, along with an abundant exudation of a reddish sap in stems and branches, with a tanning of vascular tissues. The knowledge of the availability and dispersion of inoculant of pathogenic fungi, as well as of the environmental factors that favor the release of spores is crucial to determine the periods of risk of infection and to implement preventive control measures. The aim of this investigation was to study the seasonal fluctuation of spores of the main pathogenic fungi of mangoes in Guerrero and their relation with weather conditions.
Materials and methods
The work was carried out in a period of 14 months, from the stage before flowering to the summer production and the beginning of the next flowering cycle (October 2011 to December 2012), in a mango orchard (Mangifera indica L.) cv. Ataulfo, aged eight years, with distances between trees of 7 x 7 m, in San Jerónimo, in the municipality of Benito Juárez, in the Costa Grande region, in Guerrero, (18° 15’ 38.3’’ North and 99° 28’ 53’’ from West, 30 m above sea level), which presents a warn subhumid climate (Aw1) (García, 1988).
Management of the orchard
The orchard was managed as follows: fertigation of N, P, and K, with the formula 120-40-80 fractioned in three applications; biofertilizer, Rhizophagus irregularis (=Glomus intrarradices), and Azospirillum brasilenses, 1.3 kg ha-1; dolomite (Ca 53 % y Mg 44 %) 980 kg ha-1; foliar fertilizer, 2 L ha-1 of chelated micronutrients, with three applications; periodical irrigation management, 6 hours every three days between November and May, using a micro spraying system, keeping the tree’s drip area within field capacity; management of pests and diseases with seven doses of chemical fungicides and insecticides.
Spreading of spores
In a period of 14 months of study, we examined the abundance of spores in the air, at the treetop level, in the experimental orchard, using a Burckard volumetric trap with a 7-day register (Gadoury and MacHardy, 1983). The spore trap was placed in the center of the experimental field, at a height of 2 m in the first third of the tree canopy. The spores were impacted on a cylindrical drum covered by transparent tape where they were deposited; the tape was cut into 39.5 mm sections that corresponded to each 24-hour period, and placed on a slide. Spores were counted in three transects at a 400x magnification, calculating the average observed per day. At least five spores were observed, another three transects were counted, and the average was calculated to report the concentration of spores accumulated in seven days. The trap was operated on a daily basis during flowering, setting and growth of the fruits.
The weather data were obtained using a Davis Vantage PRO2 weather station, which recorded the variables of temperature, relative humidity, wind speed and dew point. The weather variables were used to calculate the number of hours with relative humidity equal to or higher than 90, 80, 70, and 60 %, cold hours at 20 and 18 °C (HF20 and HF18).
Isolation of fungi
From November to December 2011 and January 2012, five trees were chosen at random, considering size, age, and uniform appearance, from which four branches were chosen, each pointing at a different cardinal point, at a height of 1.0 to 1.8 m, sampling one inflorescence per branch monthly, with symptoms of smutting and which were transferred to the Plant Pathology Lab of the Universidad Autónoma de Guerrero for the isolation of fungi present in rachis and petals of the affected inflorescences. The isolations were carried out over Petri dishes with potato-dextrose-agar (PDA) and subcultures were obtained from the edges of the cultures in growth. Monoconidial cultures were carried out using the streak agar plate technique. Morphometric identification of obtained isolates was carried out by using the codes by Ellis (1971), Barnett and Huntter (1998), and Heuchert et al. (2005).
Statistical analysis
Descriptive statistics were used to analyze the fluctuation of the weekly spore population. Correlation analyses (Pearson) were carried out between the density of the spores and the environmental variables, using the program SAS version 9.3 for Windows (SAS Institute 2010).
Results
In the trees of the experimental plot there were 6 flowering stages, which appeared in the period between late November and early February. Fruit setting was recorded between December 2012 and February 2013 and the fruit was harvested in March-April.
