Introduction
Tuberculosis (TB), a disease caused by Mycobacterium tuberculosis, is a global health problem1. Up to one-third of the global population may be infected with M. tuberculosis in an asymptomatic form latent TB infection, and 5-10% of these people will eventually develop active TB, which is mainly a lung disease. In Mexico, there were 21,184 new cases of active TB in 2016, with a prevalence of 17.3 cases/100,000 people and a mortality rate of 1.7/100,000 people2. The WHO reported a total incidence of 29,000 (22,000-37,000) cases in 2018, with 1900 deaths3. Innate and adaptive immune cells control the proliferation and dissemination of M. tuberculosis through the formation of granulomas4. Antibodies also participate in the control of this infection: B-cell deficiency is associated with higher mycobacterial burdens and tissue injury5,6 and antibodies against M. tuberculosis antigens reduce M. tuberculosis dissemination7, decrease the lung mycobacterial burden8, and increase host survival9.
Control of TB incidence would be greatly aided by an effective vaccine, by a short-term drug TB treatment, and by the availability of a sensitive, fast, and inexpensive point-of-care TB diagnostic test, which would allow the immediate initiation of antibiotic therapy10. Active pulmonary TB is currently diagnosed by baciloscopy, which has low sensitivity; by mycobacterial culture, which has higher sensitivity but can only be performed in specialized laboratories; and by molecular biology methods, which are highly sensitive and specific, but require expensive equipment and training10. Serological diagnostic methods are attractive because they are fast, easy to perform, and do not require sophisticated equipment. Several M. tuberculosis antigens have been suggested as possible serodiagnostic targets; in most cases, the proposed serological diagnostic test identifies antibodies against mycobacterial antigens. However, methods targeting antibodies depend on correct immune activation and specific antibody production after M. tuberculosis infection, while methods directly targeting M. tuberculosis antigens are useful as soon as a person is infected11. In this study, we used a Western blot method to detect mycobacterial antigens in the sera of mice infected with M. tuberculosis and in patients with active pulmonary TB; these antigens could be applied to serological methods for TB diagnosis.
Materials and methods
M. tuberculosis and Mycobacterium bovis strains
M. tuberculosis H37Rv (ATCC 27294), a virulent strain susceptible to isoniazid, rifampicin, and pyrazinamide, and M. tuberculosis CIBIN99, a clinical isolate resistant to isoniazid, rifampicin, and pyrazinamide (multidrug-resistant [MDR]), were cultured in 7H9 medium at 37°C (BD Difco, Becton, Dickinson and Company, Franklin Lakes, NJ) to logarithmic growth and adjusted to 2.5 × 105 colony-forming unit (CFU) per 100 μl of phosphate-buffered saline (PBS). M. bovis 534, an avirulent strain, and M. bovis AN5, a virulent strain, were cultured in 7H9 medium enriched with glycerol, albumin, catalase, and dextrose at 37°C to logarithmic growth and adjusted to 2.5 × 105 CFU per 100 μl of PBS.
Laboratory animals
Rabbits and BALB/c mice were housed and manipulated according to the Guide for the Care and Use of Laboratory Animals (National Academy of Sciences, 2011). All our protocols for animal care and use were reviewed and approved by the Bioethics Committee of ENCB-IPN (CEI-ENCB 007/2013).
Infection of BALB/c mice with mycobacteria
Male, 6-8-week-old BALB/c mice were infected by intratracheal administration of 2.5 × 105 CFU of M. tuberculosis H37Rv, M. tuberculosis CIBIN99, M. bovis 534, or M. bovis AN5, and serum samples were collected on the indicated days post-infection. Sera were filtered through a 0.45 μm pore size membrane and stored at −20°C. In some experiments, infected mice were treated with isoniazid (10 mg/kg), rifampicin (10 mg/kg), and pyrazinamide (30 mg/kg); antibiotic treatment was started on day 60 post-infection and was administered daily.
