Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Revista mexicana de fitopatología
versión On-line ISSN 2007-8080versión impresa ISSN 0185-3309
Rev. mex. fitopatol vol.39 no.3 Texcoco sep. 2021 Epub 13-Dic-2021
https://doi.org/10.18781/r.mex.fit.2105-2
Scientific articles
Etiology of soybean (Glycine max) leaf spot in Sinaloa, Mexico
1 Departamento de Ciencias Biológicas, Universidad Autónoma de Occidente, Unidad Los Mochis, Boulevard Macario Gaxiola y Carretera Internacional s/n Los Mochis, Sinaloa, CP 81223, México;
2 Departamento de Biotecnología Agrícola, Centro Interdisciplinario de Investigación para el Desarrollo Integral Regional, Instituto Politécnico Nacional-IPN, Unidad Sinaloa. Boulevard Juan de Dios Bátiz Paredes No. 250. Guasave, Sinaloa, CP 81101, México.
Soybean is one of the most important legumes worldwide. In recent years, in the North of Sinaloa the incidence of a leaf spot has been observed at the growth stages of full seed and beginning maturity. The objective of the present study was to identify the causal agent of the disease. Symptomatic soybean leaflets were collected from eight commercial fields, from which eight fungal isolates were obtained and identified morphologically and molecularly. Pathogenicity tests were performed on soybean var. Nainari. The Koch postulates were fullfilled by the re-isolation of the original isolates from the inoculated plants. The morphological analyses determined the genus Curvularia associated to the soybean leaf spot. The phylogenetic analysis of the gene gapdh allowed the identification of Curvularia ahvazensis, C. muehlenbeckiae, C. spicifera and Curvularia sp. These species were pathogenic on soybean var. Nainari presenting variation in virulence since the area foliage diseased varied from 3 to 31%. The results open new research lines regarding the impact of the disease on yield and quality of soybean in Sinaloa.
Key words: Curvularia; leguminous; pathogenicity; gapdh
La soya es una de las leguminosas más importantes en el mundo. En años recientes en el norte de Sinaloa se ha observado incidencia de mancha foliar en la etapa de llenado de grano e inicio de la madurez. El objetivo del presente trabajo fue identificar el agente causal de esta enfermedad. Se colectaron muestras de foliolos sintomáticos en ocho lotes comerciales de soya, de los cuales se obtuvieron ocho aislados fúngicos, y se identificaron morfológica y molecularmente. Se realizaron pruebas de patogenicidad en plantas de soya var. Nainari y se confirmaron los postulados de Koch, mediante el reaislamiento de los aislados originales. Los análisis morfológicos determinaron al género Curvularia asociado a la mancha foliar de la soya. El análisis filogenético del gen gadph permitió la identificación de Curvularia ahvazensis, C. muehlenbeckiae, C. spicifera y Curvularia sp. Estas especies resultaron patogénicas en soya var. Nainari mostrando variación en virulencia, pues el área foliar afectada varió de 3 a 31%. Los resultados abren nuevas líneas de investigación relacionadas con el impacto de la enfermedad en el rendimiento y calidad de la cosecha de la soya en Sinaloa.
Palabras clave: Curvularia; leguminosa; patogenicidad; gapdh
The soybean (Glycine max) is one of the world’s most important legumes, with countries such as the United States, Brazil and Argentina standing out as the largest producers, the first two of which produce around 125 million tons and the latter, 50 million (FAO, 2020). The importance of the soybean in Mexico lies in the application of sub-products that have a high demand in the food market for the production of flour and edible oils. Mexico planted 155,000 ha of the crop in 2019, with a production of 233, 000 t and an average yield of 1.6 t ha-1 (SIAP, 2020). Sinaloa is the state with the most soybean planted under irrigation, with 10,331 ha, a production of 24,718 t and a yield of 2.4 t ha-1 during the spring-summer cycle (SIAP, 2020). The production and quality of the soybean worldwide has been limited by abiotic and biotic agents, including, particularly, diseases.
In the United States, annual losses due to diseases are estimated in 4.5 billion dollars (Bandara et al., 2020). Foliar diseases include brown spot (Septoria glycines), frogeye leaf spot (Cercospora sojina), leaf blight and purple seed stain (Cercospora kikuchii), anthracnosis (Colletotrichum truncatum), pod and stem blight (Phomopsis sojae), mildew (Peronospora manshurica), leaf spot by Alternaria (Alternaria spp.), ring spot (Corynespora cassiicola), bacterial pustule (Xanthomonas campestris pv. glycines) and bacterial blight (Pseudomonas syringae pv. glycinea) (Carmona et al., 2010). These diseases are known as late-season diseases (EFC), since they take place in plants in the stage of maturity, which coincides with a warm and rainy environment (Carmona et al., 2015).
In Sinaloa, C. sojina, C. cassiicola, C. kikuchii and P. manshurica have been confirmed as causal agents of the foliar spot disease in soybean (Rodríguez-Cota et al., 2019). Given the high incidence of the disease in recent agricultural cycles, this investigation was performed with the following aims: a) to identify the causal agent with morphometric and molecular studies, and b) to determine the pathogenicity of the fungi related to the disease in commercial fields of this legume during the summer of 2018.
