Introduction
Infective endocarditis (IE) is a rare but life-threatening disease worldwide.1,2 In addition to heart failure and systemic embolization, patients with IE also suffer from valvular destruction and valve aneurysms, which lead to increased morbidity and mortality.3
Over the years, the epidemiology of IE has gradually changed; Staphylococcus aureus is now the most common cause of IE in most studies at 26.6%; Viridans group streptococci (VGS) account for 18.7% of all cases, other streptococci account for 17.5%, and enterococci make up 10.5%; together, these organisms account for 80-90% of IE cases.2 Many endocarditis pathogens are still found in the oral cavity and may have been acquired through everyday dental routines or invasive procedures. VGS bacteria have a low level of virulence and are typically found in the oral cavity, upper airways, gastrointestinal tract, and female genitalia.1 Regarding the VSG classification, Streptococci mitis is the most common cause of IE, while Streptococcus gordonii has historically been an uncommon cause of IE.4
The mitral valve aneurysm (MVA) is a rare complication associated with IE of the aortic valve. MVA incidence in the setting of IE has decreased from approximately 3.5% to less than 0.3%.5 Once MVA ruptures and severe mitral regurgitation with hemodynamic instability develops, immediate surgical intervention is required.3,6Streptococcus gordonii is an extremely rare cause of IE complicated with abscesses, fistulas, aneurysms, or valve perforation.6,7 In addition to a literature review, we describe a case of native valve endocarditis complicated with perforated MVA and aortic valve perforation caused by Streptococcus gordonii.
Case presentation
A 60-year-old man presented to our center due to a four-week history of evening predominant fever, diaphoresis, asthenia, adynamia, and involuntary weight loss of 10 kg. Only active smoking for 36 years was relevant in his past medical history; IE-related risk factors were not identified, such as recent dental procedures, invasive procedures, or valve heart disease. Vital signs at admission were unaltered: BP 113/56 mmHg, HR 71 bpm, temperature 36.2 oC, RR 23 rpm, and SaO2 95% at ambient air. An electrocardiogram revealed no abnormalities, although the blood tests showed elevated C-reactive protein level (5 mg/dL) and white blood cell count (11,000 mm3).
During the physical examination, a grade 5 holodiastolic murmur was detected in the aortic area, while the mitral area had a grade 5 holosystolic murmur with radiation to the armpit and aortic arch; no signs of IE vascular phenomena were observed.
Based on the suspicion of IE, we obtained paired blood cultures and, subsequently, started vancomycin 1 g IV bid and ceftriaxone 1 g IV bid. A transesophageal echocardiogram (TEE) revealed an 11 × 5 mm vegetation on the P1 annulus of the mitral valve, a ruptured anterior mitral leaflet aneurysm resulting in severe mitral regurgitation accompanied by turbulent flow within the ruptured aneurysm (Figure 1) as well as a perforated non-coronary cusp of the aortic valve resulting in severe aortic regurgitation with regurgitation jet impinging on the anterior mitral leaflet (Figure 2). The blood cultures were positive for multi-sensitive Streptococcus gordonii, and antibiotics were deescalated to only ceftriaxone. Blood tests revealed a reduction in white blood cell count (8,400 mm3) and a decrease in inflammation markers (C-reactive protein 1 mg/dL, erythrocyte sedimentation rate 5 mm/h).
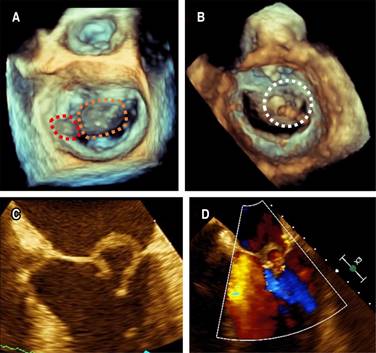
Figure 1: Transesophageal echocardiography: A) 3D view from the left atrium demonstrating an 11 × 5 mm vegetation on the P1 annulus of the mitral valve (red dotted line mark) and an anterior mitral leaflet perforated aneurysm (orange dotted line mark), B) 3D zoom-mode acquisition of mitral valve in mid-diastole from a ventricular perspective with discontinuity of the anterior mitral leaflet at segment A2 (white dotted line mark), C) 2D image at 30o showing an abnormal ring-like structure on anterior mitral leaflet compatible with a perforated aneurysm, D) 2D image with color Doppler showed mitral regurgitation and turbulent flow inside the saccular like image.