Patient selection criteria
This study was approved by the Ethics and Research Committee of the General Hospital of Mexico (DIC/07/406/03/007). We obtained serum samples from patients (18 to 60 years old) with a first episode of active pulmonary TB; these patients had not been previously treated for TB. Active pulmonary TB was diagnosed according to the clinical presentation of the disease, the thoracic X-ray image, and the baciloscopy. Patients with pregnancy, recent (< 4 weeks) major surgery, immunosuppressive treatment, immunodeficiency, HIV infection, cancer, diabetes mellitus, or autoimmune disease were not included in this study. A group of healthy volunteers was also included in this study. All the individuals gave written informed consent to participate in this study, in accordance with the Declaration of Helsinki.
Preparation of M. tuberculosis H37Rv extracts
M. tuberculosis H37Rv was cultured in Proskauer-Beck-Youmans medium at 37°C for 4 weeks. Bacteria were resuspended in saline solution with protease inhibitors (Complete Mini Protease Inhibitor Cocktail, Roche, Mannheim, Germany). To obtain M. tuberculosis total extract (MTTE), bacteria were subjected to six lysing cycles (2500 psi each) in a French press (Sim-Aminco, Spectronic Instruments, Rochester, NY). To obtain M. tuberculosis soluble extract (MTSE), the MTTE was centrifuged twice at 9000 g for 10 min and 4°C, and the supernatant was recovered. Protein concentration in the extracts was determined with the Bradford method (Bio-Rad Laboratories, Hercules, CA). The extracts were stored at −20°C.
Preparation of a rabbit serum with antibodies against anti-MTTE
A 2-month-old male rabbit was immunized by subcutaneous injection of 2.5 mg of MTTE, which was mixed 1:2 with incomplete Freund's adjuvant (Sigma-Aldrich, St. Louis, MO). After 2 weeks, another subcutaneous injection of 2.5 mg of MTTE with incomplete Freund's adjuvant was administered and, after 1 more week, the rabbit was anesthetized (ketamine, 100 mg), bled by intracardiac puncture and euthanized. The blood was collected in a glass tube and allowed to clot for 3 h at 4°C. Serum was recovered by centrifugation and stored at −20°C. Blood was also collected from a non-immunized rabbit, to use as a specificity control.
Detection of M. tuberculosis antigens in sera by Western blot
MTTE or MTSE (16 mg/well), filtered mice sera (87.5 mg/well), or filtered human sera (87.5 mg/well) were mixed with a buffer (Tris-HCl 1 M pH 8.4, sucrose 0.5 M, EDTA 0.5 M, bromophenol blue 0.03%, DTT 1%, and SDS 10%), boiled for 5 min, and separated by SDS 3/10% or 3/6% polyacrylamide gel electrophoresis (PAGE). The proteins were then transferred to a polyvinylidene fluoride membrane, which was soaked in PBS with 0.05% Tween 20 for 20 min at room temperature, and blocked in PBS with 0.05% Tween 20 and 1% skimmed milk for 45 min at 37°C. The rabbit serum with antibodies anti-MTTE was diluted 1:5000 in blocking buffer, added to the membranes, and left overnight at 4°C. The membranes were then washed 10 times with PBS with 0.05% Tween 20 and 5 times with PBS, and the secondary antibody (horseradish peroxidase-conjugated donkey anti-rabbit immunoglobulin G (IgGs), BioLegend, San Diego, CA) diluted 1:2000 in blocking buffer, was added, and left for 1 h at 37. The membranes were revealed with diaminobenzidine and hydrogen peroxide, and photographed in a MultiImage Light Cabinet with AlphaImager software (Alpha Innotech Corporation, San Leandro, CA). The photographs were analyzed with ImageJ Software 1.8.0 (NIH, Bethesda, MD), to determine the area under the curve (AUC) of the graph that represents each band. Statistical analyses were performed with Prism 6.0 (GraphPad Software, La Jolla, CA).