Materials and methods
Collection of samples and obtaining monosporic isolates. Symptomatic soybean plant leaflets were collected in the del Carrizo and del Fuerte valleys between September and October, 2018 (Cuadro 1). Samples were taken from eight commercial soybean var. Nainari fields. These varied between 10 and 30 ha of crops, five in the stage of grain filling (R6) and three in the stage of the beginning of maturity (R7) (Fehr and Caviness, 1977); in each lot, the sampling method used (five of gold) was to select five plants, out of which five leaflets, each with light to dark brown foliar spots surrounded by a yellow halo were taken. The isolates of fungi related to symptomatic leaflets were carried out in agar water (AA; Bioxon; Cuautitlán Izcalli, State of Mexico, Mexico) following procedures described by Maraite et al. (1997). They were incubated at 25 ± 2 °C for 72 h, until mycelial growth was observed. To purify the isolates, hypha tips were transferred to potato-dextrose-agar (PDA; Bioxon; Cuautitlán Izcalli, State of Mexico, Mexico).
The monosporic isolates were obtained by serial dilutions, which were planted in the AA at 1.6% and incubated for 4 h; a germinated conidium was transferred from there to a PDA medium and the plants were incubated at 25 °C. Monosoporic isolates were stored at 15 °C in tilted test tubes with PDA, with added mineral oil (Flores-Córdova et al., 2013).
Table 1 Areas in which fungal isolates were obtained from symptomatic leaflets of soybean plants var. Nainari were collected in the del Carrizo and del Fuerte valleys.
| Muestra | |||||
|---|---|---|---|---|---|
| Aislado | Localidad | Fecha | Latitud Norte | Longitud Oeste | |
| Cv-3 | Ejido Chihuahuita, Ahome | 01/sept/2018 | 26° 9’ 56” | 109° 2’ 57” | |
| Cv-4 | Ejido Chihuahuita, Ahome | 01/sept/2018 | 26° 9’ 46” | 109° 2’ 26” | |
| Cv-5 | Poblado 5, Ahome | 01/sept/2018 | 26° 11’ 26” | 109° 3’ 24” | |
| Cv-8 | Poblado 5, Ahome | 01/sept/2018 | 26° 11’ 26” | 109° 3´43” | |
| Cv-11 | Ejido Chihuahuita, Ahome | 01/sept/2018 | 29° 9’ 12” | 109° 2’ 57” | |
| Cv-19 | El Carrizo, Ahome | 15/sept/2018 | 26° 18’ 44.43” | 108° 59’ 30.27” | |
| Cv-21 | El Carrizo, Ahome | 15/sept/2018 | 26° 18’ 1.01” | 109° 2’ 3.81” | |
| Cv-29 | Bachoco, Guasave | 02/oct/2018 | 25° 43’ 55.83” | 108° 46’ 33.98” | |
Phenotypic characteristics of mycelial cultures. The phenotypic characteristics of the cultures were determined with PDA. The cultures were incubated for 11 days at 25 °C, with a 12 h light-darkness regime, and the mycelial growth rate was determined (Kim et al., 2005), along with the color of the culture, using standard Kelly and Judd colors (1976).
Morphometry of the conidia of isolates. Fragments, 5 mm in diameter, were cut from the margin of the four-day-old cultures in PDA and transferred to the same medium. The shape and color of the septa, as well as the length and width of 40 conidia from each isolate were determined seven days after planting in the same medium and incubated at 25 °C. Preparations were then made with lactophenol blue to be measured in a micrometer installed in a compound microscope (Labmed; Lab America, Inc. Los Angeles, CA, USA).
Extraction of DNA, PCR and sequencing. The extraction of genonic DNA was carried out using 50-100 mg of mycelia in seven-day-old PDA cultures, using DNAzol as a reagent (Molecular Research Center Inc., Car. No. DN127, Cincinnati, OH, USA), following the instructions by the manufacturer. The quality and concentration of the DNA extracted was determined in a NanoDrop 2000c spectrophotometer (Thermo Scientific, Wilmington, DE). To confirm the identity of the isolates, the gene from the glyceraldehyde-3-phosphate dehydrogenase (gapdh) was amplified using primers gpd1 (5’-CAA CGG CTT CGG TCG CAT TG-3’) and gpd2 (5’-GCC AAG CAG TTG GTT GTG C-3’), which amplify a fragment of approximately 580 pairs of bases (Berbee et al., 1999). The PCR’s reaction took place in a final volume of 50 mL, which contained 1 mL of genomic DNA, GoTaq Flexi 1X Buffer, 1.25 mM of MgCl2, 0.2 mM of each primer, 0.2 mM of dNTP´s and 1 U of DNA polymerase GoTaq Flexi (Promega, Madison, WI, USA). Amplification conditions were as follows: initial denaturalization at 95 °C for 2 min, 35 denaturalization cycles at 95 °C for 1 min, alignment at 60 °C for 1 min, an extension at 72 °C for 45 s and a final extension at 72 °C for 10 min. The amplified fragments were visualized in a 2% agarose gel stained with ethidium bromide in a Chemidoc (BIO-RAD) and the bands of interest were split for their later purification using the QIAquick Gel Extraction kit (Qiagen, Hilden, Germany). The quantification od the purified PCR products were carried out with a NanoDrop 2000c spectrophotometer. The fragments were sequenced unidirectionally with the primer gpd1 in an AB3730 (Applied Biosystems, Foster City, CA, USA), in the genomic services lab of the National Genonic Laboratory for Biodiversity (LANGEBIO).