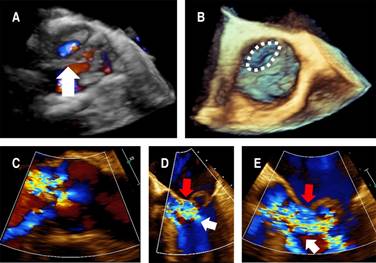
Figure 2: Transesophageal echocardiography: A) 3D image with color Doppler showing flow on the non-coronary cusp of the aortic valve (white arrow), B) 3D image with discontinuity of non-coronary cusp (white dotted line mark), C) 2D image with color Doppler at 125o showing lateral and central jets of aortic regurgitation, D, E) 2D image with Color Doppler at 45o showing the aortic regurgitation jet (white arrow) impinging on the anterior mitral leaflet (red arrow).
The patient underwent surgery after two weeks of antibiotic treatment. Severe damage was observed intraoperatively in the aortic and mitral valves, along with active signs of inflammation; the anterior mitral valve leaflet (AMVL) displayed a perforated aneurysm, while the aortic valve had a perforation on the non-coronary cusp. Mitral and aortic valves were replaced with a 21-mm mechanical prosthesis (St. Jude Medical). The postoperative TEE showed a normal function of the prosthetic valves. Intravenous ceftriaxone was continued, and postoperative blood cultures were negative.
Unfortunately, the patient developed several complications following surgery: hypovolemic shock caused by left internal mammary artery injury, which caused severe postoperative bleeding and necessitated surgical artery repair; complete heart block requiring permanent cardiac pacing; and at day 20 post-surgery, he died of septic shock caused by Klebsiella pneumonia acute mediastinitis.
Discussion
Streptococcus gordonii is a VSG species that colonizes oral biofilms on tooth surfaces and forms dental plaques. During tooth brushing, tooth extraction, or oral trauma, bacteria can be released from oral biofilms and enter the bloodstream, resulting in systemic infection.4Streptococcus gordonii bacteremia contributes to the pathogenesis of IE by inducing platelet aggregation and excessive inflammatory conditions by stimulating various host cells.4,8
As soon as Streptococcus gordonii enters the bloodstream, it attaches to platelets or erythrocytes using their numerous cell surface proteins, and then hematogenously spreads to damaged heart valves. Upon binding to human vascular endothelial cells, it forms biofilms on heart valves, which can further exacerbate the inflammatory response by aggregating platelets into bacterium-platelet-fibrin complexes. In addition, it activates human valve interstitial cells to cause them to release IL-6 and IL-8, which in turn leads to the infiltration of immune cells via the Nuclear Factor-kappa B (NF-κB) signaling pathway. Streptococcus gordonii secretes nitric oxide through the toll-like receptor-2 pathway to activate immune cells in heart lesions recruited from chemokines. When human monocytes are stimulated, they produce proinflammatory cytokines and express more cell surface markers, including clusters of differentiation (CD) 40, CD54, and CD80. Furthermore, it stimulates dendritic cells to produce inflammatory cytokines such as IL-6, IL-12, tumor necrosis factor-α (TNF-α), and co-stimulatory receptors.8
The prevalence of IE in streptococcal bloodstream infections is highly dependent on species, as reported by Chamat-Hedemand et al. in 6,506 cases involving streptococcal bloodstream infections (BSIs). BSI due to S. gordonii showed a very high prevalence (44.2% [95% CI 34-54.8]) and high risk for IE (OR 80.8 [95% CI 43.9-149]), with the highest requirement of cardiac surgery (31%) compared to the most common isolated streptococcal species (S. pneumonia, S. pyogenes). These findings suggest that an echocardiogram should be performed in all patients with streptococcal BSI with a «high» or «very high» risk of IE.9