To determine the optimal dilution of the rabbit serum with antibodies anti-MTTE, several dilutions were tested (1:2000, 1:3000, 1:5000, 1:10,000, 1:15,000, 1:20,000, and 1:30,000); the 1:5000 dilution was selected because it allows a sharper separation of bands in MTTE.
Results
BALB/c mice were infected intratracheally with M. tuberculosis H37Rv; sera were collected on days 1, 3, 7, 14, 21, and 28 post-infection and analyzed by Western blot, with anti-MTTE rabbit serum as the primary antibody. Mycobacterial antigens were detected in the sera of these mice for the 1st day post-infection, with molecular masses of 112, 50, 45, 29-30 (two bands), and < 15 kDa (Fig. 1A). Since the 112 kDa antigen band is located in the top fourth of the 3/10% polyacrylamide gel, we performed 3/6% SDS-PAGE to determine the molecular mass of this antigen with higher accuracy (Fig. 2A); this antigen is hereafter referred to as the 97 kDa antigen. A decrease in antigen concentration on days 14 and 21 post-infection (when granulomas are formed in this mouse model of pulmonary TB)12 was noted in some mice (Fig. 1A), but overall, the antigen concentration remained constant up to day 28 post-infection (Fig. 1C). Decreased antigen concentration on days 14 and 21 post-infection could be caused by the capture of M. tuberculosis antigens inside granulomas; after day 28, a predominant Th2 response decreases the integrity of the granuloma12 and correlates with increased circulating antigens at this time.

Figure 1 Mice infected with Mycobacterium tuberculosis H37Rv have mycobacterial antigens in their sera. Filtered sera from mice infected with M. tuberculosis H37Rv were separated by 3/10% SDS-polyacrylamide gel electrophoresis and then transferred to polyvinylidene fluoride membranes, which were analyzed by Western blot using an anti-M. tuberculosis total extract rabbit serum as primary antibody. A-C: early infection (days 0-28 post-infection). The graph in C represents the area under the curve (AUC) of 97 kDa antigen bands from 5 to 7 mice/day. B-D: progressive infection (days 60-180 post-infection). The graph in D represents the AUC of 97 kDa antigen bands from 2 to 7 mice/day. Data were analyzed with Kruskal-Wallis test followed by Dunn's multiple comparisons test (*p > 0.05). M, molecular mass standard. MTSE, M. tuberculosis soluble extract.
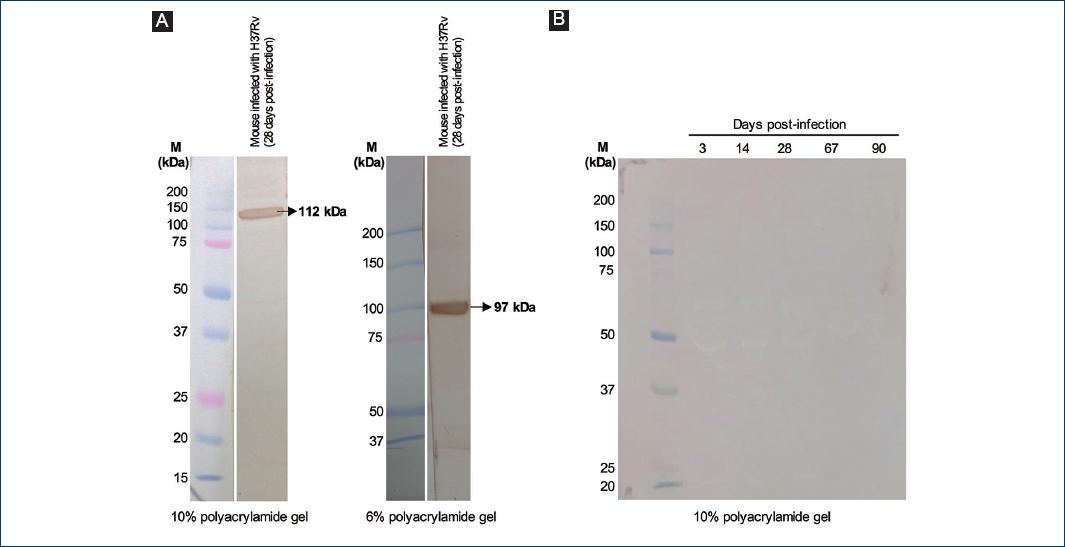
Figure 2 Controls for the Western blot detection of Mycobacterium tuberculosis antigens. A: left panel: filtered sera from a mouse infected with M. tuberculosis H37Rv (28 days post-infection) were separated by 3/10% SDS-polyacrylamide gel electrophoresis (PAGE) and then transferred to a polyvinylidene fluoride (PVDF) membrane, which was analyzed by Western blot using an anti-M. tuberculosis total extract rabbit serum as primary antibody. Right panel: filtered sera from a mouse infected with M. tuberculosis