Phylogenetic analysis. The sequences were edited in the software BioEdit version 7.0.5.3 (Hall, 1999) and compared with other sequences deposited in the GenBank data base, using the BlastN algorithm. The sequences were aligned along with a set of 68 references sequences of the genus Curvularia and a sequence of Bipolaris maydis as an external group (Madrid et al., 2014; Manamgoda et al., 2015; Tomaso-Peterson et al., 2016; Mehrabi-Koushki et al., 2018; Tan et al., 2018; Kiss et al., 2019), using the MUSCLE aligner (Edgar, 2004) implemented in MegaX (Kumar et al., 2018). The phylogenetic analysis was carried out in MegaX, using the maximum likelihood method and the Hasegawa-Kishino-Yano (HKY) model, with a gamma distribution (four categories) and unvariable sites. All the gaps were considered in the analysis and the robustness of the topology was evaluated with 1000 bootstrap replications. Finally, the phylogram was edited using the software FigTree version 1.4.0 (Rambaut, 2014).
Pathogenicity tests in a greenhouse. To determine the pathogenicity of the eight isolates associated with the disease, soybean plants var. Nainari were used, which were planted in 5L plastic pots with a pasteurized substrate (sand 48.0, silt 19.4 and clay 32.5%, respectively and a pH of 7.0); one seed was placed in each pot. The plants were watered according to each one’s requirements and fertilized every week with Miracle-Gro (The Scotts Company LLC; Marysville, Ohio, USA), following the manufacturer’s recommendations. Four replicates were used for each isolate (four pots with one plant each).
Preparation of inoculum. The monoconidial isolates were developed in PDA at 25 °C, with a 12 h light-darkness regime for 10 days. The conidia were obtained by adding 10 mL of sterile distilled water to each culture and scraping with a sterilized spatula to release them. The conidial suspensions were filtered through a double cheese mesh to remove the mycelia (Prieto et al., 2016) and the inoculum concentrations of each isolate were adjusted from 5 x 104 to 7 x 104 conidia mL-1 with a hematocytometer.
The conidial suspensions were sprayed on plants (in the R6 stage) to the point of run-off. The control plants were sprayed with sterile distilled water. The plants were then covered for 24 h with 90 x 20 cm plastic bags with paper towels saturated with distilled water to achieve a relative humidity of 85-90% in their interior and they were kept at 22 to 28 °C; they underwent the same moisture and temperature conditions for 12 h a day, every day for seven consecutive days. The plants then remained for 21 days on the greenhouse benches, where temperatures ranged from 20 to 24 °C with relative humidity ranges of 50 to 75%. The inoculated and control plants were distributed in a totally random design. The pathogenicity of the isolates was determined 30 days after inoculation. Ten leaflets were collected at random from each plant and the percentage of the leaf area affected (LAA) was determined, following procedures described Soares et al. (2009). To comply with Koch’s postulates, after the experiments, isolates were carried out from symptomatic leaflets taken from inoculated plants. The identity of the isolates was confirmed based on the morphology of the cultures and the morphometry of twenty conidia from each isolate.
Statistical analysis. The percentages of LAA were transformed using the arcsine function (Gómez y Gómez, 1984) before performing an ANOVA. The comparison of means was carried out with Tukey’s test (Little and Hills, 1973), using the statistical package SAS® version 9.4 (1999). The experiments were held twice. Given that the statistical analyses indicated a significant interaction between isolates and the experiments, the results of both experiments are presented.
Results and discussion
Cultural and morphometric characteristics of fungal isolates. The phenotypic characteristics of the cultures, the conidiophores and conidia of eight isolates obtained during the sampling period (Table 1) were determined in PDA after eight days of incubation. This helped locate the isolates in the genus Curvularia (Manamgoda et al., 2015) genus. No fungal teleomorph was found during the sampling period, although the genus Cochliobolus has been confirmed as the teleomorph of some species of Curvularia (Marin-Felix et al., 2020). The phenology of the colonies, the characteristics of conidiophores and the morphometry of the Curvularia species are described below.
Isolates Cv-3, Cv-5 and Cv-8. Cultures with a mycelial growth rate of 3.7 - 4.4 mm/day on PDA, with a velvet-like aspect, olive-gray, with faint concentric circles (Figure1A); reverse side of the cultures with olive-brown pigmentation (Figure 1B). Straight, dark brown septate conidiophores, geniculated in the terminal part (Figure 1C); conidia, 15.0 - 27.5 μm in length by 7.5 - 15.0 μm in width, with 2 to 4 transversal septa, the middle septum being the bulkiest one and with the most pigmentation, a pronounced curvature and a dark hilum (Table 2; Figure 1D). The morphometric characteristics of these are similar to those recorded for C. muehlenbeckiae (Berbee et al., 1999; Madrid et al., 2014).
Isolate Cv-4. Cultures with a cotton- or suede-like appearance, with a mycelial growth rate of 4.0 mm/day on PDA, colored dark gray with uneven tones of brown to light gray (Figure 1E) and the front, dark golden brown in color (Figure 1F). Light-brown conidiophore and geniculated on the top part. Cylindrical conidia, with three to four distosepts, ellipsoidal, golden maroon in color (Figure 1G and H), 10.0 - 22.5 µm in length by 7.5 - 10.0 µm in width (Table 2). The cultural and morphometric characteristics are similar to those found in C. spicifera (Ayoubi et al., 2017; Qostal et al., 2019).
Isolate Cv-11. Olive-gray cultures, scarcely dense, with a velvetlike aspect, very noticeable concentric rings (Figure 1I) and a mycelial growth rate of 3.9 mm/day on PDA. The reverse side of the cultures dark to olive green (Figure 1J). Isolated and geniculated conidiophore in the terminal part, without ramifications (Figure 1K). Conidia between 16.2 and 27.5 µm in length by 6.2 to 10.0 µm in width (Table 2), with a smooth wall, ellipsoidal to cylindrical, rounded tips, with 2 to 4 transversal distosepta (Figure 1L), protuberant hilum (Arrow in Figure 1L), coinciding with the characteristics described for C. ahvazensis (Mehrabi-Koushki et al., 2018).