Dispersion of spores
Figure 1 shows the total fluctuation of spores captured, where we can notice the presence of spores throughout almost all the cycle of flowering, harvest, and vegetative growth, with a clear monthly variation. At the end of the stage of vegetative growth, in preflowering, the highest population was recorded, with 1,844 spores in September and October. In the stage of flowering and fruit growth, the highest capture took place in the third week of April with 1,139 spores, followed by the third and second weeks of January and November with 648 and 531 spores, respectively. The lowest populations were found in May, June, July, and August, when the vegetative growth of summer and most of the rainy season takes place. Also shown are the spore dispersions of Colletotrichum sp., Cladosporium sp., and Lasiodiplodia sp. The Colletotrichum sp. and Cladosporium sp. Spores have similar dispersions during the planting cycle, although Cladosporium sp. displayed a greater amount of spores caught. The highest peaks of both fungi appeared in September, October, April, and early June; practically since preflowering, the beginning of flowering until the end of the fruit harvest. In the case of Lasiodiplodia sp., the greatest spore populations appeared in September, October, and April, in preflowering, at the end of the fruit setting period and in the middle of the period of harvesting. These greater populations of spores from three fungi corresponds to the lowest temperatures in the dew point of 23.7, 23.7, 20.1, 18.2, and 23.1 °C corresponding to September, October, January, April, and June, where the existing water vapor begins to condense upon reaching these temperatures to form dew or fog. These increases also coincide with the humidity values above 75 % (Figure 2), excellent conditions for the formation and the release of spores.

Figure 1. Total capture of spores from October 2011 to December 2012, in the Ataulfo mango orchard, San Jerónimo, Municipality of Benito Juárez. Guerrero, Mexico.

Figure 2. Dispersion of Colletotrichum sp., Cladosporium sp., Lasiodiplodia sp. spores, temperature of dew point and relative humidity from October 2011 to December 2012, in the Ataulfo mango orchard. San Jerónimo, Municipality of Benito Juárez, Guerrero, Mexico.
The highest spore populations were found in the genus Cladosporium with 1,533 spores/week in October, which coincided with the period for Colletotrichum with 160 spores/week. On the other hand, the highest concentration for Lasiodiplodia was observed in September, with 92 spores/week. In these months, the phenological state of mango is preflowering.
The correlation analysis was applied considering the quantification of Colletotrichum, Cladosporium, Lasiodiplodia spores, total spores, and the weather parameters reported on the same evaluation period. The correlation coefficient was significant in three weather variables (Table 1). The weather variables that best explain the fluctuation of spores were relative humidity, the dew point and the rainfall present during the weeks of spore sampling. The weather parameter with a high correlation was relative humidity, for the fungus Colletotrichum (α≤0.011), followed by Lasiodiplodia (α≤0.009), and total spores (α≤0.035). The dew point was positively correlated with Colletotrichum (α≤0.008) and total spores (α≤0.059). Rainfall took place between May and October 2012 and the rate of rainfall was calculated by measuring the interval of time between each increase in precipitation; this variable showed a positive correlation with the populations of Colletotrichum (α≤0.002) and Lasiodiplodia (α≤0.031).
Table 1. Analysis of correlation between the concentration of spores and weather factors, applying Pearson’s correlation coefficient. Cycle 2011-12. San Jerónimo, Municipality of Benito Juárez, Guerrero, Mexico.
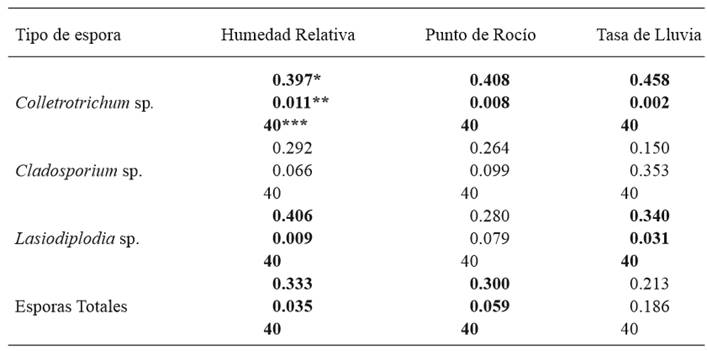
*Correlation coefficient; **level of significance; ***number of observations in the analysis of correlation.