H37Rv (the same sera that were used in the left panel and in Fig. 4) were separated by 3/6% SDS-PAGE and then transferred to a PVDF membrane, which was analyzed by Western blot using an anti-M. tuberculosis total extract rabbit serum as primary antibody. B: filtered sera from mice infected with M. tuberculosis H37Rv were separated by 3/10% SDS-PAGE and then transferred to a PVDF membrane, which was analyzed by Western blot using the serum of a non-immunized rabbit as primary antibody.
Circulating mycobacterial antigens were also found during progressive mycobacterial infection (days 60-180 post-infection) and, on day 120, in addition to the antigens of the molecular masses indicated above, a 22 kDa antigen was also present in the sera (Fig. 1B and D). There were no significant differences in the antigens detected in unfiltered sera or in sera filtered through a 0.45 μm pore size membrane, so the detected antigens were not part of whole circulating mycobacteria (data not shown). No antigens were detected when the Western blot was performed with the serum of a non-immunized rabbit as the primary antibody (Fig. 2B), which confirms that this method specifically detects mycobacterial antigens.
In days 67-180 post-infection, mice infected with M. tuberculosis H37Rv and treated with isoniazid, rifampicin, and pyrazinamide showed a marked decrease in circulating mycobacterial antigens (Fig. 3A and C). In contrast, mice infected with MDR M. tuberculosis CIBIN99 showed no decrease in the concentration of circulating mycobacterial antigens (Fig. 3B and D); the AUC of the 97 kDa antigen in the sera of mice infected with M. tuberculosis CIBIN99 was significantly higher than the AUC of the 97 kDa antigen in the sera of mice infected with M. tuberculosis H37Rv on day 74 post-infection (Fig. 3C and D).

Figure 3 Treatment with isoniazid, rifampicin, and pyrazinamide decreases circulating mycobacterial antigens. A-C: filtered sera from mice infected with antibiotic-susceptible Mycobacterium tuberculosis H37Rv; B-D: filtered with multidrug-resistant M. tuberculosis CIBIN99, were separated by 3/10% SDS-polyacrylamide gel electrophoresis and then transferred to polyvinylidene fluoride (PVDF) membranes, which were analyzed by Western blot using an anti-M. tuberculosis total extract rabbit serum as primary antibody. Antibiotic treatment (with isoniazid, rifampicin, and pyrazinamide) was started on day 60 post-infection (vertical arrow) and was administered daily. C-D: the area under the curve (AUC) of 97 kDa antigen bands from 2 to 8 mice/day. Data were analyzed with Kruskal-Wallis test followed by Dunn's multiple comparisons test (*p > 0.05). Data were also analyzed with Mann-Whitney U-test, to compare the 97 kDa antigen AUC in mice infected with H37Rv with the corresponding AUC in mice infected with CIBIN99 (&, p < 0.05 between the indicated groups). M, molecular mass standard.