Isolates Cv-19, Cv-21 and Cv-29. Cultures with a mycelial growth rate of 2.8 - 4.3 mm/day in PDA, with a suede-like aspect, undefined concentric rings and whitish plumes (Figure 1M), and a dark maroon to black reverse side (Figure 1N). Conidiophore are erect to flexible, septated, often geniculated (Figure 1O). Conidia are 10 to 35 µm in length by 5 to 12.5 µm in width, ellipsoidal or rounded on the tips, pale to dark maroon, 2 a 5 distosepts (generally 4), smooth conidial wall (Table 2; Figure 1P y Q). The phenotypical characteristics of the cultures, as well as the morphometry of the conidia are similar to those in species phylogenetically related to C. hawaiensis (Manamgoda et al., 2015).

Figure 1 Morphology species of Curvularia in PDA related to leaf spot in soybean. A-D) Representative morphology of isolates Cv-3, Cv-5 and Cv-8 (C. muehlenbeckiae); A) Front side of the culture; B) Reverse side of the culture; C-D) Conidiophores and conidia. E-H) Isolate Cv-4 (C. spicifera); E) Front side of the culture; F) Reverse side of the culture; G and H) Conidiophores and conidia. I-L) Isolate Cv-11 (C. ahvazensis); I) Front side of the culture; J) Reverse side of the culture; K-L) Conidiophores and conidia; L) Conidia. M-Q) Representative morphology of isolates Cv-19, Cv-21, Cv-29 (C. hawaiensis); M) Front side of the culture; N) Reverse side of the culture; O-P-Q) Conidiophore and conidia. Scale bar=20 µm.
Table 2 Morphometry of fungal conidia associated with leaf spot in soybean var. Nainari.
| Especie/Aislado | Longitud (µm) | Ancho (µm) | Relación (L/A) (µm) | Septos/Distoseptas |
|---|---|---|---|---|
| C. muehlenbeckiae Cv-3 | 15.0-27.5x | 7.5-12.5 | 2.0 | 2.0-4.0 |
| 21.75y | 11.25 | 3.6 | ||
| 1.0z | 0.6 | 0.7 | ||
| C. spicifera Cv-4 | 10.0-22.5 | 7.5-10.0 | 2.1 | 3.0-4.0 |
| 18.75 | 9.0 | 3.9 | ||
| 1.0 | 0.5 | 0.4 | ||
| C muehlenbeckiae Cv-5 | 20.0-27.5 | 10.0-15.0 | 1.9 | 2.0-4.0 |
| 23.25 | 12.0 | 3.6 | ||
| 0.9 | 0.5 | 0.7 | ||
| C. muehlenbeckia Cv-8 | 17.5-25.0 | 7.5-15.0 | 2.0 | 3.0-4.0 |
| 22.0 | 11.0 | 3.6 | ||
| 0.7 | 0.6 | 0.5 | ||
| C. ahvazensis Cv-11 | 16.2-27.5 | 6.2-10.0 | 2.9 | 2.0-4.0 |
| 22.3 | 7.6 | 4.2 | ||
| 2.7 | 0.5 | 1.1 | ||
| Curvularia sp. Cv-19 | 10.0-22.5 | 7.5-10.0 | 2.1 | 3.0-4.0 |
| 18.75 | 9.0 | 3.9 | ||
| 1.0 | 0.5 | 0.4 | ||
| Curvularia sp. Cv-21 | 10.0-35.0 | 5.0-7.5 | 3.7 | 2.0-5.0 |
| 20 | 5.5 | 3.8 | ||
| 1.5 | 0.4 | 0.6 | ||
| Curvularia Cv-29 | 17.5-27.5 | 7.5-12.5 | 2.4 | 2.0-5.00 |
| 21.75 | 9.0 | 3.8 | ||
| 1.1 | 4.0 | 0.7 |
x The first row represents the maximum and minimum length of the structures; ythe second row indicates the mean of the 40 structures in each sample; zthe third row refers to the standard deviation of the data.
Molecular identification. The partial sequences of the gapdh gene were deposited in the GenBank database (accession MT680146-MT680150 and MT680152-MT680154). The analyses confirmed the identity of the isolates at a genus level. In turn, the comparison of the sequences in the GenBank database displayed high percentages of similarity with diverse Curvularia species; isolates Cv-3, Cv-5 and Cv-8 were similar to C. muehlenbeckiae strain UTHSC 08-2905 (accession LT715807; 99% identity) and CBS 144.63 (accession LT715806, 100% identity); isolate Cv-4 was similar to C. spicifera strain CBS 198.31 (accession LT715814.1, 99% identity); isolate Cv-11 recorded high percentages of similarity with C. hawaiiensis strain CBS 173.57 (accession LT715812, 99% identity), C. nodosa strain CPC 28812 (accession MF490840, 98.5% identity), C. perotidis strain CBS 350.90 (accession LT715813, 97.9% identity) and C. dactyloctenii strain BRIP 12846 (accession KJ415401, 98.5% identity); whereas isolates Cv-19, Cv-21 and Cv-29 shared high percentages of similarity with C. beasleyi strain BRIP 15854 (accession MH433639, 99.1% identity), C. dactyloctenii strain BRIP 12846 (accession KJ415401, 98.9% identity), C. hawaiiensis strain CBS 173.57 (accession LT715812, 98.7% identity), C. nodosa strain CPC 28812 (accession MF490840, 98.5% identity) and C. perotidis strain CBS 350.90 (accession LT715813, 97.9% identity).