MVAs are extremely rare, usually associated with IE of the aortic valve, and the incidence is between 0.2 and 0.3% on echocardiography in general.6 There are several mechanisms of MVA formation: in the presence of aortic valve IE, the jet lesion may result in secondary destruction of the mitral valve due to: A) the jet damaging the endothelial surface of the mitral valve, B) retrograde dissemination of bacteria, or C) the presence of neovessels (prominent in AMVL) which results in localized inflammation, valvulitis, protrusion of weakened MV into the left atrium cavity, and subsequently aneurysmal formation.3,5,10 Respect retrograde dissemination might result from 1) direct contact between the aortic vegetation and the AMVL during diastole, known as «mitral kissing vegetations» when they exceed 6 mm in length, 2) secondary infection of the damaged endothelium by bacteria from regurgitation blood flow, or 3) local spread of the infection through the mitral-aortic intervalvular fibrosa.5
Approximately two-thirds of MVAs rupture or perforate; the size of the aneurysm does not correlate with the risk of perforation; the AMVL is much more commonly involved than the posterior leaflet for unknown reasons.5
The echocardiographic appearance of MVA is characterized by a saccular bulge of the mitral leaflets that extends into the left atrium during systole and collapses during diastole.5,11 Other echocardiographic features vary from small saccular bulges, often challenging to identify due to vegetation, to large leaflet protrusions towards the left atrium, which may be associated with various degrees of mitral regurgitation and thrombosis.12 Among the differential diagnoses of MVA are mitral valve diverticulum, blood cysts of the papillary muscle, cardiac masses, chordal rupture, non-bacterial thrombotic endocarditis, mitral valve prolapse, flailing mitral leaflets, myxomatous degeneration, and infective vegetations. The color flow Doppler can support a correct diagnosis. A high-velocity regurgitant jet and direct communication between the aneurysm and the left ventricle support the diagnosis of a perforated aneurysm.3,5
Abscess, pseudoaneurysm, and formation of valve aneurysm in a patient with IE indicate uncontrolled infection and the need for urgent cardiac surgery (within seven days), except if there is severe co-morbidity. In other cases, the surgery can be postponed for one or two weeks while the patient receives antibiotic treatment under careful observation to allow the infected tissue to recover and heal and avoid unnecessary extensive surgical procedures.6,11,13
Streptococcus gordonii IE has been reported in 27 patients worldwide (Table 1), most male (74%). The median age was 48 (range 11-83 years), and fever was the most common symptom; 66% of patients experienced embolisms, 50% had to undergo valve replacement or repair surgery, and 13.6% died. In most cases, IE was diagnosed by TEE. The most common valve affection was the isolated native mitral valve (42%), followed by native mitro-aortic compromise (27%) and the isolated native aortic valve (23%). 94% of IE cases featured vegetations (mean diameter 11 mm), 27% a valve perforation, and 16.6% a valve aneurysm (the most common of both was the anterior mitral leaflet). Among patients who presented a perforated MVA with valve perforation, mortality was the highest.