To evaluate if intratracheal infection with other mycobacteria of M. tuberculosis complex also led to the presence of mycobacterial antigens in circulation, we analyzed the sera of mice infected with M. bovis AN5 strain (virulent) or 534 strain (avirulent). Circulating mycobacterial antigens, including a prominent band of 97 kDa, were found on days 14 and 21 post-infection in the sera of mice infected with the AN5 strain (Fig. 4A and C), while the sera of mice infected with the 534 strain contained low amounts of mycobacterial antigens even up to day 120 post-infection, but did not contain the 97 kDa band (Fig. 4B and D).

Figure 4 Mice infected with virulent Mycobacterium bovis AN5, but not with avirulent M. bovis 534, present a 97 kDa antigen in their sera. A-C: filtered sera from mice infected with virulent M. bovis AN5 (days 0-21 post-infection), B-D: filtered with avirulent M. bovis 534 (days 0-120 post-infection), were separated by 3/10% SDS-polyacrylamide gel electrophoresis and then transferred to polyvinylidene fluoride membranes, which were analyzed by Western blot using an anti- Mycobacterium tuberculosis total extract rabbit serum as primary antibody. C-D: the area under the curve (AUC) of 97 kDa antigen bands from 2 to 4 mice/day. Data were analyzed with Kruskal-Wallis test followed by Dunn's multiple comparisons test (*p > 0.05). AUC, area under the curve. M, molecular mass standard. MTSE, M. tuberculosis soluble extract.
To determine if the 97 kDa antigen could potentially be used as a diagnostic marker for TB, we analyzed the sera of 19 TB patients with positive baciloscopy and 12 healthy volunteers. The sera of the TB patients contained an antigen of 97 kDa, which is in the same position as the antigen found in the sera of mice infected with M. tuberculosis H37Rv (Fig. 5A). The sera of TB patients contained significantly higher concentrations of the 97 kDa antigen than the sera of healthy volunteers (Fig. 5A and B); the area under the receiver-operator characteristic curve is 0.75 (0.58-0.92, p > 0.019) and, for an AUC cutoff value of 2336, the sensitivity is 68% (44-87%) and the specificity is 67% (35-90%) (Fig. 5C).
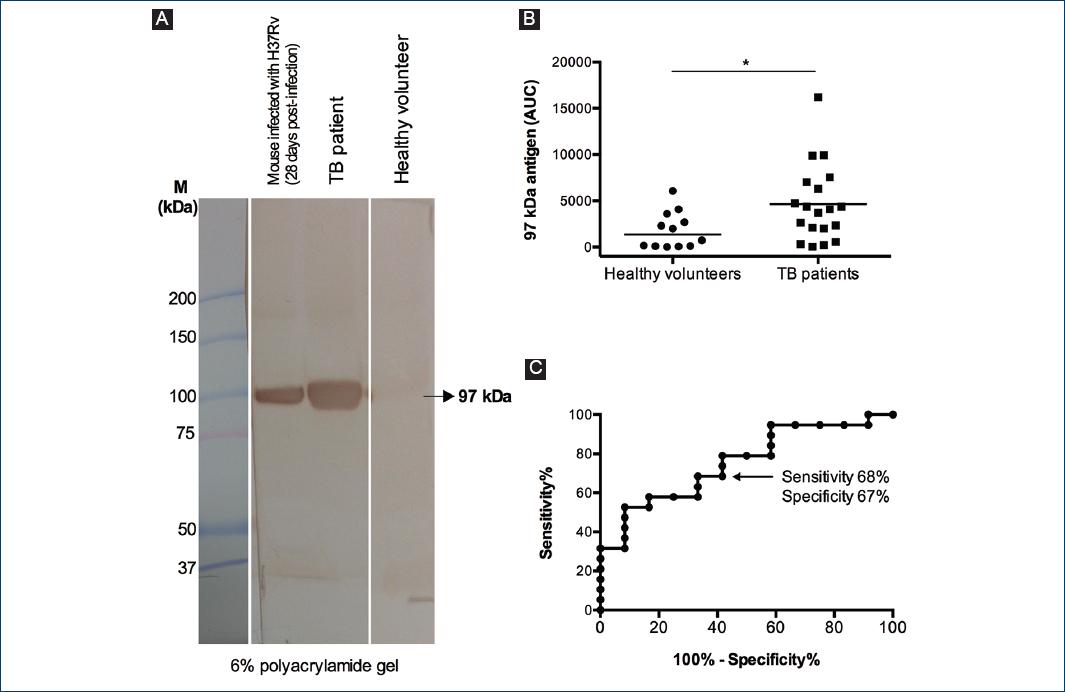