Figure 2 shows the phylogram inferred from the partial sequence of gene gapdh. Isolates Cv-3, Cv-5 and Cv-8 were grouped, along with the C. muehlenbeckiae type sequence, with a high bootstrap support (90.9%), which coincides with the morphological description of these isolates for the species at hand (Figure 1A-D). Isolate Cv-4 was identified as C. spicifera, since it grouped with the type sequence of this species (bootstrap 94.1%) and its morphological characteristics coincide with those reported previously. Isolate Cv-11 is grouped with C. ahvazensis (52.1% bootstrap) and its morphological characteristics coincide with those reported for the species. The phylogram shows that isolates Cv-19, Cv-21 and Cv-29 are different and do not correspond to any of the species of Curvularia used as references in the analysis, and were therefore considered as Curvularia sp. The three isolates are phylogenetically near to C. beasleyi, the species with which they share 99 - 99.2% similarities.
This group of fungi has a high morphological complexity and diversity of cryptic species; therefore, its taxonomy has not been entirely solved (Manamgoda et al., 2015). Currently, the genus covers just over 100 described species, out of which only ~80 have been accepted and their taxonomic locations have been established through multigene phylogenetic analyses (Manamgoda et al., 2015; Tan et al., 2018; Kiss et al., 2019). However, the gene gapdh on its own can solve the Curvularia species (Manamgoda et al., 2015), and it is the locus with the most phylogenetic information among the loci analyzed for this fungal genus (Ferdinandez et al., 2019). For isolates Cv-19, Cv-21 and Cv-29, the identities of which could not be determined, we recommend carrying out a more detailed molecular characterization to corroborate them as a new lineage within the genus and give them a name. In order to do this, the sequencing of the ITS region and/or the translation elongation factor (TEF) gene must be included, as it has been done previously in the registration of new species of Curvularia (Heidari et al., 2018; Mehrabi-Koushki et al., 2018).
Pathogenicity test. All the isolates obtained from symptomatic soybean leaflets were pathogenic in soybean var. Nainari and caused similar symptoms to those observed in the field (Figure 3). The first symptoms were found in the basal third and middle of the plants six days after inoculation, while defoliation began 15 days after inoculation; in turn, the symptoms caused by the different Curvularia species were similar, but with variations in the degree of virulence. In experiment 1, 30 days after inoculation, the plants inoculated with the C. ahvazensis (Cv-11) and Curvularia sp. (Cv-21) isolates presented an LAA of 30%, without differences (F=73.3; P<0.0001) between these species, although there were differences with isolates Cv-3 and Cv-8 from C. muehlenbeckiae and isolates Cv-19 and Cv-29 of Curvularia sp., which caused an AFA of 10%. The isolates of C. spicifera (Cv-4) and C. muehlenbeckiae (Cv-3 and Cv-5) were the least virulent, since they caused an AFA of 3 to 9% (Table 3). In experiment 2, the percentages of AFA caused by isolates of C. ahvazensis (Cv-11) and Curvularia sp. (Cv-21) were 24.0 and 19.0%, respectively, with differences (F=27.0; P<0.0001) between these isolates, and the rest belonged to diverse species of Curvularia, which caused between 5.0 and 15.0% of LAA. In both experiments, the control plants sprayed with distilled water remained asymptomatic throughout the investigation (Figure 1B).
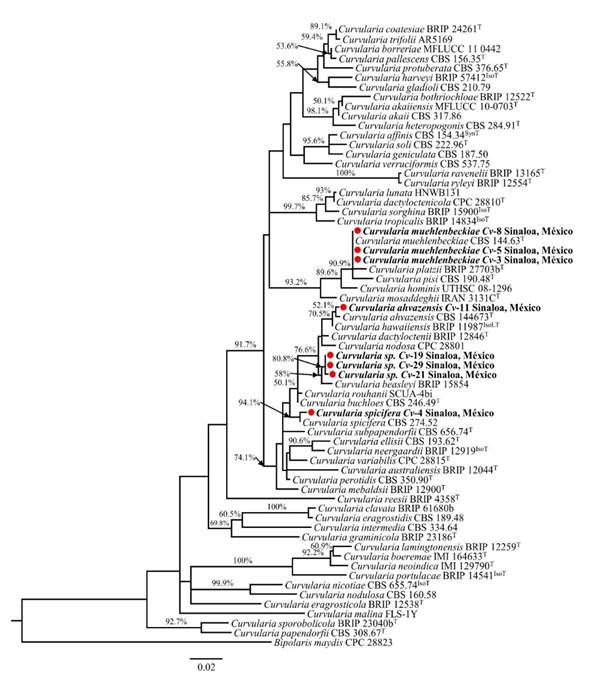
Figure 2 Phylogenetic tree of the glyceraldehyde-3-phosphate dehydrogenase (gapdh) gene, inferred by maximum likelihood (lnL -4286.90) in MEGA X. The scale represents the average of situations per site. Bootstrap values ≥70% are presented on the branches, near the internodes. The species found in this study are shown in bold, followed by a red circle. ATCC, American Type Culture Collection, Virginia, USA; BRIP, Queensland Plant Pathology Herbarium, Brisbane, Australia; CBS, Westerdijk Fungal Biodiversity Institute, Utrecht, the Netherlands; MFLUCC, Mae Fah Luang University Culture Collection, Chian Ria, Thailand; UTHSC, Fungus Testing Laboratory, Department of Pathology of the University of Texas Health Science Center, San Antonio, Texas, USA; CPC, Culture Collection of Pedro Crous, Westerdijk Fungal Biodiversity Institute; IMI, International Mycological Institute, CABI-Bioscience, Egham, Bakeham Lane, UK. ET , IsoT , IsoLT , SynT and T indicate ex-epitype, ex-isotype, ex-isolectotype, ex-syntype and ex-type strains, respectively.