Isolation of inflorescences. The fungi Colletrotrichum sp. and Lasiodiplodia sp. showed the highest frequency of 51 and 19 % in rachis respectively and 58 and 16 % in petals for both fungi. Meanwhile, Cladosporium sp. assumed a low frequency of isolation in the rachis of 3 % and was not isolated from the petals. Colletrotrichum sp. had an average isolation of 54.7 %, Lasiodiplodia sp., of 17.2 %, and Cladosporium sp., of 1.4 % per inflorescence (Table 2). The isolations were identified morphometrically; the Colletotrichum conidia were straight, cylindrical, with an obtuse apex and a truncated base; average size was 13.2 x 4.2 µm, which corresponded to the values indicated by Sutton (1992) for Colletrotrichum gloesporioides. In the case of Lasiodiplodia conidia, average size was 22.82 x 11.06 µm, located in the range indicated by Punithalingam (1976) for Lasiodiplodia theobromae. The measurement of Cladosporium conidia was carried out in digital images taken from a scanning electron microscope (Jeol® model 5800 LV), measuring, on average, 4.8 x 1.78 µm, with a variation of 1.5 - 1.6 x 0.28 - 0.62 µm. These values indicate a reduced variation, but they are 1.05 and 1.15 µm less than the size of spores reported by Guillén-Sánchez et al. (2007) for Cladosporium tenuissimum, who carried out measurements in a photomicroscope, and therefore with a different treatment to the study material, which explains the numerical differences recorded.
Discussion
Mango anthracnose, the causal agents of which are C. gloesporioides (Morales and Rodriguez, 2009) and C. acutatum (Rivera-Vargas et al., 2006), constitutes one of the most important diseases worldwide, since it affects both vegetative and reproductive organs, and it causes important post-harvest losses by causing rotting in the fruits (Acosta-Ramos et al., 2003; Ploetz, 2003; Benítez-Camilo et al., 2003). In an epidemiological study, Huerta et al. 2009 made similar observations with Colletotrichum gloesporioides; they reported that the greatest increase in anthracnose took place between December and April, when the minimum temperature at dew point (TminPR) as between 21 and 24 °C, which propitiate the condensation of water on leaves, flowers, and fruits, favoring the infection and dispersion by spore’s drag (tearing symptom) and spattering. This study found that the dew point temperature values were between 18.2 and 23.7 °C, recorded in September, October, January, April, and June; in these months, the phenological state is preflowering, flower blossoming, fruit setting until harvest. This wider dew point temperature range observed can be related to the specific temperature requirements for spore germination and formation of C. gloeosporioides appressoria in mango in different locations (Dodd et al., 1991; Arauz, 2000).
Cladosporium tenuissimum was recently reported as a mango pathogen (Guillén-Sánchez et al., 2007), in a study on the cv. Haden in the states of Guerrero and Michoacán, showing that C. tenuissimum caused necrosis in flowers, peduncle and small fruits in the inoculated panicles. They also reported that the affected organs are covered with a cottony grey mycelium and a grayish-olive green sporulation. The organs were susceptible from flowering until fruition. This report indicates the presence of the same causal agent in mango cv. Ataulfo, causing similar damages to those recorded in cv. Haden. Until recently, Cladosporium sp. was only pointed out to be an opportunistic and endophytic fungus, and an important contaminant of environments and cultivation media (Morales et al., 2009). However, there are reports on the fluctuation of spores and their relation with the environment, up to this paper, where a similar dispersal was found between Cladosporium sp. and Colletotrichum sp.; their spore populations increase when the dew point temperature was between 18.2 and 23.7 °C, causing the saturation of humidity in the air, forming dew and/or fog on the surface of the tree, favoring the infection and dispersal of spores from both fungi in the preflowering and flowering stages, up to the harvest of the fruit.
The results of the isolations in smutted inflorescences (rachis and petals) indicate the presence of well-known mango pathogenic fungi C. gloesporioides and L. theobromae (Fitzell and Peak 1984; Arauz, 2000; Ploetz, 2003; González et al., 1999). On the other hand, the low frequency of C. tenuissimum isolations may indicate that there was no colonization in petals and/or a heterogenous distribution of the fungus in the tissue, but it may also be a residual effect of the applications of fungicides and the possible presence of other microorganisms that interfere with the isolations. This work confirms the colonization of C. gloesporioides and L. theobromae as an important source for the development of the smut of the inflorescence and scarce fruit setting per inflorescence.
Anthracnose heavily damages the mango value chain in Mexico, reducing its productivity up to 90 %, and consequently, important economic losses for farmers (Espinosa et al., 2004). In addition, for its control, excessive contact and systemic fungicides are used. One proposal is of an integrated management with eight applications, starting with two applications in vegetative growth and the rest, during flowering and fruit growth (Acosta-Ramos et al., 2003). Another proposal is a program called “INIFAP”, in which they propose the use of 4 to 6 fungicide applications, from the beginning of flowering up to the first stages of fruit development (Espinosa et al., 2004). Knowing the stages of greater vulnerability to fungal attacks on mango trees will help adjust fungicide applications, reducing the negative impacts on the field workers, consumers and the environment.