Figure 5 An antigen of 97 kDa is present in the sera of patients with active pulmonary tuberculosis. A: filtered sera from a mouse infected with Mycobacterium tuberculosis H37Rv (28 days post-infection), from a tuberculosis (TB) patient and from a healthy volunteer, were separated by 3/6% SDS-polyacrylamide gel electrophoresis and then transferred to a polyvinylidene fluoride membrane, which was analyzed by Western blot using an anti-M. tuberculosis total extract rabbit serum as primary antibody. B: area under the curve (AUC) of the 97 kDa antigen in the sera of 19 TB patients and 12 healthy volunteers. Data were analyzed with Mann-Whitney U-test (*p < 0.05). C: receiver-operator characteristic curve of the data in C. AUC, area under the curve. M, molecular mass standard.
Discussion
To curb the current TB epidemic10, a sensitive, fast, and inexpensive point-of-care TB diagnostic test is necessary, and serological diagnostic methods fulfill these requirements. To develop these methods, it is necessary to identify M. tuberculosis antigens that are found in individuals with active TB, but not in individuals successfully treated with antibiotics or in healthy individuals. To identify such M. tuberculosis antigens, we first analyzed the sera of mice infected with M. tuberculosis using a Western blot method with a rabbit primary antibody that binds to MTTE antigens. We found that infected mice had mycobacterial antigens in their sera from the 1st day after infection, with a predominance of a 97 kDa antigen; antigen concentrations remained constant up to 180 days post-infection.
The mouse model of pulmonary TB that we used in this study is well characterized. Histopathological studies of these mice clearly define two phases of the disease: an acute phase and a progressive phase. The acute phase lasts from days 1 to 28 post-infection and is characterized by the development of an inflammatory infiltrate in the alveolar-capillary interstitium (days 1-7) and by the formation of granulomas (from day 14 onward). The progressive phase, from day 60 onward, is characterized by progressive pneumonia, with proteinaceous deposits in the alveolar space and focal necrosis12. Without treatment, 80-90% of mice die as a consequence of the infection, showing pneumonic areas in 75-80% of the lung12. Antibiotic treatment decreases lung parenchyma damage and increases the survival of mice infected with antibiotic-susceptible M. tuberculosis strains13. We report here for the 1st time that mice health improvement after successful antibiotic treatment is associated with a marked decrease in circulating mycobacterial antigens; moreover, when mice were infected with a MDR M. tuberculosis strain, the progressive disease was associated with persistence of circulating mycobacterial antigens. These results open the possibility of studying these antigens as potential biomarkers for monitoring disease progression and response to antibiotic treatment.