To comply with Koch’s standards, the inoculated Curvularia species were re-isolated from the symptomatic plants and their identity was confirmed by comparing the phenotypical characteristics of the cultures and morphometry of 20 conidia with the original isolates.
The species of Curvularia mentioned previously were proven to cause leaf spot in soybean plants, which contrasts with reports in Sinaloa in which Corynespora casiicola, Cercospora sojina and Cercospora kikushii are mentioned as causal agents of the disease (Rodríguez-Cota et al., 2019), as well as in Brazil (Teramoto et al., 2017), the United States (Sumabat et al., 2018) and Argentina (Reznikov et al., 2018). Although the symptoms caused by the Curvularia species in our study are similar to those caused by other fungi (Stewart and Rodríguez, 2016; Mwase and Kapooria, 2000), these were not found in the sampling period of our investigation. Fungal foliar diseases in soybean that occur in the stage of grain filling are called late-season diseases (LSD), since they take place in plants in intermediate to advanced crop stages of maturity (De Lisi et al., 2015). Given that foliar spot in soybean caused by C. spicifera, C. muehlenbeckiae, C. ahavenzis and Curvularia spp. in Sinaloa appears in the grain-filling stage, it must also be considered an LSD. This disease appeared in September and October, when rainfalls varied between 321 and 373 mm in the region and the temperature fluctuated between 26 and 39 °C (Junta Local del Valle del Carrizo weather station, Sinaloa, 2019). In other soybean-producing regions, Colletotrichum truncatum, Septoria glycines, Phomopsis sojae, Peronospora manshurica and Alternaria spp. have been registered as LSD causal agents (Carmona et al., 2015), yet these pathogens were not found at the time the present investigation was being carried out. Studies in Argentina indicate that LSD reduce crop yield by 8 to 10%, and a maximum of up to 30% (Carmona et al., 2004); in this sense, the investigations carried out in Sinaloa to determine the impact of the disease in crop yield and quality are justifiable, in the same way the implantation of strategies for their control would be, if needed.

Figure 3 A) Symptoms of foliar spot in leaflets of soybean plants from natural infections in the field; B) Non-inoculated control plant leaflets; C) Leaflets with symptoms, typical of leaf spot in soybean plants, caused by species of Curvularia (isolate Cv-11) in greenhouse inoculations.
Table 3. Percentage of foliar area affected in soybean plants, variety Nainari, inoculated with different species of Curvularia.
| Especie/Aisladox | Experimento 1 | Experimento 2 |
|---|---|---|
| C. ahvazensis Cv-11 | 30.0 az | 24.0 az |
| Curvularia sp. Cv-21 | 30.0 a | 19.0 ab |
| C. muehlenbeckiae Cv-8 | 21.0 b | 15.0 bc |
| Curvularia sp. Cv-19 | 10.0 c | 8.0 d |
| Curvularia sp. Cv-29 | 10.0 c | 11.0 cd |
| C. muehlenbeckiae Cv-3 | 9.0 c | 9.0 cd |
| C. spicifera Cv-4 | 9.0 c | 10.0 cd |
| C. muehlenbeckiae Cv-5 | 3.0 d | 5.0 de |
| Testigo sin inoculación | 0.0 d | 0.0 e |
| CV | 29.8 | 39.2 |
xThe pathogenicity of the isolates was evaluated 30 days after inoculation.
zMeans with the same letter in each column are not different in Tukey’s test (p≤0.05).
Conclusions
The fungi Curvularia spicifera, C. muehlenbeckiae, C. ahvazensis and Curvularia sp. were implicated in foliar spot in soybean, which began in the stage of grain filling (R6). The species were identified with the analysis of its morphometric characteristics and phylogenetic reconstruction, based on a fragment of the gene gapdh. The Curvularia species were found to be pathogenic in soybean var. Nainari and they displayed differen degrees of virulence. This is the first report in Mexico involving species of Curvularia in the etiology of leaf spot in soybean.
Acknowledgements
We would like to thank the partial funding of the present study by PFECE-SES-SEP, 2017.