Conclusions
Spore dispersal in the Ataulfo mango orchard had two important increases in the months corresponding to the final stage of vegetative growth and preflowering. The other increase in spores took place in flowering, fruit setting and growth, overlapped with the harvesting period.
Colletotrichum sp., Cladosporium sp., and Lasiodiplodia sp. spores had a high concentration between September and June, during the stages of preflowering, flowering, and up to fruit harvest.
The concentration of Colletotrichum sp. spores was positively and significantly correlated with dew point, relative humidity and rate of rainfall. Total spores were correlated with relative humidity and dew point.
Colletotrichum sp. and Lasiodiplodia sp. occurred as the main fungi in inflorescences with symptoms of smutting.
Acknowledgments
To the Foundation Produce de Guerrero A.C. for the funding of the projects “Management of anthracnose in mango in the Costa Grande Region” and “Technological validation of the management of anthracnose in mango in the potential regions of the state of Guerrero” in the 2011-13 announcement.
REFERENCES
Arauz, L.F. 2000. Mango Anthracnose: Economic impact and current options for integrated management. Plant Disease 84: 600-611. DOI D-2000-0328-01F [ Links ]
Acosta-Ramos M., Noriega-Cantú D.H., Nieto-Angel D., Téliz-Ortiz D. 2003. Efecto del manejo integrado del mango (Mangifera indica L.) en la incidencia de enfermedades y en la calidad de frutos. Revista Mexicana de Fitopatología 21: 46-55. Disponible en linea http://www.redalyc.org/articulo.oa?id=61221107 [ Links ]
Ali K.M., Mubeen L.A. and Shahzad S. 2004. Pathogenicity of Lasiodiplodia theobromae and Fusarium solani on mango. Pakistan Journal Botanical 36: 181-189. Disponible en línea https://www.researchgate.net/profile/Saleem_Shahzad/publication/266069789_Pathogenicity_of_Lasiodiplodia_theobromae_and_Fusarium_solani_on_mango/links/543d23540cf2c432f742531c.pdf [ Links ]
Benítez-Camilo, F.A., Huerta-Palacios, G., Holguín-Melendez, F., y Toledo-Arreola, J. 2003. Efecto de Colletotrichum gloeosporioides (Penz.) Penz. y Sacc. en la caída de frutos de mango cv. Ataulfo en el Soconusco, Chiapas, México. Revista Mexicana de Fitopatología 21:223-227. Disponible en líena http://www.redalyc.org/articulo.oa?id=61221218 [ Links ]
Dodd, J.C.; Estrada, A.B.; Matcham, J.; Jeffries, P.; Jeger, M.J. 1991. The effect of climatic factors on Colletotrichum gloeosporioides, causal agent of mango anthracnose, in the Philippines. Plant Pathology 40: 568-575. DOI: 10.1111/j.1365-3059.1991.tb02421.x [ Links ]
Espinosa, A. J., Arias S. J. F., Rico P. H. R., Miranda S. M. y Chávez C. X. 2004. Dinámica de daño y control de la antracnosis Colletotrichum gloeosporioides (Penz.) en mango en Michoacán. Folleto Técnico No. 2. INIFAP. CIRPAC. Campo Experimental Apatzingán, Apatzingán Michoacán, México. 21 p. Disponible en línea http://biblioteca.inifap.gob.mx:8080/jspui/bitstream/handle/123456789/1258/Dinamica_da%c3%b1o_1258.pdf?sequence=1 [ Links ]
FAO 2016. Food and Agriculture Organization of the United Nations. FAOSTAT www.faostat3.org, (consulta, marzo 2016) [ Links ]
Fitzell R.D. and Peak C.M. 1984. The epidemiology of anthracnose disease of mango: inoculum sources, spore production and dispersal. Annals of Applied Biology 104: 53-59. DOI: 10.1111/j.1744-7348.1984.tb05586 [ Links ]