The previous studies have identified M. tuberculosis antigens in the sera of TB patients, including ESAT-6 (Rv3875, 9.9 kDa)14, CFP-10 (Rv3874, 10.8 kDa)14, alpha-crystallin protein (Rv2031c, 16.2 kDa)15,16, MPT32 (Rv1860, 32.7 kDa)17, MPT64 (Rv1980c, 24.9 kDa)18, mycothiol acetyltransferase (Rv0819, 33.6 kDa)19, the antigen 85 complex (Rv3804c, Rv1886c, and Rv0129c; 35.7, 34.6, and 36.8 kDa)20, and heat-shock protein 65 (Rv0440, 56.7 kDa)21. The main antigens that we identified in the sera of infected mice have molecular masses of < 15 kDa (which could correspond to ESAT-6 and/or CFP-10), 29-30 kDa (which could correspond to proteins from the antigen 85 complex)20,22, 45 kDa (which could correspond to trehalose-6-phosphate phosphatase, Rv3372, 41.7 kDa), 50 kDa, and 97 kDa. Shende et al. separated sera from three active TB patients by SDS-PAGE and analyzed them by Western blot with a goat IgG antibody raised against a M. tuberculosis avirulent strain H37Ra sonicate; they found that this antibody binds to circulating mycobacterial antigens of 16, 20, 43, 55, 85, 140, and 170 kDa23. This pattern is similar, although not identical, to the pattern of circulating mycobacterial antigens that we report in this study; the differences could be explained by the different mycobacteria strain used to raise the primary antibodies (H37Ra vs. H37Rv) and by the hosts of these primary antibodies (goat vs. rabbit).
The 97 kDa antigen is a mycobacteria-specific antigen since it is detected by the serum from rabbits immunized with MTTE, but not by the serum from a non-immunized rabbit. We propose the 97 kDa antigen as a virulence-specific antigen related to M. tuberculosis complex infection since this protein is detected in the sera of mice infected with either M. tuberculosis H37Rv or a virulent M. bovis strain, but not in the sera of mice infected with an avirulent M. bovis strain. Moreover, the 97 kDa antigen is increased in the sera of patients with active pulmonary TB, compared to healthy volunteers. The 97 kDa was present in the sera of some healthy volunteers, which may reflect a latent or a subclinical infection with M. tuberculosis, but the detection of the 97 kDa antigen still had a sensitivity of 68% and a specificity of 67% for the identification of active TB. M. tuberculosis H37Rv (ATCC 25618) proteome24 reports four proteins with a molecular mass (as calculated from the full precursor canonical sequence) close to 97 kDa: DNA polymerase I (Rv1629, 98.5 kDa), PPE family protein PPE35 (Rv1918c, 97.9 kDa), alanine-tRNA ligase (Rv2555c, 97.4 kDa), and replicative DNA helicase (Rv0058, 96.9 kDa). Sequencing of the 97 kDa antigen is required to confirm its identity.
The presence of intracellular M. tuberculosis antigens in the blood of infected individuals could be explained by whole mycobacteria that reach the bloodstream while disseminating from granulomas25. We observed no significant differences in the antigens detected in unfiltered sera or in sera filtered through a 0.45 μm pore size membrane, so the antigens that we detected were not part of whole mycobacteria. In this mouse model of TB, live M. tuberculosis can be found in the circulation from the 1st day post-infection, and antigens released from these mycobacteria, through the mycobacteria secretion system26 or from vesicles released by M. tuberculosis27, could explain why mycobacterial antigens were detected in serum for day 1 post-infection. Circulating mycobacterial antigens could also come from dead mycobacteria released from necrotic immune cells or from extracellular vesicles released from infected immune cells28-30.
Circulating mycobacterial antigens have been proposed as targets for serological diagnostic methods for TB14,31,32. Our study demonstrates a strategy that can be used to identify additional targets for these serological diagnostic methods; in particular, we report a 97 kDa antigen that identifies patients with active pulmonary TB with 68% sensitivity and 67% specificity. The sensitivity and specificity could be increased using a combination of antigens or by analyzing the whole pattern of circulating mycobacterial antigens that are present in the patient sera.
Conclusion
We report a method that can be used to detect circulating mycobacterial antigens and identified a novel 97 kDa antigen in the sera of mice infected with M. tuberculosis and of patients with active pulmonary TB. The presence of the 97 kDa antigen in mice sera correlated with lung infection with M. tuberculosis H37Rv or with virulent M. bovis, and this antigen decreased after successful antibiotic treatment. The 97 kDa antigen could be used as a target, alone or in combination with other antigens, in serological diagnostic methods for TB.











 nueva página del texto (beta)
nueva página del texto (beta)