REFERENCES
Ayoubi N, Javad M, Zare R and Zafari D. 2017. First report of Curvularia inaequalis and C. specifera causing leaf blight and fruit rot of strawberry in Iran. Nova Hedwigia 105(1-2): 75-85. https://doi.org/10.1127/nova_hedwigia/2017/0402 [ Links ]
Bandara AY, Weerasooriya DK, Bradley CA, Allen TW and Esker PD. 2020. Dissecting the economic impact of soybean diseases in the United States over two decades. PLoS ONE 15: e0231141. https://doi.org/10.1371/journal.pone.0231141 [ Links ]
Berbee M, Pirseyedi M and Hubbard S. 1999.Cochliobolusphylogenetics and the origin of known, highly virulent pathogens, inferred from ITS and glyceraldehyde-3-phosphate dehydrogenase gene sequences. Mycologia 91(6): 964-977. https://doi.org/10.2307/3761627 [ Links ]
Carmona M, Sautua F, Perelman S, Gally M and Reis EM. 2015. Development and validation of a fungicide scoring system for management of late season soybean diseases in Argentina. Crop Protection 70: 83-91. https://doi.org/10.1016/j.cropro.2015.01.019 [ Links ]
Carmona M, Moscchini R, Cazenave G y Sautua F. 2010. Relación entre la precipitación registrada en estados productivos de la soja y la severidad de Septoria glycines y Cercospora kikushii. Tropical Plant Pathology 35(2): 71-78. http://dx.doi.org/10.1590/S1982-56762010000200001 [ Links ]
Carmona M, Gally M, Grijalba P, Sugia V and Jaeggi E. 2004. Frequency and chemical control of causal pathogens of soybean late season diseases in the Pampeana Region. 7th World Soybean Research Conference, 4th International Soybean Processing and Utilization Conference, III Congresso Mundial de Soja, 29 de febrero al 5 de marzo de 2004. Foz de Iguazú, Brasil. 159p. [ Links ]
De Lisi V, Reznikov S, Aguaysol NC, Martínez CV, Claps MP, Bernal ML, Gomez EN, Martinez-Monteros MJ, Villafañe P, Escobar M, Gonzalez V y Ploper LD. 2015. Enfermedades presentes en la campaña de soja 2014/2015 en el NOA. Publicación Especial EEAOC 51: 112-118. http://hdl.handle.net/11336/15007 [ Links ]
Edgar RC. 2004. MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Research 32(5):1792-1797. https://doi.org/10.1093/nar/gkh340 [ Links ]
FAO, Food and Agriculture Organization of the United Nations. 2020. Statistical Yearbook. World Food and Agriculture. 366p. https://doi.org/10.4060/cb1329en. (Consulta junio 2021). [ Links ]
Fehr WR and Caviness CE. 1977. Stages of soybean development. Iowa State University. Special Report 87. 13p. http://lib.dr.iastate.edu/specialreports/87 [ Links ]
Ferdinandez RSKHS, Udayanga D, Deshappriya N, Munasinghe MLAMS and Manamgoda DS. 2019. Species limits in Curvularia: Updated backbone phylogeny and fresh collections from Sri Lanka. International Research Conference of UWU-2019.http://www.erepo.lib.uwu.ac.lk/bitstream/handle/123456789/65/24.pdf?sequence=1&isAllowed=y [ Links ]
Flores-Córdova MA, Martínez-Damián MT, Nieto-Ángel D, Rodríguez-Pérez JE, Colinas-León MT y Martínez-Solís J. 2013. Reducción en la germinación in vitro de conidios de Alternaria alternata aislada de Eruca sativa con jugo de brócoli. Revista Mexicana de Fitopatología 31(2): 180-190. http://www.redalyc.org/articulo.oa?id=61231509009 [ Links ]
Gómez KA and Gómez AA. 1984. Statistical procedures for agricultural research. John Wiley and sons Inc. New York, USA. 680p. [ Links ]
Hall TA. 1999. BioEdit: a user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic acids symposium series 41:95-98. [ Links ]
Heidari K, Mehrabi-Koushki M and Farokhinejad R. 2018. Curvularia mosaddeghii sp. nov., a novel species from the family Pleosporaceae. Mycosphere 9(4): 635-646. https://doi.org/10.5943/mycosphere/9/4/2 [ Links ]
Kelly KL and Judd DE. 1976. Color Universal language and dictionary of names. National Bureau of standards (U.S.). Special publication 440. 184p. [ Links ]
Kim YK, Xiao CK and Rogers JD. 2005. Influence of culture media and environmental factors on mycelial growth and pycnidial production of Sphareopsis pyriputrescens. Mycologia 97(1): 25-32. https://doi.org/10.3852/mycologia.97.1.25 [ Links ]
Kiss N, Homa M, Manikandan P, Mythili A, Krizsán K, Revathi R., Varga M, Papp T, Vágvölgyic C, Kredics L and Kocsubé S. 2019. New species of the genus Curvularia: C. tamilnaduensis and C. coimbatorensis from fungal keratitis cases in South India. Pathogens 9(1):9. https://doi.org/10.3390/pathogens9010009. [ Links ]
Kumar S, Stecher G, Li M, Knyaz C and Tamura K. 2018. MEGA X: Molecular Evolutionary Genetics Analysis across Computing Platforms. Molecular Biology Evolution 35(6): 1547-1549. https://doi.org/10.1093/molbev/msy096 [ Links ]
Little TM and Hills FJ. 1973. Agricultural Experimentation and Analysis. John Wiley and Sons. New York, USA. 350p. [ Links ]
Madrid H, Da Cunha KC, Gené J, Dijksterhuis J, Cano J, Sutton DA, Guarro J and Crous PW. 2014. Novel Curvularia species from clinical specimens. Persoonia 33(1): 48-60. https://doi.org/10.3767/003158514X683538 [ Links ]