Gadoury, D. M., and MacHardy, W. E. 1983. A 7-day recording volumetric spore trap. Phytopathology 73:1526-1531. Disponible en línea http://www.apsnet.org/publications/phytopathology/backissues/Documents/1983Abstracts/Phyto73_1526.htm [ Links ]
García, E. 1988. Modificaciones al sistema de clasificación climática de Köppen. México. Offset Larios, 217 p. [ Links ]
González E., Umaña G., Arauz L.F. 1999. Fluctuación poblacional de Botryodiplodia theobromae Pat. en mango. Agronomía Costarricense 23: 21-29. Disponible en línea http://www.mag.go.cr/rev_agr/v23n01_021.pdf [ Links ]
Guillén-Sánchez D., Yañez-Morales Ma. de J., Téliz-Ortíz D., Siebe-Grabach C. Bautista-Baños S. 2007. Morphological and molecular characterization of Cladosporium tenuissimum Cooke (Deuteromycotina: Hyphomycetes) on mango tree panicles: symptoms, pathogenicity and severity of the fungus. Fruits 62: 361-368. doi: 10.1051/fruits:2007032 [ Links ]
Huerta-Palacios G., Holguín-Meléndez F, Benítez-Camilo F.A., Toledo-Arreola J. 2009. Epidemiología de la Antracnosis [Colletotrichum gloeosporioides (Penz.) Penz. and Sacc.] en Mango (Mangifera indica L.) cv. Ataulfo en el Soconusco, Chiapas, México. Revista Mexicana de Fitopatología 27: 93-105. Disponible en línea http://www.redalyc.org/articulo.oa?id=61212195002 [ Links ]
Morales G. J.L., Rodríguez G. M. del P., Azpíroz R.H.S. y Pedraza S.M.E. 2009. Temperatura base in vitro de Colletotrichum gloeosporioides Penz aislado de frutos de aguacate (Persea americana Mill.) cv. Hass en Michoacán, México. Revista Científica UDO Agrícola 9: 414-420. Disponible en línea http://udoagricola.udo.edu.ve/V9N2UDOAg/V9N2TabladeContenido.pdf [ Links ]
Morales R. V. y Rodríguez G. M. 2009. Micobiota endofítica asociada al cultivo del mango ‘Haden’ (Mangifera indica L.) en el oriente de Venezuela. Revista Científica UDO Agrícola 9: 393-402. Disponible en línea http://udoagricola.udo.edu.ve/V9N2UDOAg/V9N2TabladeContenido.pdf [ Links ]
Noriega-Cantú, D.H., Téliz-Ortíz, D., Mora-Aguilera, G., Rodríguez-Alcazar, J., Zavaleta-Mejía, E., Otero-Colinas, G., and Campbell, C.L. 1999. Epidemiology of mango malformation in Guerrero, Mexico, with traditional and integrated management. Plant Disease 83:223-228. http://dx.doi.org/10.1094/PDIS.1999.83.3.223 [ Links ]
Ploetz, R.C. 2003. Diseases of Mango. Pp. 327-363. In: Ploetz R.C. (ed.) Diseases of Tropical Fruit Crops. CABI Publishing. Wallingford, UK. 544 p. http://dx.doi.org/10.1079/9780851993904.0327 [ Links ]
Punithalingam, E. 1976. Botryodiplodia theobromae. CMI description of pathogenic fungi and bacteria. No. 519. Commonwealth Mycological Institute. Kew, Surrey, England. doi:10.1007/BF01795180 [ Links ]
Rivera-Vargas, L.I, Lugo-Noel, Y., McGovern, R.J., Seiji, T., and Davis, M.J. 2006. Ocurrence and distribution of Colletotrichum spp. on mango (Mangifera indica L.) in Puerto Rico and Florida, USA. Plant Pathology Journal 5:191-198 http://dx.doi.org/10.3923/ppj.2006.191.198 [ Links ]
SAGARPA 2016. Secretaria de Agricultura, Ganadería, Desarrollo Rural, Pesca y Alimentación. Servicio de información agroalimentaria y pesquera. www.infosiap.siap.gob.mx (consulta, marzo 2016) [ Links ]
SAS Institute 2010. User’s Guide: Statistics, version 9.3, SAS Institute Inc. Cary, North Caroline, USA. [ Links ]
Sutton, B.C. 1992. The Genus Glomerella and its Anamorph Colletotrichum. Pp. 1-26. In: Bailey, J.A & M.J. Jeger (eds). Colletotrichum: Biology, Pathology and Control. CAB International, UK, 389 p. [ Links ]
Received: October 26, 2016; Accepted: February 09, 2017











 texto en
texto en 