Table 1: Reported cases of infective endocarditis due to Streptococcus gordonii.
| Author | Country | Pt. no. | Year | Age/gender | Symptoms (time) | Valve | Embolism | Diagnostic modality | Vegetation (valve/number/size) | Aneurysm (valve) | Perforation (valve) | AR | MR | Antibiotic treatment (duration) | Surgery | Outcome |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Tomsic et al.(6) | United States | 1 | 2016 | 48/M | Fever, Fatigue, cough (2 months) | Bicuspid AV, MV | Yes | TTE, TEE | AV/1/< 10 mm, MV/NR/NR | Yes (NCC AV, anterior MVL) | Yes (anterior MVL) | Severe | Severe | IV penicilin (6 wk), IV gentamicin (2 wk) | AVR, MVR | Cure |
| Baca et al.(7) | Peru | 2 | 2017 | 58/M | Fever, vomiting, low back pain (2 months) | AV, MV | Yes | TEE | AV/multiples/9 mm, MV/NR/NR | Yes (anterior MVL) | Yes (NCC AV) | Severe | Severe | NR | AVR, MVR | NR |
| Dadon et al.(14) | Israel | 3 | 1998 | 23/F | Fever (NR) | MV | NR | TEE | NR | NR | NR | NR | NR | IV gentamicin (2 wk) | None | Cure |
| 4 | 2006 | 37/M | Dyspnea (NR) | AV | NR | TTE | NR | NR | NR | NR | NR | NR | AVR | Died | ||
| 5 | 2006 | 45/M | Fever (NR) | MV | NR | TEE | NR | NR | NR | NR | NR | IV penicilin (6 wk), gentamicin (1 wk) | MVR | Cure | ||
| 6 | 2007 | 75/M | Fever (NR) | MV, AV | NR | TEE | NR | NR | NR | NR | NR | IV penicilin (6 wk) | No | Cure | ||
| 7 | 2013 | 83/F | Fever (NR) | MV | NR | TEE | NR | NR | NR | NR | NR | IV ceftriaxone (6 wk) | No | Cure | ||
| 8 | 2014 | 78/M | Fever (NR) | MV, AV | NR | TEE | NR | NR | NR | NR | NR | IV penicilin (6 wk) | AVR, MVR | Cure | ||
| 9 | 2014 | 71/M | Fever (NR) | NR | NR | TEE | NR | NR | NR | NR | NR | IV penicilin (6 wk) | No | Cure | ||
| 10 | 2015 | 31/M | Fever (NR) | MV | NR | TTE | NR | NR | NR | NR | NR | IV penicilin (4 wk) | MVR | Cure | ||
| 11 | 2016 | 82/M | Low back pain (2 wk) | MV | Yes | TEE | MV/1/14 mm | None | None | None | None | IV penicilin (8 wk) | None | Cure | ||
| 12 | 2016 | 63/M | Fever, low back pain (2 wk) | AV | Yes | TEE | AV/1/13 mm | None | None | None | None | IV penicilin (8 wk), IV gentamicin (2 wk), oral amoxycyline (4 wk) | None | Cure | ||
| 13 | 2017 | 71/M | General deterioration (NR) | AV | NR | TEE | NR | NR | NR | NR | NR | IV penicilin (6 wk) | AVR, MVR | Cure | ||
| Callejo-Goena et al.(15) | Spain | 14 | 2018 | 60/M | Fever (2 wk) | MV | Yes | TEE | MV/1/NR | None | None | None | Severe | Ceftriaxone (3 months) | None | Cure |
| Mosailova et al.(1) | United States | 15 | 2019 | 31/M | Bilateral lower extremity edema (2 days) | MV | Yes | TTE | MV/1/NR | None | None | None | Severe | IV ceftriaxone (6 wk) | MVR | NR |
| Komorovsky et al.(16) | Ukraine | 16 | 2019 | 11/F | Fever, weakness, joint pain (NR) | MV | None | NR | MV/1/6 mm | None | None | None | Moderate | NR | MV annuloplasty | Cure |
| Peechakara et al.(17) | India | 17 | 2019 | 42/M | Fever, back pain, hemoptysis, joint pain (3 months) | AV, MV | None | TTE, TEE | AV/NR/NR | None | Yes (anterior MVL) | Moderate | NR | IV ceftriaxone (6 wk), IV gentamicin (2 wk) | None | NR |