Manamgoda DS, Rossman AY and Castlebury LA. 2015. A taxonomic and phylogenetic reappraisal of the genusCurvularia(Pleosporaceae): human and plant pathogens. Phytotaxa 212(3): 175-198. https://doi.org/10.11646/phytotaxa.212.3.1 [ Links ]
Maraite M, Zinno TD, Longree H, Daumerie V and Duveiller E. 1997. Fungi associated with foliar blight of wheat in warm areas. In: Duveiller E, Duvin HJ, Reeves J, and MacNab A (eds). Helminthosporium blight of wheat: Spot blotch and tan spot. CIMMYT. 293-300p. [ Links ]
Marin-Felix Y, Hernández-Restrepo M and Crous PW. 2020. Multi-locus phylogeny of the genus Curvularia and description of ten new species. Mycological Progress 19(6):559-588. https://doi.org/10.1007/s11557-020-01576-6 [ Links ]
Mehrabi-Koushki M, Pooladi P, Eisvand P and Babaahmadi G. 2018. Curvularia ahvazensis and C. rouhanii spp. nov. from Iran. Mycosphere 9(6):1173-1186. https://doi.org/10.5943/mycosphere/9/6/7 [ Links ]
Mwase WF and Kapooria RG. 2000. Incidence and severity of frogeye leaf spot and associated yield losses in soybeans in agroecological zone II of Zambia. Mycopathologia 149(2):73-78. https://doi.org/10.1023/A:1007126225457 [ Links ]
Prieto KR, de Medeiros LS, Isidoro MM, Toffano L, da Silva MFGF, Fernandes JB, Vieira PC, Forim MR, Rodrigues-Filho E, Stuartb RM and Machadob MA. 2016. Rapid determination of ACTG-and AK-toxins in Alternaria alternata by LC-ESI-MS/MS Analysis and antifungal properties of citrus compounds. Journal of the Brazilian Chemical Society 27(8):1493-1505. https://doi.org/10.5935/0103-5053.20160195 [ Links ]
Qostal S, Kribel S, Chliyeh M, Selmaoui K, Ouazzani A, Serghat S, Zaarati H, Benkirane R and Douira A. 2019. Curvularia spicifera, a parasite of the fungal complex of root rot of wheat and barley in Morocco. Plant Cell Biotechnology and Molecular Biology 20(9-10):354-365. https://www.ikprress.org/index.php/PCBMB/article/view/4627 [ Links ]
Rambaut A. 2014. FigTree v1.4.2, A Graphical Viewer of Phylogenetic Trees. Available from <http://tree.bio.ed.ac.uk/software/ figtree/>. [ Links ]
Reznikov S, de Lisi V, Escobar M, Claps MP, Bleckwedel J, Gutierrez H, Daniel F, Gonzalez V, Ledesma F, Devani MR and Ploper LD. 2018. Panorama sanitario del cultivo de la soja en el noroeste argentino durante la campaña 2017/2018. Estación Experimental Agroindustrial Obispo Colombres; Soja en el NOA; 54(9):1-5 http://hdl.handle.net/11336/95779 [ Links ]
Rodríguez-Cota FG, Cortez-Mondaca E, Sauceda-Cota RH, Valenzuela-Herrera V y Macías-Cervantes J. 2019. Tecnología para producir soya en Sinaloa. Campo Experimental Valle del Fuerte, INIFAP, Folleto Técnico No. 45. Juan José Ríos, Sin., México. 48p. [ Links ]
SAS, Institute Inc. 1999. SAS user’s guide: Statistics. Release 6.03. Ed. SAS Institute Incorporation. Cary, NC USA. 1028 p. [ Links ]
SIAP, Servicio de Información Agroalimentaria y Pesquera. 2020. Cierre de la Producción Agrícola por Cultivo “Modalidad riego + temporal, Primavera-verano”. SAGARPA, D.F. México. http://infosiap.siap.gob.mx:8080/agricola_siap_gobmx/ResumenDelegacion.do [ Links ]
SIAP, Servicio de Información Agroalimentaria y Pesquera. 2020. Panorama Agroalimentario 2020. Ciudad de México. 200p. https://www.inforural.com.mx/wp-content/uploads/2020/11/Atlas-Agroalimentario-2020.pdf [ Links ]
Soares RM, Godoy CV y Oliveira MCN. 2009. Diagrammatic scale for severity evaluation of soybean target spot. Tropical Plant Pathology 34(5): 333-338. https://doi.org/10.1590/S1982-56762009000500007 [ Links ]
Stewart S y Rodríguez M. 2016. Manual de identificación de enfermedades de la soja. 2a edición ampliada y corregida Instituto Nacional de Investigación Agropecuaria, Uruguay. Boletín de Divulgación No 4. 74p. [ Links ]
Sumabat LG, Kemerait RC, Kim DK, Mehta YR and Brewer MT. 2018. Clonality and geographic structure of host-specialized populations ofCorynespora cassiicolacausing emerging target spot epidemics in the southeastern United States. PLoS ONE 13: e0205849. https://doi.org/10.1371/journal.pone.0205849. [ Links ]
Tan YP, Crous PW and Shivas RG. 2018. Cryptic species ofCurvulariain the culture collection of the Queensland Plant Pathology Herbarium. MycoKeys 35: 1-25. https://doi.org/10.3897/mycokeys.35.25665 [ Links ]
Teramoto A, Meyer MC, Suassuna ND and Cunha MG. 2017. In vitro sensitivity of Corynespora cassiicola isolated from soybean to fungicides and field chemical control of target spot. Summa Phytopathologica 43(4): 281-289. https://doi.org/10.1590/0100-5405/2195 [ Links ]
Tomaso-Peterson M, Jo YK, Vines PL and Hoffmann FG. 2016. Curvularia malina sp. nov. incites a new disease of warm-season turfgrasses in the southeastern United States. Mycologia 108(5): 915-924. https://doi.org/10.3852/15-238 [ Links ]
Received: May 11, 2021; Accepted: July 16, 2021











 texto en
texto en 