| Quan Li et al.(18) | China | 18 | 2020 | 36/M | Fever (2 months) | Bicuspid AV | None | TTE, TEE | AV/ NR/NR | None | None | Severe | NR | NR | AVR | Cure |
| Yue Wang et al.(19) | China | 19 | 2020 | 39/F | Fever, headache (6 months) | MV | Yes | TEE | MV/multiples/NR | None | None | None | None | IV vancomycin (12 days) | None | Died |
| Bridwell et al.(20) | United States | 20 | 2020 | 63/M | Fever (3 wk) | MV | None | TEE | MV/NR/NR | None | None | None | NR | IV gentamicin (2 wk), IV penicilin (4 wk) | None | Cure |
| Arbune et al.(21) | Romania | 21 | 2021 | 23/F | Fever, malaise (9 wk) | AV | None | TTE | AV/1/5 mm | None | None | NR | NR | IV ceftriaxone (6 wk) | None | Cure |
| Jiménez Melo et al.(22) | Spain | 22 | 2021 | 76/F | Dyspnea (4 months) | Mechanical MV and AV | Yes | TEE, FDG-PET/CT | MV/2/7 mm, AV/1/NR | None | None | None | None | IV ceftriaxone (NR) | None | NR |
| Chang et al.(4) | Malaysia | 23 | 2021 | 28/M | Fever (1 wk), abdominal pain (3 days) | Bicuspid AV, MV | Yes | TTE | AV/2/11 × 6 mm, 16 × 8 mm, MV/1/5 × 8 mm | None | None | Severe | Moderate | IV penicilin (6 wk) | NR | NR |
| Lim et al.(23) | Malaysia | 24 | 2022 | 40/M | Fever (1 month) | PV | Yes | TTE, CT | PV/1/23 × 12 mm | None | None | None | None | IV penicilin (6 wk) | PVR | Cure |
| Chawla et al.(24) | United States | 25 | 2023 | 27/F | Sudden loss of vision, Severe headache (4 days) | MV | Yes | TEE | MV/NR/NR | None | None | None | NR | IV ceftriaxone (6 wk) | None | Cure |
| Qu Yi-Fan et al.(25) | China | 26 | 2023 | 61/M | Fever, malaise (4 wk) | Bicuspid AV | Yes | TEE | None | None | Yes (AV) | Moderate-severe | Mild | NR | AVR, AV valvuloplasty | Cure |
| Alanís-Naranjo et al. (present case) | Mexico | 27 | 2023 | 60/M | Fever, weight loss (3 wk) | AV, MV | None | TEE | MV/1/11 × 5 mm | Yes (anterior MVL) | Yes (NCC AV, anterior MVL) | Severe | Moderate | IV ceftriaxone (1 month) | AVR, MVR | Died |
AR = aortic regurgitation. AV = aortic valve. AVR = aortic valve replacement. CT = computed tomography. F = female. FDG-PET/CT = F-fluorodeoxyglucose positron emission tomography/computed tomography. IV = intravenous. M = male. MR = mitral regurgitation. MV = mitral valve. MVL = mitral valve leaflet. MVR = mitral valve replacement. NCC = noncoronary cusp. NR = not reported. Pt = patient. PV = pulmonary valve. PVR = pulmonary valve replacement.
TEE = trans-esophageal echocardiogram. TTE = trans-thoracic echocardiogram.
Conclusions
Streptococcus gordonii is considered a commensal of the oral cavity and a non-pathogenic bacterium, but it could be an opportunistic pathogen and cause various infectious diseases. A perforated MVA with AV perforation is a rare but life-threatening complication of IE, even rarer in Streptococcus gordonii-related IE. In order to identify these complications, TEE is the method of choice that allows for more accurate morphological characterization of the tissue. All patients with streptococcal BSIs with a high risk of IE should be evaluated with an echocardiogram to rule out IE and its complications and offer prompt surgical intervention if necessary.











 nueva página del texto (beta)
nueva página del texto (beta)



